Abstract
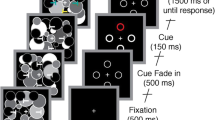
Salient objects often capture our attention, serving as distractors and hindering our current goals. It remains unclear when and how salient distractors interact with our goals, and our knowledge on the neural mechanisms responsible for attentional capture is limited to a few brain regions recorded from non-human primates. Here we conducted a multivariate analysis on human intracranial signals covering most brain regions and successfully dissociated distractor-specific representations from target-arousal signals in the high-frequency (60–100 Hz) activity. We found that salient distractors were processed rapidly around 220 ms, while target-tuning attention was attenuated simultaneously, supporting initial capture by distractors. Notably, neuronal activity specific to the distractor representation was strongest in the superior and middle temporal gyrus, amygdala and anterior cingulate cortex, while there were smaller contributions from the parietal and frontal cortices. These results provide neural evidence for attentional capture by salient distractors engaging a much larger network than previously appreciated.




Similar content being viewed by others
Data availability
As the iEEG data are being used in ongoing studies, we are unable to share them at this time. However, should researchers express interest in replicating our study, we will share the derived iEEG data upon request.
Code availability
The code we used can be accessed through https://github.com/wangbenchi/Shared_data (ref. 78).
References
Theeuwes, J. Top-down and bottom-up control of visual selection. Acta Psychol. 135, 77–99 (2010).
Itti, L. & Koch, C. Computational modelling of visual attention. Nat. Rev. Neurosci. 2, 194–203 (2001).
Theeuwes, J. Perceptual selectivity for color and form. Percept. Psychophys. 51, 599–606 (1992).
Theeuwes, J. Exogenous and endogenous control of attention: the effect of visual onsets and offsets. Percept. Psychophys. 49, 83–90 (1991).
Anderson, B. A., Laurent, P. A. & Yantis, S. Value-driven attentional capture. Proc. Natl Acad. Sci. USA 108, 10367–10371 (2011).
Schmidt, L. J., Belopolsky, A. V. & Theeuwes, J. Attentional capture by signals of threat. Cogn. Emot. 29, 687–694 (2015).
Luck, S. J., Gaspelin, N., Folk, C. L., Remington, R. W. & Theeuwes, J. Progress toward resolving the attentional capture debate. Vis. Cogn. 29, 1–21 (2021).
Constantinidis, C. & Steinmetz, M. A. Neuronal responses in Area 7a to multiple-stimulus displays: I. Neurons encode the location of the salient stimulus. Cereb. Cortex 11, 581–591 (2001).
Constantinidis, C. Posterior parietal cortex automatically encodes the location of salient stimuli. J. Neurosci. 25, 233–238 (2005).
Katsuki, F. & Constantinidis, C. Early involvement of prefrontal cortex in visual bottom-up attention. Nat. Neurosci. 15, 1160–1166 (2012).
Bichot, N. P., Schall, J. D. & Thompson, K. G. Visual feature selectivity in frontal eye fields induced by experience in mature macaques. Nature 381, 697–699 (1996).
Schall, J. D. & Hanes, D. P. Neural basis of saccade target selection in frontal eye field during visual search. Nature 366, 467–469 (1993).
Bichot, N. P. & Schall, J. D. Priming in macaque frontal cortex during popout visual search: feature-based facilitation and location-based inhibition of return. J. Neurosci. 22, 4675–4685 (2002).
Klink, P. C., Teeuwen, R. R. M., Lorteije, J. A. M. & Roelfsema, P. R. Inversion of pop-out for a distracting feature dimension in monkey visual cortex. Proc. Natl Acad. Sci. USA 120, e2210839120 (2023).
Ipata, A. E., Gee, A. L., Gottlieb, J., Bisley, J. W. & Goldberg, M. E. LIP responses to a popout stimulus are reduced if it is overtly ignored. Nat. Neurosci. 9, 1071–1076 (2006).
Cosman, J. D., Lowe, K. A., Zinke, W., Woodman, G. F. & Schall, J. D. Prefrontal control of visual distraction. Curr. Biol. 28, 414–420 (2018).
Moran, J. & Desimone, R. Selective attention gates visual processing in the extrastriate cortex. Science 229, 782–784 (1985).
Treue, S. & Trujillo, J. C. M. Feature-based attention influences motion processing gain in macaque visual cortex. Nature 399, 575–579 (1999).
Hickey, C., McDonald, J. J. & Theeuwes, J. Electrophysiological evidence of the capture of visual attention. J. Cogn. Neurosci. 18, 604–613 (2006).
Brouwer, G. J. & Heeger, D. J. Decoding and reconstructing color from responses in human visual cortex. J. Neurosci. 29, 13992–14003 (2009).
Sprague, T. C. & Serences, J. T. Attention modulates spatial priority maps in the human occipital, parietal and frontal cortices. Nat. Neurosci. 16, 1879–1887 (2013).
Foster, J. J., Sutterer, D. W., Serences, J. T., Vogel, E. K. & Awh, E. Alpha-band oscillations enable spatially and temporally resolved tracking of covert spatial attention. Psychol. Sci. 28, 929–941 (2017).
Sprague, T. C., Saproo, S. & Serences, J. T. Visual attention mitigates information loss in small- and large-scale neural codes. Trends Cogn. Sci. 19, 215–226 (2015).
Ossandon, T. et al. Efficient ‘pop-out’ visual search elicits sustained broadband gamma activity in the dorsal attention network. J. Neurosci. 32, 3414–3421 (2012).
Clayton, M. S., Yeung, N. & Cohen Kadosh, R. The roles of cortical oscillations in sustained attention. Trends Cogn. Sci. 19, 188–195 (2015).
Yeo, B. T. et al. The organization of the human cerebral cortex estimated by intrinsic functional connectivity. J. Neurophysiol. 106, 1125–1165 (2011).
Kunz, L. et al. Hippocampal theta phases organize the reactivation of large-scale electrophysiological representations during goal-directed navigation. Sci. Adv. 5, eaav8192 (2019).
Buschman, T. J. & Miller, E. K. Top-down versus bottom-up control of attention in the prefrontal and posterior parietal cortices. Science 315, 1860–1862 (2007).
Chen, X. et al. Parietal cortex regulates visual salience and salience-driven behavior. Neuron 106, 177–187 (2020).
Norman, Y. et al. Hippocampal sharp-wave ripples linked to visual episodic recollection in humans. Science 365, eaax1030 (2019).
Solomon, E. A. et al. Widespread theta synchrony and high-frequency desynchronization underlies enhanced cognition. Nat. Commun. 8, 1704 (2017).
Banaie Boroujeni, K., Oemisch, M., Hassani, S. A. & Womelsdorf, T. Fast spiking interneuron activity in primate striatum tracks learning of attention cues. Proc. Natl Acad. Sci. USA 117, 18049–18058 (2020).
Gueguen, M. C. et al. Anatomical dissociation of intracerebral signals for reward and punishment prediction errors in humans. Nat. Commun. 12, 3344 (2021).
Goldstein, A. et al. Shared computational principles for language processing in humans and deep language models. Nat. Neurosci. 25, 369–380 (2022).
Khalighinejad, B., Herrero, J. L., Mehta, A. D. & Mesgarani, N. Adaptation of the human auditory cortex to changing background noise. Nat. Commun. 10, 2509 (2019).
Gaspelin, N. & Luck, S. J. Inhibition as a potential resolution to the attentional capture debate. Curr. Opin. Psychol. 29, 12–18 (2019).
Geng, J. J. Attentional mechanisms of distractor suppression. Curr. Dir. Psychol. Sci. 23, 147–153 (2014).
Theeuwes, J., Bogaerts, L. & van Moorselaar, D. What to expect where and when: how statistical learning drives visual selection. Trends Cogn. Sci. 26, 860–872 (2022).
Wang, B. & Theeuwes, J. Statistical regularities modulate attentional capture. J. Exp. Psychol. Hum. Percept. Perform. 44, 13–17 (2018).
Jonides, J. & Yantis, S. Uniqueness of abrupt visual onset in capturing attention. Percept. Psychophys. 43, 346–354 (1988).
Folk, C. L., Remington, R. W. & Johnston, J. C. Involuntary covert orienting is contingent on attentional control settings. J. Exp. Psychol. Hum. Percept. Perform. 18, 1030–1044 (1992).
Gaspelin, N., Leonard, C. J. & Luck, S. J. Direct evidence for active suppression of salient-but-irrelevant sensory inputs. Psychol. Sci. 26, 1740–1750 (2015).
Wang, B., Van Driel, J., Ort, E. & Theeuwes, J. Anticipatory distractor suppression elicited by statistical regularities in visual search. J. Cogn. Neurosci. 31, 1535–1548 (2019).
Baudena, P., Halgren, E., Heit, G. & Clarke, J. M. Intracerebral potentials to rare target and distractor auditory and visual stimuli. III. Frontal cortex. Electroencephalogr. Clin. Neurophysiol. 94, 251–264 (1995).
Clark, V. P., Fannon, S., Lai, S., Benson, R. & Bauer, L. Responses to rare visual target and distractor stimuli using event-related fMRI. J. Neurophysiol. 83, 3133–3139 (2000).
Kiehl, K. A., Laurens, K. R., Duty, T. L., Forster, B. B. & Liddle, P. F. Neural sources involved in auditory target detection and novelty processing: an event-related fMRI study. Psychophysiology 38, 133–142 (2001).
Uddin, L. Q. Salience processing and insular cortical function and dysfunction. Nat. Rev. Neurosci. 16, 55–61 (2015).
Menon, V. & Uddin, L. Q. Saliency, switching, attention and control: a network model of insula function. Brain Struct. Funct. 214, 655–667 (2010).
Sani, I. et al. The human endogenous attentional control network includes a ventro-temporal cortical node. Nat. Commun. 12, 360 (2021).
Stemmann, H. & Freiwald, W. A. Evidence for an attentional priority map in inferotemporal cortex. Proc. Natl Acad. Sci. USA 116, 23797–23805 (2019).
Bogadhi, A. R., Bollimunta, A., Leopold, D. A. & Krauzlis, R. J. Spatial attention deficits are causally linked to an area in macaque temporal cortex. Curr. Biol. 29, 726–736 (2019).
Ramezanpour, H. & Fallah, M. The role of temporal cortex in the control of attention. Curr. Res. Neurobiol. 3, 100038 (2022).
Peck, C. J., Lau, B. & Salzman, C. D. The primate amygdala combines information about space and value. Nat. Neurosci. 16, 340–348 (2013).
Knight, R. T. Contribution of human hippocampal region to novelty detection. Nature 383, 256–259 (1996).
Rao, H., Zhou, T., Zhuo, Y., Fan, S. & Chen, L. Spatiotemporal activation of the two visual pathways in form discrimination and spatial location: a brain mapping study. Hum. Brain Mapp. 18, 79–89 (2003).
Posner, M. I. Orienting of attention. Q. J. Exp. Psychol. 32, 3–25 (1980).
Corbetta, M. & Shulman, G. L. Control of goal-directed and stimulus-driven attention in the brain. Nat. Rev. Neurosci. 3, 201–215 (2002).
Vossel, S., Geng, J. J. & Fink, G. R. Dorsal and ventral attention systems: distinct neural circuits but collaborative roles. Neuroscientist 20, 150–159 (2014).
Van Rossum, G. & Drake, F. L. Python Reference Manual http://docs.python.org (2006).
Delorme, A. & Makeig, S. EEGLAB: an open source toolbox for analysis of single-trial EEG dynamics including independent component analysis. J. Neurosci. Methods 134, 9–21 (2004).
Lopez-Calderon, J. & Luck, S. J. ERPLAB: an open-source toolbox for the analysis of event-related potentials. Front. Hum. Neurosci. 8, 213 (2014).
Liu, J. et al. Stable maintenance of multiple representational formats in human visual short-term memory. Proc. Natl Acad. Sci. USA 117, 32329–32339 (2020).
Bijanzadeh, M. et al. Decoding naturalistic affective behaviour from spectro-spatial features in multiday human iEEG. Nat. Hum. Behav. 6, 823–836 (2022).
Grossman, S. et al. Convergent evolution of face spaces across human face-selective neuronal groups and deep convolutional networks. Nat. Commun. 10, 4934 (2019).
Feldmann-Wüstefeld, T., Weinberger, M. & Awh, E. Spatially guided distractor suppression during visual search. J. Neurosci. 41, 3180–3191 (2021).
Gaspelin, N., Leonard, C. J. & Luck, S. J. Suppression of overt attentional capture by salient-but-irrelevant color singletons. Atten. Percept. Psychophys. 79, 45–62 (2017).
Wang, B., Samara, I. & Theeuwes, J. Statistical regularities bias overt attention. Atten. Percept. Psychophys. 81, 1813–1821 (2019).
Fischl, B., Sereno, M. I. & Dale, A. M. Cortical surface-based analysis: II: inflation, flattening, and a surface-based coordinate system. NeuroImage 9, 195–207 (1999).
Groppe, D. M. et al. iELVis: an open source MATLAB toolbox for localizing and visualizing human intracranial electrode data. J. Neurosci. Methods 281, 40–48 (2017).
Mukamel, R. et al. Coupling between neuronal firing, field potentials, and fMRI in human auditory cortex. Science 309, 951–954 (2005).
Parvizi, J. & Kastner, S. Promises and limitations of human intracranial electroencephalography. Nat. Neurosci. 21, 474–483 (2018).
Watson, B. O., Ding, M. & Buzsáki, G. Temporal coupling of field potentials and action potentials in the neocortex. Eur. J. Neurosci. 48, 2482–2497 (2018).
Jensen, O., Kaiser, J. & Lachaux, J.-P. Human gamma-frequency oscillations associated with attention and memory. Trends Neurosci. 30, 317–324 (2007).
Benjamini, Y. & Yekutieli, D. The control of the false discovery rate in multiple testing under dependency. Ann. Stat. 29, 1165–1188 (2001).
Norman, Y., Yeagle, E. M., Harel, M., Mehta, A. D. & Malach, R. Neuronal baseline shifts underlying boundary setting during free recall. Nat. Commun. 8, 1301 (2017).
Miller, J., Patterson, T. & Ulrich, R. Jackknife-based method for measuring LRP onset latency differences. Psychophysiology 35, 99–115 (1998).
Wang, L., Kuperberg, G. & Jensen, O. Specific lexico-semantic predictions are associated with unique spatial and temporal patterns of neural activity. eLife 7, e39061 (2018).
Wang, B. Shared_data, GitHub, https://github.com/wangbenchi/Shared_data (2024).
Acknowledgements
This research was supported by the National Science and Technology Innovation 2030 Major Program (grant no. 2022ZD0204802 to B.W.), the National Natural Science Foundation of China (grant no. 32000738 to B.W.) and the Sanming Project of Medicine in Shenzhen (grant no. SZSM202003006 to X.M.). The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
R.L., X.M., F.C., X.L. and B.W. designed the experiment. R.L., X.M. and F.C. collected the data. R.L. and B.W. analysed the data. R.L., O.J., J.T. and B.W. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Human Behaviour thanks Jacqueline Gottlieb and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary results (including ten figures).
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lin, R., Meng, X., Chen, F. et al. Neural evidence for attentional capture by salient distractors. Nat Hum Behav 8, 932–944 (2024). https://doi.org/10.1038/s41562-024-01852-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41562-024-01852-5
- Springer Nature Limited
This article is cited by
-
Attentional capture
Nature Reviews Neuroscience (2024)