Abstract
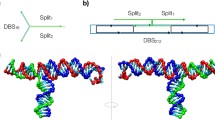
DNA origami is capable of spatially organizing molecules into sophisticated geometric patterns with nanometric precision. Here we describe a reconfigurable, two-dimensional DNA origami with geometrically patterned CD95 ligands that regulates immune cell signalling to alleviate rheumatoid arthritis. In response to pH changes, the device reversibly transforms from a closed to an open configuration, displaying a hexagonal pattern of CD95 ligands with ~10 nm intermolecular spacing, precisely mirroring the spatial arrangement of CD95 receptor clusters on the surface of immune cells. In a collagen-induced arthritis mouse model, DNA origami elicits robust and selective activation of CD95 death-inducing signalling in activated immune cells located in inflamed synovial tissues. Such localized immune tolerance ameliorates joint damage with no noticeable side effects. This device allows for the precise spatial control of cellular signalling, expanding our understanding of ligand–receptor interactions and is a promising platform for the development of pharmacological interventions targeting these interactions.





Similar content being viewed by others
Data availability
The data that support the findings of this study are available within the Article and its Supplementary Information files. Source data can be found on figshare at https://doi.org/10.6084/m9.figshare.25111502 (ref. 51). Source data are provided with this paper.
References
Vital, E. M. & Emery, P. The development of targeted therapies in rheumatoid arthritis. J. Autoimmun. 31, 219–227 (2008).
Smolen, J. S. & Aletaha, D. Rheumatoid arthritis therapy reappraisal: strategies, opportunities and challenges. Nat. Rev. Rheumatol. 11, 276–289 (2015).
Ju, S.-T. et al. Fas (CD95)/FasL interactions required for programmed cell death after T-cell activation. Nature 373, 444–448 (1995).
Vanamee, É. S. & Faustman, D. L. Structural principles of tumor necrosis factor superfamily signaling. Sci. Signal. 11, eaao4910 (2018).
Scott, F. L. et al. The Fas–FADD death domain complex structure unravels signalling by receptor clustering. Nature 457, 1019–1022 (2009).
Wang, L. et al. The Fas–FADD death domain complex structure reveals the basis of DISC assembly and disease mutations. Nat. Struct. Mol. Biol. 17, 1324–1329 (2010).
Balta, G. S. G. et al. 3D cellular architecture modulates tyrosine kinase activity, thereby switching CD95-mediated apoptosis to survival. Cell Rep. 29, 2295–2306 (2019).
Rothemund, P. W. Folding DNA to create nanoscale shapes and patterns. Nature 440, 297–302 (2006).
Chandrasekaran, A. R., Anderson, N., Kizer, M., Halvorsen, K. & Wang, X. Beyond the fold: emerging biological applications of DNA origami. ChemBioChem 17, 1081–1089 (2016).
Shaw, A. et al. Spatial control of membrane receptor function using ligand nanocalipers. Nat. Methods 11, 841–846 (2014).
Kwon, P. S. et al. Designer DNA architecture offers precise and multivalent spatial pattern-recognition for viral sensing and inhibition. Nat. Chem. 12, 26–35 (2020).
Wang, Y., Baars, I., Fordos, F. & Hogberg, B. Clustering of death receptor for apoptosis using nanoscale patterns of peptides. ACS Nano 15, 9614–9626 (2021).
Comberlato, A., Koga, M. M., Nüssing, S., Parish, I. A. & Bastings, M. M. C. Spatially controlled activation of Toll-like receptor 9 with DNA-based nanomaterials. Nano Lett. 22, 2506–2513 (2022).
Fang, T. et al. Spatial regulation of T-cell signaling by programmed death-ligand 1 on wireframe DNA origami flat sheets. ACS Nano 15, 3441–3452 (2021).
Shaw, A. et al. Binding to nanopatterned antigens is dominated by the spatial tolerance of antibodies. Nat. Nanotechnol. 14, 184–190 (2019).
Wickham, S. F. J. et al. Complex multicomponent patterns rendered on a 3D DNA-barrel pegboard. Nat. Commun. 11, 5768 (2020).
Eklund, A. S., Comberlato, A., Parish, I. A., Jungmann, R. & Bastings, M. M. C. Quantification of strand accessibility in biostable DNA origami with single-staple resolution. ACS Nano 15, 17668–17677 (2021).
Goldie, I. & Nachemson, A. Synovial pH in rheumatoid knee-joints. I. The effect of synovectomy. Acta Orthop. Scand. 40, 634–641 (1969).
Stevens, C. R., Williams, R. B., Farrell, A. J. & Blake, D. R. Hypoxia and inflammatory synovitis: observations and speculation. Ann. Rheum. Dis. 50, 124–132 (1991).
Farr, M., Garvey, K., Bold, A. M., Kendall, M. J. & Bacon, P. A. Significance of the hydrogen ion concentration in synovial fluid in rheumatoid arthritis. Clin. Exp. Rheumatol. 3, 99–104 (1985).
Roskams, T., Libbrecht, L., Van Damme, B. & Desmet, V. Fas and Fas ligand: strong co-expression in human hepatocytes surrounding hepatocellular carcinoma; can cancer induce suicide in peritumoural cells? J. Pathol. 191, 150–153 (2000).
Li, S. et al. A DNA nanorobot functions as a cancer therapeutic in response to a molecular trigger in vivo. Nat. Biotechnol. 36, 258–264 (2018).
Liu, S. et al. A DNA nanodevice-based vaccine for cancer immunotherapy. Nat. Mater. 20, 421–430 (2021).
Manuguri, S., Nguyen, M. K., Loo, J., Natarajan, A. K. & Kuzyk, A. Advancing the utility of DNA origami technique through enhanced stability of DNA-origami-based assemblies. Bioconjug. Chem. 34, 6–17 (2023).
Wang, P. F., Meyer, T. A., Pan, V., Dutta, P. K. & Ke, Y. G. The beauty and utility of DNA origami. Chem 2, 359–382 (2017).
Hahn, J., Wickham, S. F. J., Shih, W. M. & Perrault, S. D. Addressing the instability of DNA nanostructures in tissue culture. ACS Nano 8, 8765–8775 (2014).
Ponnuswamy, N. et al. Oligolysine-based coating protects DNA nanostructures from low-salt denaturation and nuclease degradation. Nat. Commun. 8, 15654 (2017).
Perrault, S. D. & Shih, W. M. Virus-inspired membrane encapsulation of DNA nanostructures to achieve in vivo stability. ACS Nano 8, 5132–5140 (2014).
Mei, Q. A. et al. Stability of DNA origami nanoarrays in cell lysate. Nano Lett. 11, 1477–1482 (2011).
Chandrasekaran, A. R. Nuclease resistance of DNA nanostructures. Nat. Rev. Chem. 5, 225–239 (2021).
Schneider, P. et al. Conversion of membrane-bound Fas (CD95) ligand to its soluble form is associated with downregulation of its proapoptotic activity and loss of liver toxicity. J. Exp. Med. 187, 1205–1213 (1998).
Algeciras-Schimnich, A. et al. Molecular ordering of the initial signaling events of CD95. Mol. Cell. Biol. 22, 207–220 (2002).
Lee, K. H. et al. The role of receptor internalization in CD95 signaling. EMBO J. 25, 1009–1023 (2006).
Malyala, P. & Singh, M. Endotoxin limits in formulations for preclinical research. J. Pharm. Sci. 97, 2041–2044 (2008).
Hong, E. et al. Structure and composition define immunorecognition of nucleic acid nanoparticles. Nano Lett. 18, 4309–4321 (2018).
Namekawa, T., Wagner, U. G., Goronzy, J. J. & Weyand, C. M. Functional subsets of CD4 T cells in rheumatoid synovitis. Arthritis Rheum. 41, 2108–2116 (1998).
Gizinski, A. M. & Fox, D. A. T cell subsets and their role in the pathogenesis of rheumatic disease. Curr. Opin. Rheumatol. 26, 204–210 (2014).
Reparon-Schuijt, C. C. et al. Secretion of anti-citrulline-containing peptide antibody by B lymphocytes in rheumatoid arthritis. Arthritis Rheum. 44, 41–47 (2001).
Okamoto, K. et al. Induction of apoptosis in the rheumatoid synovium by Fas ligand gene transfer. Gene Ther. 5, 331–338 (1998).
Zhang, H. et al. Amelioration of collagen-induced arthritis by CD95 (Apo-1/Fas)-ligand gene transfer. J. Clin. Investig. 100, 1951–1957 (1997).
Choy, E. H. & Panayi, G. S. Cytokine pathways and joint inflammation in rheumatoid arthritis. N. Engl. J. Med. 344, 907–916 (2001).
Smolen, J. S. & Steiner, G. Therapeutic strategies for rheumatoid arthritis. Nat. Rev. Drug Discov. 2, 473–488 (2003).
Fullerton, J. N., O’Brien, A. J. & Gilroy, D. W. Pathways mediating resolution of inflammation: when enough is too much. J. Pathol. 231, 8–20 (2013).
Elliott, M. R., Koster, K. M. & Murphy, P. S. Efferocytosis signaling in the regulation of macrophage inflammatory responses. J. Immunol. 198, 1387–1394 (2017).
Achkar, I. W. et al. Oncogenic role of dysregulated leptin signaling in the pathogenesis of ovarian cancer. Transl. Med. Commun. 4, 1 (2019).
Akhtar, S. et al. Cytokine-mediated dysregulation of signaling pathways in the pathogenesis of multiple myeloma. Int. J. Mol. Sci. 21, 5002 (2020).
Mehta, M. et al. Cellular signalling pathways mediating the pathogenesis of chronic inflammatory respiratory diseases: an update. Inflammopharmacology 28, 795–817 (2020).
Brand, D. D., Latham, K. A. & Rosloniec, E. F. Collagen-induced arthritis. Nat. Protoc. 2, 1269–1275 (2007).
Zhou, B. et al. Therapeutic effects of a novel BAFF blocker on arthritis. Signal Transduct. Target. Ther. 4, 19 (2019).
Wang, H. et al. TACI-ligand interactions are required for T cell activation and collagen-induced arthritis in mice. Nat. Immunol. 2, 632–637 (2001).
Li, L. A DNA origami device spatially controls CD95 signalling to induce immune tolerance in rheumatoid arthritis. figshare https://doi.org/10.6084/m9.figshare.25111502.v2 (2024).
Acknowledgements
We acknowledge financial support from the National University of Singapore (grant nos. NUHSRO/2020/133/Startup/08, NUHSRO/2023/008/NUSMed/TCE/LOA and NUHSRO/2021/034/TRP/09/Nanomedicine), National Medical Research Council (grant no. MOH-OFIRG23jan-0005), Singapore Ministry of Education (grant no. MOE-000387-00), National Research Foundation (grant no. NRF-000352-00), National Natural Science Foundation of China (grant nos. NSFC 82202311, 62288102, 82302356 and 22274081), Science Foundation of Fujian Normal University (grant no. Y07204080K13) and Natural Science Foundation of Jiangsu Province, Major Project (grant no. BK20212012). We thank H. Liu at the National University of Singapore for providing A20 cells. We thank X. Song at the West China School of Public Health and West China Fourth Hospital, Sichuan University for help with confocal microscopy imaging. We thank B. Zhou at the State Key Laboratory of Biotherapy and Cancer Center, West China Hospital, Sichuan University for help with establishing the CIA model. We thank Guangzhou Sagene Biotech Co., Ltd. for help in making the pattern diagrams.
Author information
Authors and Affiliations
Contributions
X.C., J.C., Z.Y., L.L., J.Y., W.M. and L.T. conceived and designed the experiments. L.L., J.Y., W.M., L.T. and L.Y. performed experiments and wrote the manuscript. J.Z., T.D. and Y.Z. performed data analysis. X.C., J.C. and Z.Y. supervised the entire project. L.W. and C.F. conducted proofreading of the manuscript. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Materials thanks Maartje Bastings, Mauro Perretti, Marcus Peter and Liangfang Zhang for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 The spatial arrangement of the CD95L array directs the activation of CD95 signaling in Jurkat cells.
a, Clustering of CD95 receptors in Jurkat cells 2 h following treatment with NS-empty (G2), sCD95L (G3), NS-10 (G4), ND-10 in the culture medium (pH 7.4) (G5), ND-10 in the culture medium (pH 6.5) (G6). Untreated (G1); Scale bar, 5 μm. The experiment was performed three times with similar results. (b, c), Colocalization of CD95 with the endosomal marker-early endosome antigen (EEA1) following NS-10 stimulation (b) or ND-10 stimulation in the culture medium (pH 6.5) (c). Scale bar, 5 μm. The experiment was performed three times with similar results. d, Immunocytochemistry analysis of Cleaved Caspase-8 in Jurkat cells 6 h following treatment with NS-empty (G2), sCD95L (G3), NS-10 (G4), ND-10 in the culture medium (pH 7.4) (G5), ND-10 in the culture medium (pH 6.5) (G6). Untreated (G1); Scale bar, 5 μm. The experiment was performed three times with similar results.
Extended Data Fig. 2 DNA origami nanodevice exhibits conformational transition in response to pH trigger in vivo.
Representative in vivo fluorescence images of CIA mice after intravenous injection with pH-sensitive nanodevice with i-motif-based fasteners labeled with both Cy5.5 fluorophore and BBQ650 quencher (w/ Cy5.5 & BBQ650), pH-sensitive nanodevice with i-motif-based fasteners labeled with Cy5.5 fluorophore (w/ Cy5.5), or pH-non-sensitive nanodevice with the pH-non-sensitive fasteners labeled with both Cy5.5 fluorophore and BBQ650 quencher (w/ Cy5.5 & BBQ650).
Extended Data Fig. 3 Designer DNA origami shows minimal immunomodulation potential.
(a, b) Concentration of TNF-α, IL-6, and IL-1β in the supernatants of RAW 264.7 cells (a) and BMDMs (b) following ND-empty treatment at indicated concentration. LPS (lipopolysaccharide, Invitrogen) was used as a control. n = 3 biologically independent samples per group. Note that the cytokine concentrations in some samples were below the threshold of the assay range and they were determined by extrapolating from the standard curve. Data presented as means ± s.d. Statistical significance was calculated via One-way ANOVA with Tukey post-hoc test (a, b).
Extended Data Fig. 4 Designer DNA origami yields long-term therapeutic efficacy in CIA mice.
a, Schematic illustration of the long-term treatment regimen. b, Clinical arthritis scores of the CIA mice following indicated treatments. n = 12 biologically independent mice per group. c, The paw swelling measurement of the CIA mice following indicated treatments. n = 12 biologically independent mice per group. Data presented as means ± s.d. Statistical significance was calculated via One-way ANOVA with Tukey post-hoc test (b, c). The experiment was performed twice with similar results.
Supplementary information
Supplementary Information
Supplementary Figs. 1–43.
Supplementary Data
DNA sequences of the staple strand pool and functional staple strands.
Source data
Source Data Fig. 2
Statistical source data.
Source Data Fig. 3
Statistical source data.
Source Data Fig. 4
Statistical source data.
Source Data Fig. 5
Statistical source data.
Source Data Extended Data Fig. 3.
Statistical source data.
Source Data Extended Data Fig. 4.
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, L., Yin, J., Ma, W. et al. A DNA origami device spatially controls CD95 signalling to induce immune tolerance in rheumatoid arthritis. Nat. Mater. (2024). https://doi.org/10.1038/s41563-024-01865-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41563-024-01865-5
- Springer Nature Limited