Abstract
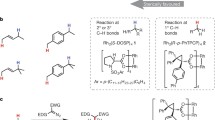
Carbon–hydrogen (C–H) bonds have long been considered unreactive and are inert to traditional chemical reagents, yet new methods for the transformation of these bonds are continually being developed1,2,3,4,5,6,7,8,9. However, it is challenging to achieve such transformations in a highly selective manner, especially if the C–H bonds are unactivated10 or not adjacent to a directing group11,12,13. Catalyst-controlled site-selectivity—in which the inherent reactivities of the substrates14 can be overcome by choosing an appropriate catalyst—is an appealing concept, and substantial effort has been made towards catalyst-controlled C–H functionalization6,15,16,17, in particular methylene C–H bond functionalization. However, although several new methods have targeted these bonds in cyclic alkanes, the selectivity has been relatively poor18,19,20. Here we illustrate an additional level of sophistication in catalyst-controlled C–H functionalization, whereby unactivated cyclohexane derivatives can be desymmetrized in a highly site- and stereoselective manner through donor/acceptor carbene insertion. These studies demonstrate the potential of catalyst-controlled site-selectivity to govern which C–H bond will react, which could enable new strategies for the production of fine chemicals.




Similar content being viewed by others
Data availability
Crystallographic data for the structures reported have been deposited at the Cambridge Crystallographic Data Centre, under deposition numbers CCDC 1855619, 1855620 and 1855295. Copies of the data can be obtained free of charge from www.ccdc.cam.ac.uk/data_request/cif. Complete experimental procedures and compound characterization data are available in the Supplementary Information, or from the corresponding author upon request.
References
Yamaguchi, J., Yamaguchi, A. D. & Itami, K. C–H bond functionalization: emerging synthetic tools for natural products and pharmaceuticals. Angew. Chem. Int. Ed. 51, 8960–9009 (2012).
Davies, H. M. L. & Morton, D. Recent advances in C–H functionalization. J. Org. Chem. 81, 343–350 (2016).
Gutekunst, W. R. & Baran, P. S. C–H functionalization logic in total synthesis. Chem. Soc. Rev. 40, 1976–1991 (2011).
Wencel-Delord, J. & Glorius, F. C–H bond activation enables the rapid construction and late-stage diversification of functional molecules. Nat. Chem. 5, 369–375 (2013).
Fier, P. S. & Hartwig, J. F. Synthesis and late-stage functionalization of complex molecules through C–H fluorination and nucleophilic aromatic substitution. J. Am. Chem. Soc. 136, 10139–10147 (2014).
Liao, K. et al. Site-selective and stereoselective functionalization of non-activated tertiary C–H bonds. Nature 551, 609–613 (2017).
McMurray, L., O’Hara, F. & Gaunt, M. J. Recent developments in natural product synthesis using metal-catalysed C–H bond functionalisation. Chem. Soc. Rev. 40, 1885–1898 (2011).
Newhouse, T. & Baran, P. S. If C–H bonds could talk: selective C–H bond oxidation. Angew. Chem. Int. Ed. 50, 3362–3374 (2011).
Davies, H. M. L. & Manning, J. R. Catalytic C–H functionalization by metal carbenoid and nitrenoid insertion. Nature 451, 417–424 (2008).
Davies, H. M. L., Hansen, T. & Churchill, M. R. Catalytic asymmetric C–H activation of alkanes and tetrahydrofuran. J. Am. Chem. Soc. 122, 3063–3070 (2000).
He, J., Wasa, M., Chan, K. S. L., Shao, Q. & Yu, J. Q. Palladium-catalyzed transformations of alkyl C–H bonds. Chem. Rev. 117, 8754–8786 (2017).
Colby, D. A., Bergman, R. G. & Ellman, J. A. Rhodium-catalyzed C–C bond formation via heteroatom-directed C–H bond activation. Chem. Rev. 110, 624–655 (2010).
Hartwig, J. F. & Larsen, M. A. Undirected, homogeneous C–H bond functionalization: challenges and opportunities. ACS Cent. Sci. 2, 281–292 (2016).
Davies, H. M. L. & Morton, D. Guiding principles for site selective and stereoselective intermolecular C–H functionalization by donor/acceptor rhodium carbenes. Chem. Soc. Rev. 40, 1857–1869 (2011).
Liao, K., Negretti, S., Musaev, D. G., Bacsa, J. & Davies, H. M. L. Site-selective and stereoselective functionalization of unactivated C–H bonds. Nature 533, 230–234 (2016).
Qin, C. et al. D 2-symmetric dirhodium catalyst derived from a 1,2,2-triarylcyclopropanecarboxylate ligand: design, synthesis and application. J. Am. Chem. Soc. 133, 19198–19204 (2011).
Qin, C. & Davies, H. M. L. Role of sterically demanding chiral dirhodium catalysts in site-selective C–H functionalization of activated primary C–H bonds. J. Am. Chem. Soc. 136, 9792–9796 (2014).
Chen, M. S. & White, M. C. Combined effects on selectivity in Fe-catalyzed methylene oxidation. Science 327, 566–571 (2010).
Czaplyski, W. L., Na, C. G. & Alexanian, E. J. C–H Xanthylation: a synthetic platform for alkane functionalization. J. Am. Chem. Soc. 138, 13854–13857 (2016).
Schmidt, V. A., Quinn, R. K., Brusoe, A. T. & Alexanian, E. J. Site-selective aliphatic C–H bromination using N-bromoamides and visible light. J. Am. Chem. Soc. 136, 14389–14392 (2014).
Quinn, R. K. et al. Site-selective aliphatic C–H chlorination using N-chloroamides enables a synthesis of chlorolissoclimide. J. Am. Chem. Soc. 138, 696–702 (2016).
Wasa, M. et al. Ligand-enabled methylene C(sp3)–H bond activation with a Pd(ii) catalyst. J. Am. Chem. Soc. 134, 18570–18572 (2012).
Chen, K., Eschenmoser, A. & Baran, P. S. Strain release in C–H bond activation? Angew. Chem. Int. Ed. 48, 9705–9708 (2009).
Dondi, D. et al. Regio- and stereoselectivity in the decatungstate photocatalyzed alkylation of alkenes by alkylcyclohexanes. Chem. Eur. J. 15, 7949–7957 (2009).
Liao, K. et al. Design of catalysts for site-selective and enantioselective functionalization of non-activated primary C–H bonds. Nat. Chem. 10, 1048–1055 (2018).
Guptill, D. M. & Davies, H. M. L. 2,2,2-Trichloroethyl aryldiazoacetates as robust reagents for the enantioselective C–H functionalization of methyl ethers. J. Am. Chem. Soc. 136, 17718–17721 (2014).
Saito, H. et al. Enantio- and diastereoselective synthesis of cis-2-aryl-3-methoxycarbonyl-2,3-dihydrobenzofurans via the Rh(ii)-catalyzed C–H insertion process. Org. Lett. 4, 3887–3890 (2002).
Kitagaki, S. et al. Enantiocontrol in tandem carbonyl ylide formation and intermolecular 1,3-dipolar cycloaddition of α-diazo ketones mediated by chiral dirhodium(ii) carboxylate catalyst. J. Am. Chem. Soc. 121, 1417–1418 (1999).
DeAngelis, A. et al. The chiral crown conformation in paddlewheel complexes. Chem. Commun. 46, 4541–4543 (2010).
Nakamura, E., Yoshikai, N. & Yamanaka, M. Mechanism of C–H bond activation/C–C bond formation reaction between diazo compound and alkane catalyzed by dirhodium tetracarboxylate. J. Am. Chem. Soc. 124, 7181–7192 (2002).
Acknowledgements
Financial support was provided by the National Science Foundation (NSF) under the CCI Center for Selective C–H Functionalization (CHE-1700982). D.G.M. gratefully acknowledges NSF MRI-R2 grant (CHE-0958205) and the use of the resources of the Cherry Emerson Center for Scientific Computation. NMR and X-ray instrumentation used in this work was supported by the NSF (CHE-1531620 and CHE-1626172).
Reviewer information
Nature thanks V. Gevorgyan and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
J.F. performed the synthetic experiments. Z.R. and D.G.M. conducted the computational studies. J.B. conducted the X-ray crystallographic studies. J.F. and H.M.L.D. designed and analysed the synthetic experiments and prepared the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Structures of previously established catalysts.
We have previously shown that, through catalyst-directed C–H functionalization, the most accessible primary, secondary and tertiary C–H bonds within a linear alkane substrate could be selectively functionalized by using catalyst 2, 3 or 4.
Supplementary information
Supplementary Information
This file contains Supplementary Text and Data Sections 1-7.
Rights and permissions
About this article
Cite this article
Fu, J., Ren, Z., Bacsa, J. et al. Desymmetrization of cyclohexanes by site- and stereoselective C–H functionalization. Nature 564, 395–399 (2018). https://doi.org/10.1038/s41586-018-0799-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-018-0799-2
- Springer Nature Limited
This article is cited by
-
Photochemical diversification of strong C(sp3)–H bonds enabled by allyl bromide and sodium fluoride
Nature Synthesis (2023)
-
Silver-catalyzed site-selective C(sp3)−H benzylation of ethers with N-triftosylhydrazones
Nature Communications (2022)
-
Asymmetric 1,2-oxidative alkylation of conjugated dienes via aliphatic C–H bond activation
Nature Synthesis (2022)
-
Unconventional mechanism and selectivity of the Pd-catalyzed C–H bond lactonization in aromatic carboxylic acid
Nature Communications (2022)
-
Visible-light-mediated tungsten-catalyzed C-H amination of unactivated alkanes with nitroarenes
Science China Chemistry (2022)