Abstract
This data set describes the long-term observation and morphological study of the eggs of the great black-headed gull Larus ichthyaetus in the gull nesting colonies on the islands of Lake Chany. Lake Chany is located in the Baraba forest-steppe of the West Siberian Plain, Russia, between the Ob and Irtish rivers. Lake Chany is protected by the Ramsar Convention on the Wetlands of International Importance, indicating that the lake is an important site for migratory birds, including L. ichthyaetus. This dataset contains the size and fate of all eggs, as well as the size of hatched chicks in 1164 observed L. ichthyaetus nests from 1993 to 2003.
Measurement(s) | size |
Technology Type(s) | Observation |
Factor Type(s) | egg and chick size |
Sample Characteristic - Organism | Larus ichthyaetus |
Sample Characteristic - Environment | lake |
Sample Characteristic - Location | Russia |
Similar content being viewed by others
Background & Summary
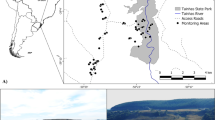
Lake Chany is located in the Baraba forest-steppe of the West Siberian Plain, Russia, between the Ob and Irtish Rivers (54°30′–55°09′ N, 76°48′–78°12′ E). The Lake Chany basin is among the largest in Russia, and the lake itself has the greatest water surface in West Siberia1. The waters, coastal zones, and islands of Lake Chany are favorable for breeding, molting, and feeding of more than 280 species of birds. In spring and autumn, numerous migratory waterbirds (over 300 000), such as swans, geese, ducks, coots, waders, gulls, terns, cranes, stop at the lake during migration1,2. Some of the species are rare and endangered and are listed in the Red Data Book of Russia and the Red List of the International Union for Conservation of Nature and Natural Resources (IUCN)3. Lake Chany is protected by the Ramsar Convention on Wetlands of International Importance, indicating that the lake is an important site for migratory birds, including L. ichthyaetus. Currently, most of the great black-headed gull colonies are in a vulnerable state; therefore, this species is recorded in the Red Data Books of the Russian Federation and Kazakhstan4,5. Therefore, it is important to observe the dynamics of their reproduction. Inter-annual changes in the onset of egg-laying and morphological parameters of eggs were pronounced during the years of our research and were probably determined by the course of spring and feeding conditions6,7. Egg size may be affected by food availability8,9. Some studies suggested a relationship between egg size and morphological traits, survival, and growth rate of chicks hatching from them [reviewed in10,11].
Here, we provide eight years of raw field data (1993, 1994, 1996–1998, 2001–2003) from our egg and chick morphological study on great black-headed gull Larus ichthyaetus breeding on islands of Lake Chany, Russia12.
Methods
We surveyed three islands of Lake Chany: Uzkoredkii (54° 58′ 15′′ N, 77°27′04′′ E), Reden’kii (54° 56′ 05′′ N, 77° 22′ 27′′ 52 E), Korablik (54° 59′ 31′′ N, 77° 40′ 38′′ E). The studied intertidal habitats are rarely reached by humans.
Gull nests were counted in colonies by regular surveys over eight years (1993, 1994, 1996–1998, 2001–2003) on the islands of Lake Chany. Colonies were visited daily or sometimes every other day. To minimize the disturbance caused by the investigation, the time spent working, within view of the gulls was restricted to a maximum of forty minutes per study plots. We noted nest content at every visit for the presence of eggs or chicks. In total, there were 1 164 nests under observation. Nests contained 1 (n = 140), 2 (n = 518), 3 (n = 504) or 4 (n = 2) eggs. Modal clutch size of the great black-headed gull is two or three eggs, varying seasonally. The length and width of the eggs were measured using Vernier calipers (division accuracy 0,1 mm) and numbered with a waterproof marker. Egg volumes were estimated using Hoyt’s equation: Volume = 0.51 * Length * Width * Width/100013. We determined the volume of 2117 great black-headed gull eggs.
As the laying of eggs has already started by the first visit to the colony, the date of the beginning of egg laying was calculated by subtracting the average length of the incubation period of great black-headed gulls (27 days) from the hatching date of first chick in the nest (n = 559 nests). If the hatching date was not known, the clutch initiation date was determined by subtracting the number of days of incubation from the date that the nest was first discovered (n = 469 nests). The stage of incubation was estimated from the change in position of an incubated egg placed in water14,15. The technique’s accuracy varied throughout incubation and mean prediction error fall between 0–4 days. On average, egg flotation estimated an embryo’s developmental age to within 1.9 ± 1.6 days (mean ± 1 SD)16. Only 47 nests were found during egg laying. Great black-headed gulls usually laid eggs at intervals of two days. Incubation started as soon as the first egg was laid, so eggs hatched asynchronously, one or two days apart.
Whenever possible, we determined the within-clutch laying sequence of eggs (1st, 2nd, 3rd, and 4th). A complete laying sequence was established by observation in 47 cases. In about 48% of clutches the position in laying sequence was established on the basis of the sequence of hatching. In other cases, if we could distinguish within-clutch distinct flotation levels of eggs, we numbered eggs according to the stage of incubation. Sometimes this technique for distinguishing egg laying order were used in other seabirds17,18.
We recorded the pipping date (i.e. appearance of star-like bursts) and the actual hatching date of the individual eggs. Wet chicks were registered as hatchlings of that day; dry chicks were registered as 1 day old. Chicks older than two days left the nest and moved to a location nearby. Newly hatched gull chicks were captured by hand at nests, ringed, and measured. We determined wing, tarsus, and head length using a ruler with zero-stop and vernier calipers and body weight measured using Pesola spring balances for 747 chicks of great black-headed gulls, and 457 of them hatched from eggs that were measured.
Data Records
The all data were deposited in Zenodo https://doi.org/10.5281/zenodo.517555112. The data name, unit and method for the data were described in Table 1.
Technical Validation
The long-term monitoring program for was the egg and chick size in the nests of Larus ichthyaetus designed by A.K.Y. A.K.Y. identified the nest and measured the values which we provided in the database. Then A.K.Y. recorded all data in an Excel file. deceased before preparing the data base and this paper. The other authors carefully prepared the data sheet with double checking by the authors.
Code availability
We did not use the custom code for the data management.
References
Yurlov, A. K. et al. Vertebrates of Chany Lake and surrounded area. Pages 162–203 in O.F. Vasilyev, J. Veen editors. Chany Lake (West Siberia) environmental profile. Academic Publishing House GEO, Novosibirsk 2015.
Veen, J. et al. An atlas of movements of Southwest Siberian waterbirds. Wetlands International, Wageningen, The Netherlands 2005.
IUCN. The IUCN Red List of Threatened Species. Version 2021-2. https://www.iucnredlist.org 2021.
Danilov-Danilyan, V. I. (ed.). Red Data Book of Russian Federation. Animals. Astrel, Moscow 2001.
The Red List of Republic of Kazakhstan. http://www.redbookkz.info.
Brommer, J. E., Rattiste, K. & Wilson, A. J. Exploring plasticity in the wild: laying date-temperature reaction norms in the common gull Larus canus. Proceedings of the Royal Society B: Biological Sciences 275(1635), 687–693, https://doi.org/10.1098/rspb.2007.0951 (2008).
Votier, S. C., Hatchwell, B. J., Mears, M. & Birkhead, T. R. Changes in the timing of egg-laying of a colonial seabird in relation to population size and environmental conditions. Marine Ecology Progress Series 393, 225–234, https://doi.org/10.3354/meps08258 (2009).
Reid, W. V. Constraints on clutch size in the Glaucous-winged Gull. Studies in Avian Biology 10, 8–25 (1987).
Salzer, D. W. & Larkin, G. J. Impact of courtship feeding on clutch and third-egg size in Glaucous-winged Gulls. Animal Behaviour 39, 1149–1162, https://doi.org/10.1016/S0003-3472(05)80787-0 (1990).
Williams, T. D. Intraspecific variation in egg size and egg composition in birds: effects on offspring fitness. Biological Reviews 69(1), 35–59, https://doi.org/10.1111/j.1469-185X.1994.tb01485.x (1994).
Krist, M. Egg size and offspring quality: a meta-analysis in birds. Biological reviews 86(3), 692–716, https://doi.org/10.1111/j.1469-185X.2010.00166.x (2010).
Yurlov, A. K., Yurlova, N. I., Garyushkina, M. Y., Selivanova, M. A. & Doi, H. Long-term observation of the egg and chick size in the nests of Larus ichthyaetus in Lake Chany,West Siberia, Russia. Zenodo https://doi.org/10.5281/zenodo.5175550 (2022).
Hoyt, D. F. Practical methods of estimating volume and fresh weight of bird eggs. Auk 96(1), 73–77, https://doi.org/10.1093/auk/96.1.73 (1979).
Westerskov, K. Methods for determining the age of game bird eggs. Journal of Wildlife Management 14, 56–67, https://doi.org/10.2307/3795978 (1950).
Van Paassen, A. G., Veldman, D. H. & Beintema, A. J. A simple device for determination of incubation stages in eggs. Wildfowl 35, 173–178 (1984).
Ackerman, J. T. & Eagles-Smith, C. A. Accuracy of egg flotation throughout incubation to determine embryo age and incubation day in waterbird nests. The Condor 112(3), 438–446, https://doi.org/10.1525/cond.2010.090070 (2010).
Nisbet, I. C. T. Selective effects of predation in a tern colony. The Condor 77(2), 221–226, https://doi.org/10.2307/1365803 (1975).
Gochfeld, M. Intraclutch egg variation: the uniqueness of the common tern’s third egg. Bird-Banding 48(4), 325–332 (1977).
Author information
Authors and Affiliations
Contributions
A.K.Y. Investigation, Conceptualisation, Project administration, Methodology N.I.Y. Funding acquisition, Supervision, Data Curation, Writing (Original Draft), Writing (Review & Editing) M.Y.G. Data Curation, Writing (Original Draft), Writing (Review & Editing) M.A.S. Data Curation, Writing (Original Draft), Writing (Review & Editing) H.D. Formal analysis, Writing (Original Draft), Writing (Review & Editing), Visualization.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Yurlov, A.K., Yurlova, N.I., Garyushkina, M.Y. et al. Long-term observation of the egg and chick size in the nests of Larus ichthyaetus in Lake Chany, Russia. Sci Data 9, 372 (2022). https://doi.org/10.1038/s41597-022-01454-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41597-022-01454-8
- Springer Nature Limited