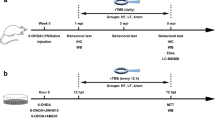
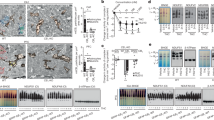
Abstract
Cannabinoids reduce tremor associated with motor disorders induced by injuries and neurodegenerative disease. Here we show that this effect is mediated by cannabinoid receptors on astrocytes in the ventral horn of the spinal cord, where alternating limb movements are initiated. We first demonstrate that tremor is reduced in a mouse model of essential tremor after intrathecal injection of the cannabinoid analog WIN55,212-2. We investigate the underlying mechanism using electrophysiological recordings in spinal cord slices and show that endocannabinoids released from depolarized interneurons activate astrocytic cannabinoid receptors, causing an increase in intracellular Ca2+, subsequent release of purines and inhibition of excitatory neurotransmission. Finally, we show that the anti-tremor action of WIN55,212-2 in the spinal cords of mice is suppressed after knocking out CB1 receptors in astrocytes. Our data suggest that cannabinoids reduce tremor via their action on spinal astrocytes.








Similar content being viewed by others
Data availability
Electrophysiological, imaging and tremor data are available at zenodo.org; https://doi.org/10.5281/zenodo.4478494. Data for immunohistochemistry are available from the corresponding author upon reasonable request. Source data are provided with this paper.
Code availability
Code written for data analysis of this study is available at https://github.com/perrierlab/Carlsen_et_al_2021.
References
Deuschl, G., Bain, P. & Brin, M. Consensus statement of the Movement Disorder Society on Tremor. Ad Hoc Scientific Committee. Mov. Disord. 13, 2–23 (1998).
Alty, J. E. & Kempster, P. A. A practical guide to the differential diagnosis of tremor. Postgrad. Med. J. 87, 623–629 (2011).
Biary, N., Cleeves, L., Findley, L. & Koller, W. Post-traumatic tremor. Neurology 39, 103–106 (1989).
Koch, M., Mostert, J., Heersema, D. & De Keyser, J. Tremor in multiple sclerosis. J. Neurol. 254, 133–145 (2007).
Rinker, J. R. 2nd et al. Prevalence and characteristics of tremor in the NARCOMS multiple sclerosis registry: a cross-sectional survey. BMJ Open 5, e006714 (2015).
Louis, E. D. Essential tremors: a family of neurodegenerative disorders? Arch. Neurol. 66, 1202–1208 (2009).
Consroe, P., Musty, R., Rein, J., Tillery, W. & Pertwee, R. The perceived effects of smoked cannabis on patients with multiple sclerosis. Eur. Neurol. 38, 44–48 (1997).
Clifford, D. B. Tetrahydrocannabinol for tremor in multiple sclerosis. Ann. Neurol. 13, 669–671 (1983).
Meinck, H. M., Schonle, P. W. & Conrad, B. Effect of cannabinoids on spasticity and ataxia in multiple sclerosis. J. Neurol. 236, 120–122 (1989).
Arjmand, S. et al. Cannabinoids and tremor induced by motor-related disorders: friend or foe? Neurotherapeutics 12, 778–787 (2015).
Baker, D. et al. Cannabinoids control spasticity and tremor in a multiple sclerosis model. Nature 404, 84–87 (2000).
Wilson, R. I. & Nicoll, R. A. Endogenous cannabinoids mediate retrograde signalling at hippocampal synapses. Nature 410, 588–592 (2001).
Kreitzer, A. C. & Regehr, W. G. Retrograde inhibition of presynaptic calcium influx by endogenous cannabinoids at excitatory synapses onto Purkinje cells. Neuron 29, 717–727 (2001).
Navarrete, M. & Araque, A. Endocannabinoids mediate neuron–astrocyte communication. Neuron 57, 883–893 (2008).
Navarrete, M. & Araque, A. Endocannabinoids potentiate synaptic transmission through stimulation of astrocytes. Neuron 68, 113–126 (2010).
Navarrete, M., Diez, A. & Araque, A. Astrocytes in endocannabinoid signalling. Philos. Trans. R. Soc. Lond. B Biol. Sci. 369, 20130599 (2014).
Metna-Laurent, M. & Marsicano, G. Rising stars: modulation of brain functions by astroglial type-1 cannabinoid receptors. Glia 63, 353–364 (2015).
Rodriguez, J. J., Mackie, K. & Pickel, V. M. Ultrastructural localization of the CB1 cannabinoid receptor in µ-opioid receptor patches of the rat caudate putamen nucleus. J. Neurosci. 21, 823–833 (2001).
Piomelli, D., Beltramo, M., Giuffrida, A. & Stella, N. Endogenous cannabinoid signaling. Neurobiol. Dis. 5, 462–473 (1998).
Kjaerulff, O. & Kiehn, O. Distribution of networks generating and coordinating locomotor activity in the neonatal rat spinal cord in vitro: a lesion study. J. Neurosci. 16, 5777–5794 (1996).
Llinas, R. & Volkind, R. A. The olivo-cerebellar system: functional properties as revealed by harmaline-induced tremor. Exp. Brain Res. 18, 69–87 (1973).
Carlsen, E. M. M., Amrutkar, D. V., Nielsen, K. S. & Perrier, J. F. Accurate and affordable assessment of physiological and pathological tremor in rodents with the accelerometer of a smartphone. J. Neurophysiol. 122, 970–974 (2019).
Handforth, A. Harmaline tremor: underlying mechanisms in a potential animal model of essential tremor. Tremor Other Hyperkinet. Mov. (N Y) 2, 02-92-769-1 (2012).
Sykova, E. & Nicholson, C. Diffusion in brain extracellular space. Physiol. Rev. 88, 1277–1340 (2008).
Wolf, D. A. et al. Dynamic dual-isotope molecular imaging elucidates principles for optimizing intrathecal drug delivery. JCI Insight 1, e85311 (2016).
McAuley, J. H. & Marsden, C. D. Physiological and pathological tremors and rhythmic central motor control. Brain 123, 1545–1567 (2000).
Chevaleyre, V., Takahashi, K. A. & Castillo, P. E. Endocannabinoid-mediated synaptic plasticity in the CNS. Annu Rev. Neurosci. 29, 37–76 (2006).
Araque, A., Castillo, P. E., Manzoni, O. J. & Tonini, R. Synaptic functions of endocannabinoid signaling in health and disease. Neuropharmacology 124, 13–24 (2017).
Zucker, R. S. & Regehr, W. G. Short-term synaptic plasticity. Annu. Rev. Physiol. 64, 355–405 (2002).
Oliveira da Cruz, J. F., Robin, L. M., Drago, F., Marsicano, G. & Metna-Laurent, M. Astroglial type-1 cannabinoid receptor (CB1): a new player in the tripartite synapse. Neuroscience 323, 35–42 (2016).
Gomez-Gonzalo, M. et al. Endocannabinoids induce lateral long-term potentiation of transmitter release by stimulation of gliotransmission. Cereb. Cortex 25, 3699–3712 (2015).
Nolte, C. et al. GFAP promoter-controlled EGFP-expressing transgenic mice: a tool to visualize astrocytes and astrogliosis in living brain tissue. Glia 33, 72–86 (2001).
Hegyi, Z. et al. CB1 receptor activation induces intracellular Ca2+ mobilization and 2-arachidonoylglycerol release in rodent spinal cord astrocytes. Sci. Rep. 8, 10562 (2018).
Lalo, U. et al. Exocytosis of ATP from astrocytes modulates phasic and tonic inhibition in the neocortex. PLoS Biol. 12, e1001747 (2014).
Carlsen, E. M. & Perrier, J. F. Purines released from astrocytes inhibit excitatory synaptic transmission in the ventral horn of the spinal cord. Front. Neural Circuits 8, 60 (2014).
Han, J. et al. Acute cannabinoids impair working memory through astroglial CB1 receptor modulation of hippocampal LTD. Cell 148, 1039–1050 (2012).
Broadhead, M. J. & Miles, G. B. Bi-directional communication between neurons and astrocytes modulates spinal motor circuits. Front. Cell. Neurosci. 14, 30 (2020).
Witts, E. C., Panetta, K. M. & Miles, G. B. Glial-derived adenosine modulates spinal motor networks in mice. J. Neurophysiol. 107, 1925–1934 (2012).
Robin, L. M. et al. Astroglial CB1 receptors determine synaptic d-serine availability to enable recognition memory. Neuron 98, 935–944 (2018).
Alger, B. E. Retrograde signaling in the regulation of synaptic transmission: focus on endocannabinoids. Prog. Neurobiol. 68, 247–286 (2002).
Williams, E. R. & Baker, S. N. Renshaw cell recurrent inhibition improves physiological tremor by reducing corticomuscular coupling at 10 Hz. J. Neurosci. 29, 6616–6624 (2009).
Marsicano, G. et al. CB1 cannabinoid receptors and on-demand defense against excitotoxicity. Science 302, 84–88 (2003).
Hirrlinger, P. G., Scheller, A., Braun, C., Hirrlinger, J. & Kirchhoff, F. Temporal control of gene recombination in astrocytes by transgenic expression of the tamoxifen-inducible DNA recombinase variant CreERT2. Glia 54, 11–20 (2006).
Acknowledgements
We thank F. Kirchhoff (University of Saarland) for providing the Tgn(hgFAPEGEP) mice, M. Beato (UCL) for his help with the adult spinal cord slice preparation, A. Fletcher-Jones and A. Eraso for technical support and L. Anson for helping edit the manuscript. The study was funded by the Offerfonden (to J.F.P.), the Aase og Ejnar Danielsens Fond (to E.M.C.), the Den Owensenske Fond (to J.F.P.), the Læge Sofus Carl Emil Friis og hustru Olga Doris Fond (to J.F.P.), the Danmarks Frie Forskningsfond (9039-00072B to J.F.P.), the European Research Council (grant MiCaBra, ERC-2017-AdG-786467 to G.M.), Region Nouvelle Aquitaine, Agence Nationale de la Recherche ANR, NeuroNutriSens (ANR-13-BSV4-0006 and ORUPS ANR-16-CE37-0010-01), BRAIN (ANR-10-LABX-0043, to G.M.) and the Fondation pour la Recherche Médicale (grant no. FRM SPF201809006908 to U.S.).
Author information
Authors and Affiliations
Contributions
E.M.C. and J.F.P. designed the project. E.M.C. performed the electrophysiological and imaging experiments. S.F. and E.M.C. performed the behavioral experiments. U.S., L.R. and A.P.Z. performed the validation of the transgenic model. G.M. and J.F.P. supervised the research. E.M.C. and J.F.P. wrote the paper. All authors approved the final version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Neuroscience thanks Alexander Gourine and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Fig. 1
Source data
Source Data Fig. 1
Statistical Source Data
Source Data Fig. 2
Statistical Source Data
Source Data Fig. 3
Statistical Source Data
Source Data Fig. 4
Statistical Source Data
Source Data Fig. 5
Statistical Source Data
Source Data Fig. 6
Statistical Source Data
Source Data Fig. 7
Statistical Source Data
Source Data Fig. 8
Statistical Source Data
Rights and permissions
About this article
Cite this article
Carlsen, E.M.M., Falk, S., Skupio, U. et al. Spinal astroglial cannabinoid receptors control pathological tremor. Nat Neurosci 24, 658–666 (2021). https://doi.org/10.1038/s41593-021-00818-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41593-021-00818-4
- Springer Nature America, Inc.