Abstract
Pneumocystis jirovecii pneumonia (PJP) is a potentially life-threatening infection that occurs in immunocompromised patients. The aim of this study was to evaluate risk factors for PJP in kidney transplantation recipients. We conducted a retrospective analysis of patient data from 500 consecutive kidney transplants performed at Severance Hospital between April 2011 and April 2014. Eighteen kidney transplantation recipients (3.6%) were diagnosed with PJP. In the univariate analysis, acute graft rejection, CMV infection, use of medication for diabetes mellitus, and lowest lymphocyte count were associated with PJP. Recipients who experienced acute graft rejection (odds ratio [OR] 11.81, 95% confidence interval [CI] 3.06–45.57, P < 0.001) or developed CMV infection (OR 5.42, 95% CI 1.69–17.39, P = 0.005) had high odds of PJP in multivariate analysis. In the acute graft rejection subgroup, patients treated with anti-thymocyte globulin (ATG) had significantly higher odds of PJP (OR 5.25, 95% CI 1.01–27.36, P = 0.006) than those who were not. Our data suggest that acute graft rejection and CMV infection may be risk factors for PJP in kidney transplant patients. The use of ATG for acute graft rejection may increase the risk of PJP.
Similar content being viewed by others
Introduction
Pneumocystis jirovecii is an opportunistic fungal pathogen1. Pneumocystis jirovecii pneumonia (PJP) previously known as Pneumocystis carinii pneumonia is a potentially life-threatening infection that occurs in immunocompromised patients1, 2. In the absence of prophylaxis, PJP occurs in approximately 5–15% of transplant patients, depending on the transplanted organ or transplant center2, 3.
In kidney transplantation, PJP is a very serious risk factor for graft loss and patient mortality4, 5. In the absence of appropriate treatment, the mortality rate of PJP is 90–100%, and can be as high as 50% despite adequate therapy6, 7. Therefore, several guidelines—such as the Kidney Disease Improving Global Outcomes (KDIGO) guideline, the European Renal Best Practice guideline, and other reports— usually recommend PJP prophylaxis by using TMP/SMX for 3–6 months after renal transplantation8,9,10. The incidence rate of PJP has decreased with the use of prophylaxis, however, an increasing number of PJP outbreaks in kidney transplant centers has been reported worldwide in recent years4, 5, 11. The causes of those outbreaks have not fully been evaluated.
Risk factors for the development of PJP in kidney transplant patients are still not confirmed. The overall load of immunosuppressive therapy, higher donor age, higher recipient age, lymphopenia, previous cytomegalovirus (CMV) infection, or treatment used for episodes of graft rejection have been reported as risk factors for PJP in kidney transplant patients12,13,14,15,16,17. However, factors identified in some studies are not always confirmed in other studies. Thus, further research is needed to evaluate the risk factors for PJP in kidney transplant patients in the era of routine PJP prophylaxis. The aim of this study was to evaluate the risk factors for PJP in kidney transplantation recipients.
Patients and Methods
Study design and population
This single center, retrospective clinical study included all kidney transplant patients aged ≥18 years who underwent kidney transplantation at the Severance Hospital, a 2000-bed university tertiary referral hospital in South Korea, from April 2011 to April 2014. During this period, 500 patients underwent kidney transplantation; they were followed up until October 2015. We divided the patients into two groups according to the occurrence of PJP—the case group developed PJP whereas the control group did not—and then compared the groups. In our center, all kidney transplant recipients receive PJP prophylaxis using trimethoprim/sulfamethoxazole (TMP/SMX) 160/800 mg per day for 12 months post-transplantation. After transplantation, patients received a tacrolimus-based combination regimen or a cyclosporine-based combination regimen for maintaining immunosuppression.
Data collection
Data from all kidney transplant recipients were collected from the hospital’s electronic medical records. Clinical data on mortality, development of PJP, demographic characteristics, graft origin (deceased vs. living), immunosuppressive regimen, data about acute graft rejection such as frequency of such episodes or treatment received, smoking status, history of infections including CMV, BK virus, hepatitis, tuberculosis (TB), and comorbidities were evaluated.
Definition
We defined PJP when the following two conditions were satisfied; first, a positive result on Pneumocystis jirovecii real-time polymerase chain reaction (PCR) testing or direct immunofluorescence testing of microbiological samples (sputum, tracheal aspirate, bronchial washing fluid or bronchoalveolar lavage fluid) and second, identification of lung infiltration on chest computed tomography (CT)18,19,20.
CMV infection was defined as a fourfold elevation of the CMV PCR titer and the use of CMV medication (ganciclovir or valganciclovir). Smoking status was categorized as ever smoker or never smoked; the latter category included those who smoked fewer than 100 cigarettes in their lifetime. Development of acute graft rejection was established by biopsy of the transplanted kidney. Steroid pulse therapy (methylprednisolone [500 mg/day × 4 for 5 days]) was considered the first line therapy for acute graft rejection. When there was an inadequate response to steroid pulse therapy, an anti-thyomcyte globulin (ATG) was used. Lowest lymphocyte count was reviewed during follow-up in the PJP negative group after first hospital discharge and within 1 month before diagnosis of PJP in the PJP group. Clinical characteristics and events were reviewed until the last follow-up date in the PJP negative group and were reviewed until the date of PJP development in the PJP group.
Ethical approval
The study protocol was approved by the Institutional Review Board (IRB) of Severance Hospital (IRB number: 4-2015-1051). All methods were performed in accordance with the relevant guidelines and regulations. Informed consent was waived by the IRB because of the study’s retrospective nature.
Statistical analysis
Statistical analysis was performed using SPSS version 20 (IBM, Armonk, New York, USA). Data are described as medians (interquartile range [IQR]) or numbers (percentages). The Chi-squared and Fisher’s exact test or the Mann-Whitney test was used to assess differences between two groups. The independent risk for PJP was identified using logistic regression modeling. The fundamental variable (such as age, gender, BMI or kidney transplantation type) and variable which were meaningful risk factors in univariate analysis were used in multivariate analysis. A two-tailed P-value < 0.05 was considered statistically significant.
Results
Characteristics of the overall study population and patients diagnosed with PJP
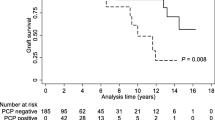
Demographic and clinical characteristics of the total study population are presented in Table 1. Overall, 500 patients were enrolled over the 3-year study period, with a similar number enrolled each year. The median age of all recipients was 47 years (range, 18–71) and men accounted for 61.4%. The major cause of kidney transplantation was hypertension (39.6%); the other cause was diabetes mellitus (17.0%). Transplanted kidneys were sourced more often from living than deceased donors (64.4% living vs. 35.6% deceased). In terms of immunosuppressive medication, 440 (88%) patients received a tacrolimus-based combination regimen and 60 (12%) received a cyclosporine-based combination regimen. The median duration of follow up was 36.2 month (range, 18.4–54.6).
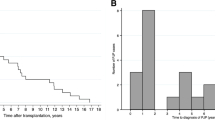
Eighteen patients were diagnosed with PJP during the study period, and most cases developed 12–24 months after kidney transplantation. All patients diagnosed with PJP were started on TMP/SMX. Five patients required second-line medication because of disease progression despite TMP-SMX use; four were treated with primaquine and clindamycin, while one was treated with pentamidine. Twelve (66.7%) patients with PJP improved after treatment, with no sequelae. However, three (16.7%) patients had residual sequelae and three (16.7%) died. Table 2 describes clinical characteristics and prognosis of PJP patients.
Risk factors for PJP in the total population
Table 3 shows the risk factors for PJP. In the univariate analysis, more men than women developed PJP, although this was not statistically significant (60.6% vs. 83.3%, respectively, P = 0.052). Median age, duration of follow up, graft source (deceased vs. living donor), proportion of re-transplant patients, and smoking status did not differ significantly between those who had PJP and those who did not. The underlying cause of kidney disease, necessitating transplantation, varied. However, hypertension, diabetes mellitus (DM), and IgA nephropathy were the major causes in both groups (P = 0.102). More acute graft rejection after transplantation wwere observed in the PJP group compared with the PJP negative group (83.3% vs. 20.1%, respectively, P < 0.001). And CMV infection was more common in the PJP group than the PJP negative group (61.1% vs. 12.9%, respectively, P < 0.001). Acute rejection and CMV infection were checked until PJP pneumonia in PJP group. Among those patients in the PJP group, 50% used medication for DM, compared to 26.1% in the PJP negative group (P = 0.032). Lowest lymphocyte counts were lower in the PJP group than in the control group (0.3 × 103/µL vs. 0.7 × 103/µL, P = 0.007). In terms of immunosuppressive agents, patients received either tacrolimus- or cyclosporine-based regimens; the regimen used did not differ between the PJP group and the control group (P = 0.710). In the multivariate analysis, acute graft rejection [odds ratio (OR), 11.81; 95% confidence interval (CI), 3.06–45.57] and CMV infection (OR, 5.42; 95% CI, 1.69–17.93) were associated with PJP, whereas sex, age, body mass index (BMI), and graft source were not.
Acute graft rejection and CMV infection were important risk factors for PJP, as presented in Table 3. To understand the impact of acute graft rejection and CMV pneumonia on PJP, we investigated the timing of the onset of PJP after acute graft rejection or CMV infection. The median interval between acute graft rejection and PJP was 6.1 (IQR, 3.3–12.9) months, while the median interval between CMV infection and PJP was 1.5 (IQR, 0.1–7.1) months (Fig. 1a and b).
Risk factors for PJP in patients experiencing acute graft rejection
As shown in Table 3, most cases of PJP (83.3%) occurred after acute graft rejection. Hence, we performed a subgroup analysis of the risk factors for PJP in patients who developed acute graft rejection (Table 4). Of the 500 kidney transplant patients, 112 experienced acute graft rejection. Of those, 15 were diagnosed with PJP. Sex, median age, BMI, graft source, smoking status, BK virus infection, use of DM medication, development of acute graft rejection within 1-year post-transplant, number of acute graft rejection episodes, and lowest lymphocyte count did not differ between patients who developed PJP and those who did not. All patients who developed acute graft rejection were managed with steroid pulse therapy. However, additional use of ATG for acute graft rejection treatment was more used in the PJP group (73.3% vs. 39.2%, P = 0.013). In addition, the proportion of patients with CMV infection was higher in the PJP group than in the PJP negative group (66.7% vs. 34%, respectively, P = 0.016). In the multivariate analysis, the independent risk factors for PJP in the acute graft rejection subgroup were identified as male sex (OR, 6.45; 95% CI, 1.24–33.73) and the use of ATG (OR, 5.25; 95% CI, 1.01–27.36).
Discussion
In our study, of 500 kidney transplant patients, 18 developed PJP. Acute graft rejection and CMV infection were identified as risk factors for PJP. Within the subgroup of patients who experienced acute graft rejection, male sex and the use of ATG were risk factors for PJP.
Our data showed that more patients in the PJP group than in the PJP negative group had CMV infection. This result is in agreement with that of previous studies17, 21. CMV is known to modify host immune responses by various mechanisms, suppressing helper T-cell and antigen presenting cell functions22. Therefore, CMV infection is both a marker of an immunocompromised state and has, itself, an immunosuppressive effect23. Therefore, when one opportunistic infection is diagnosed in transplant patients, physicians should consider that patients may at the same time or in the following weeks/months present with another opportunistic infection.
Several articles have reported that the development of acute graft rejection is a risk factor for PJP10, 13, 15, 17. Our data were similar. When acute graft rejection occurs, patients are treated with steroid pulse therapy or other immunosuppressive agents. These results support the hypothesis that severe immunosuppression may increase the occurrence of PJP. Similarly, low lymphocyte counts have been reported to be associated with the development of PJP12. Our data showed that the median lowest lymphocyte count was relatively low in the PJP group. Although this result was not evident in multivariate analysis, the univariate analysis still suggests the importance of lymphocytes in the development of PJP infection.
Previous studies have reported other risk factors for PJP in patients who have undergone kidney transplantation3, 15, 24. Lufft et al. first reported that different immunosuppressive regimens could affect the occurrence of PJP in renal transplant recipients24. In their report, tacrolimus-based regimens seemed more likely to trigger PJP, but this was not confirmed. MMF was suggested for use as an immunosuppressive agent having anti-PJP effects25, 26. In our study, there was no difference in the occurrence of PJP between the groups of patients receiving tacrolimus- or cyclosporine-based regimens. We could not analyze the effect of MMF on PJP because kidney transplant recipients at our center almost used MMF. Further research on the effect of immunosuppressive regimens on development of PJP is needed.
We performed further evaluation of patients (n = 112) who experienced acute graft rejection. Arend et al. reported that the incidence of PJP among patients treated for 0, 1, 2, or ≥3 rejection episodes increased with the increasing number of rejection episodes. The proportion of patients requiring additional ATG for an episode of rejection was higher in the PJP positive group than in the control group27. In our study, there was no association between the number of acute graft rejection episodes and PJP. Only additional use of ATG for treatment of acute graft rejection was associated with the development of PJP in the acute graft rejection subgroup. PJP occurred in four patients whilst taking PJP prophylaxis, three of whom had used additional ATG treatment prior to developing PJP. These results suggest that immunosuppressive status is more important than other factors in the development of PJP.
All guidelines recommend PJP prophylaxis for a certain period after kidney transplantation, although no universal consensus exists on the optimal duration of prophylaxis8,9,10. Our data indicated that acute graft rejection, CMV infection, and ATG use in the acute graft rejection subgroup could be the risk factors for PJP. Thus, further PJP prophylaxis is suggested in patients who have these risk factors, based on our data. In terms of the duration of prophylaxis, most cases of PJP occurring after acute graft rejection occurred within 12 months, and most cases of PJP occurring after an episode of CMV infection occurred within 6 months. Hence, we suggest a 6–12 month course of prophylaxis following either an episode of acute graft rejection or CMV infection. Although some complications have been reported with the use of TMP/SMX for PJP prophylaxis, major complications are relatively rare28; therefore, the benefits of PJP prophylaxis outweigh the risks.
There are several limitations to our study. First, we used the results of PCR as well as direct immunofluorescence to diagnose PJP. Thus, our data might include patients with false positive results. In general, confirmation of PJP requires special microbiological stains of sputum or bronchoalveolar lavage (BAL) specimens29. However, the proportion of PJP cases in HIV-uninfected patients in which is confirmed as organism in respiratory specimens is relatively low (compared with HIV-infected patients) because of the low burden of organism30,31,32. Pneumocystis jirovecii PCR assays are sensitive and increase the diagnostic yield in HIV-uninfected immunocompromised patients33,34,35. We used chest CT findings at initial diagnosis of PJP to exclude patients with false positive Pneumocystis jirovecii PCR results. Second, the number of PJP patients in our study was relatively small. However, unlike other studies, our study collected data on a large number of consecutive kidney transplant patients, instead of selecting particular patients; this point can be its strength. Third, patients with CMV infection and patients who used ATG overlapped in the acute graft rejection subgroup. Thus, we could not analyze the independent effect of CMV infection or ATG use on PJP. Fourth, this study was retrospective, thus, we could not systemically analyze multiple laboratory test results. There is a need for further, prospective studies to assess the risk factors for PJP.
Conclusion
Our data suggest that acute graft rejection and CMV infection may be risk factors for PJP in kidney transplant patients. In patients who develop acute graft rejection, the use of ATG may increase the risk of PJP. We suggest providing further PJP prophylaxis for kidney transplant patients who develop CMV infection or acute graft rejection, especially for those treated with ATG.
References
Thomas, C. F. Jr. & Limper, A. H. Pneumocystis pneumonia. N Engl J Med 350, 2487–2498, doi:10.1056/NEJMra032588 (2004).
Sepkowitz, K. A., Brown, A. E. & Armstrong, D. Pneumocystis carinii pneumonia without acquired immunodeficiency syndrome. More patients, same risk. Arch Intern Med 155, 1125–1128, doi:10.1001/archinte.1995.00430110015002 (1995).
Radisic, M. et al. Risk factors for Pneumocystis carinii pneumonia in kidney transplant recipients: a case-control study. Transpl Infect Dis 5, 84–93, doi:10.1034/j.1399-3062.2003.00018.x (2003).
Coyle, P. V. et al. Rising incidence of Pneumocystis jirovecii pneumonia suggests iatrogenic exposure of immune-compromised patients may be becoming a significant problem. J Med Microbiol 61, 1009–1015, doi:10.1099/jmm.0.043984-0 (2012).
Gordon, S. M. et al. Should prophylaxis for Pneumocystis carinii pneumonia in solid organ transplant recipients ever be discontinued? Clin Infect Dis 28, 240–246, doi:10.1086/cid.1999.28.issue-2 (1999).
Festic, E., Gajic, O., Limper, A. H. & Aksamit, T. R. Acute respiratory failure due to pneumocystis pneumonia in patients without human immunodeficiency virus infection: outcome and associated features. Chest 128, 573–579, doi:10.1378/chest.128.2.573 (2005).
Sepkowitz, K. A. Opportunistic infections in patients with and patients without Acquired Immunodeficiency Syndrome. Clin Infect Dis 34, 1098–1107, doi:10.1086/cid.2002.34.issue-8 (2002).
Kasiske, B. L. et al. KDIGO clinical practice guideline for the care of kidney transplant recipients: a summary. Kidney Int 77, 299–311, doi:10.1038/ki.2009.377 (2010).
European best practice guidelines for renal transplantation. Section IV: Long-term management of the transplant recipient. IV.7.1 Late infections. Pneumocystis carinii pneumonia. Nephrol Dial Transplant 17 Suppl 4, 36–39 (2002).
Martin, S. I. & Fishman, J. A. Pneumocystis pneumonia in solid organ transplantation. Am J Transplant 13(Suppl 4), 272–279, doi:10.1111/ajt.2013.13.issue-s4 (2013).
Higgins, R. M., Bloom, S. L., Hopkin, J. M. & Morris, P. J. The risks and benefits of low-dose cotrimoxazole prophylaxis for Pneumocystis pneumonia in renal transplantation. Transplantation 47, 558–560, doi:10.1097/00007890-198903000-00032 (1989).
Struijk, G. H. et al. Risk of Pneumocystis jiroveci pneumonia in patients long after renal transplantation. Nephrol Dial Transplant 26, 3391–3398, doi:10.1093/ndt/gfr048 (2011).
Eitner, F. et al. Risk factors for Pneumocystis jiroveci pneumonia (PcP) in renal transplant recipients. Nephrol Dial Transplant 26, 2013–2017, doi:10.1093/ndt/gfq689 (2011).
Goto, N. & Oka, S. Pneumocystis jirovecii pneumonia in kidney transplantation. Transpl Infect Dis 13, 551–558, doi:10.1111/tid.2011.13.issue-6 (2011).
Neff, R. T. et al. Analysis of USRDS: incidence and risk factors for Pneumocystis jiroveci pneumonia. Transplantation 88, 135–141, doi:10.1097/TP.0b013e3181aad256 (2009).
Arichi, N. et al. Cluster outbreak of Pneumocystis pneumonia among kidney transplant patients within a single center. Transplant Proc 41, 170–172, doi:10.1016/j.transproceed.2008.10.027 (2009).
de Boer, M. G. et al. Risk factors for Pneumocystis jirovecii pneumonia in kidney transplant recipients and appraisal of strategies for selective use of chemoprophylaxis. Transpl Infect Dis 13, 559–569, doi:10.1111/tid.2011.13.issue-6 (2011).
Thomas, C. F. Jr. & Limper, A. H. Current insights into the biology and pathogenesis of Pneumocystis pneumonia. Nat Rev Microbiol 5, 298–308, doi:10.1038/nrmicro1621 (2007).
Alanio, A. et al. Real-time PCR assay-based strategy for differentiation between active Pneumocystis jirovecii pneumonia and colonization in immunocompromised patients. Clin Microbiol Infect 17, 1531–1537, doi:10.1111/j.1469-0691.2010.03400.x (2011).
Desoubeaux, G. et al. Use of calcofluor-blue brightener for the diagnosis of Pneumocystis jirovecii pneumonia in bronchial-alveolar lavage fluids: A single-center prospective study. Med Mycol, doi:10.1093/mmy/myw068. (2016).
Borstnar, S. et al. Pneumocystis jirovecii pneumonia in renal transplant recipients: a national center experience. Transplant Proc 45, 1614–1617, doi:10.1016/j.transproceed.2013.02.107 (2013).
Muhammad Iqbal, A. H. et al. Pneumocystis jirovecii pneumonia 13 years post renal transplant following a recurrent cytomegalovirus infection. Transpl Infect Dis 14, E23–26, doi:10.1111/tid.2012.14.issue-4 (2012).
Harari, A., Zimmerli, S. C. & Pantaleo, G. Cytomegalovirus (CMV)-specific cellular immune responses. Hum Immunol 65, 500–506, doi:10.1016/j.humimm.2004.02.012 (2004).
Lufft, V. et al. Incidence of Pneumocystis carinii pneumonia after renal transplantation. Impact of immunosuppression. Transplantation 62, 421–423, doi:10.1097/00007890-199608150-00022 (1996).
Martin, S. I. & Fishman, J. A. Pneumocystis pneumonia in solid organ transplant recipients. Am J Transplant 9(Suppl 4), S227–233, doi:10.1111/j.1600-6143.2009.02914.x (2009).
Oz, H. S. & Hughes, W. T. Novel anti-Pneumocystis carinii effects of the immunosuppressant mycophenolate mofetil in contrast to provocative effects of tacrolimus, sirolimus, and dexamethasone. J Infect Dis 175, 901–904, doi:10.1086/jid.1997.175.issue-4 (1997).
Arend, S. M. et al. Rejection treatment and cytomegalovirus infection as risk factors for Pneumocystis carinii pneumonia in renal transplant recipients. Clin Infect Dis 22, 920–925, doi:10.1093/clinids/22.6.920 (1996).
Mitsides, N. et al. Complications and outcomes of trimethoprim-sulphamethoxazole as chemoprophylaxis for pneumocystis pneumonia in renal transplant recipients. Nephrology (Carlton) 19, 157–163, doi:10.1111/nep.2014.19.issue-3 (2014).
Cregan, P. et al. Comparison of four methods for rapid detection of Pneumocystis carinii in respiratory specimens. J Clin Microbiol 28, 2432–2436 (1990).
Baughman, R. P. & Liming, J. D. Diagnostic strategies in Pneumocystis carinii pneumonia. Front Biosci 3, e1–12, doi:10.2741/A363 (1998).
Limper, A. H., Offord, K. P., Smith, T. F. & Martin, W. J. 2nd Pneumocystis carinii pneumonia. Differences in lung parasite number and inflammation in patients with and without AIDS. Am Rev Respir Dis 140, 1204–1209, doi:10.1164/ajrccm/140.5.1204 (1989).
Pagano, L. et al. Pneumocystis carinii pneumonia in patients with malignant haematological diseases: 10 years’ experience of infection in GIMEMA centres. Br J Haematol 117, 379–386, doi:10.1046/j.1365-2141.2002.03419.x (2002).
Azoulay, E. et al. Polymerase chain reaction for diagnosing pneumocystis pneumonia in non-HIV immunocompromised patients with pulmonary infiltrates. Chest 135, 655–661, doi:10.1378/chest.08-1309 (2009).
Calderon, E. J., Gutierrez-Rivero, S., Durand-Joly, I. & Dei-Cas, E. Pneumocystis infection in humans: diagnosis and treatment. Expert Rev Anti Infect Ther 8, 683–701, doi:10.1586/eri.10.42 (2010).
Jiancheng, W. et al. Screening Pneumocystis carinii pneumonia in non-HIV-infected immunocompromised patients using polymerase chain reaction. Diagn Microbiol Infect Dis 64, 396–401, doi:10.1016/j.diagmicrobio.2009.04.009 (2009).
Author information
Authors and Affiliations
Contributions
S.H. Lee: participated in data collection and analysis, wrote the manuscript. M.S. Park, Y.S. Kim, S.K. Kim, J. Chang: assisted in patient data assessment and supervision of the project. K.H. Huh, D.J. Joo, M.S. Kim, S.I. Kim, J.H. Lee, Y.S. Kim: provided patient data, participated in data and follow-up of patients. S.Y. Kim: performed study conception and design, final approval of the version to be published.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lee, S.H., Huh, K.H., Joo, D.J. et al. Risk factors for Pneumocystis jirovecii pneumonia (PJP) in kidney transplantation recipients. Sci Rep 7, 1571 (2017). https://doi.org/10.1038/s41598-017-01818-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-01818-w
- Springer Nature Limited
This article is cited by
-
A case of hypercalcemia from Pneumocystis jirovecii in an immunosuppressed non-HIV patient
BMC Pulmonary Medicine (2024)
-
Risk factors for Pneumocystis pneumonia with acute respiratory failure among kidney transplant recipients
BMC Nephrology (2023)
-
Comparison of early and late Pneumocystis jirovecii Pneumonia in kidney transplant patients: the Korean Organ Transplantation Registry (KOTRY) Study
Scientific Reports (2022)
-
Synergistic impact of pre-sensitization and delayed graft function on allograft rejection in deceased donor kidney transplantation
Scientific Reports (2021)
-
Subcutaneous phaeohyphomycosis caused by Hongkongmyces snookiorum in a kidney transplant patient: a case report
BMC Infectious Diseases (2020)