Abstract
TAZ promotes growth, development and tumorigenesis by regulating the expression of target genes. However, the manner in which TAZ orchestrates the transcriptional responses is poorly defined. Here we demonstrate that TAZ forms nuclear condensates through liquid–liquid phase separation to compartmentalize its DNA-binding cofactor TEAD4, coactivators BRD4 and MED1, and the transcription elongation factor CDK9 for transcription. TAZ forms phase-separated droplets in vitro and liquid-like nuclear condensates in vivo, and this ability is negatively regulated by Hippo signalling through LATS-mediated phosphorylation and is mediated by the coiled-coil (CC) domain. Deletion of the TAZ CC domain or substitution with the YAP CC domain prevents the phase separation of TAZ and its ability to induce the expression of TAZ-specific target genes. Thus, we identify a mechanism of transcriptional activation by TAZ and demonstrate that pathway-specific transcription factors also engage the phase-separation mechanism for efficient and specific transcriptional activation.








Similar content being viewed by others
Data availability
Source data for Figs. 1–4 and 6–8 and Extended Data Figs. 1, 3, 4 and 8 are available online. The RNA-seq data are available in the Gene Expression Omnibus (GEO) with the accession number GSE142474. All other data supporting the findings of this study are available from the corresponding author on reasonable request.
References
Pan, D. The Hippo signaling pathway in development and cancer. Dev. Cell 19, 491–505 (2010).
Ma, S., Meng, Z., Chen, R. & Guan, K. L. The Hippo pathway: biology and pathophysiology. Annu. Rev. Biochem. 88, 577–604 (2018).
Tapon, N. & Harvey, K. F. The Hippo pathway—from top to bottom and everything in between. Semin. Cell Dev. Biol. 23, 768–769 (2012).
Badouel, C. & McNeill, H. SnapShot: the Hippo signaling pathway. Cell 145, 484 (2011).
Schroeder, M. C. & Halder, G. Regulation of the Hippo pathway by cell architecture and mechanical signals. Semin. Cell Dev. Biol. 23, 803–811 (2012).
Yu, F. X. & Guan, K. L. The Hippo pathway: regulators and regulations. Genes Dev. 27, 355–371 (2013).
Dong, J. X. et al. Elucidation of a universal size-control mechanism in Drosophila and mammals. Cell 130, 1120–1133 (2007).
Ota, M. & Sasaki, H. Mammalian Tead proteins regulate cell proliferation and contact inhibition as transcriptional mediators of Hippo signaling. Development 135, 4059–4069 (2008).
Enderle, L. & McNeill, H. Hippo gains weight: added insights and complexity to pathway control. Sci. Signal. 6, re7 (2013).
Shreberk-Shaked, M. & Oren, M. New insights into YAP/TAZ nucleo-cytoplasmic shuttling: new cancer therapeutic opportunities? Mol. Oncol. 13, 1335–1341 (2019).
Galli, G. G. et al. YAP drives growth by controlling transcriptional pause release from dynamic enhancers. Mol. Cell 60, 328–337 (2015).
Zanconato, F. et al. Transcriptional addiction in cancer cells is mediated by YAP/TAZ through BRD4. Nat. Med. 24, 1599–1610 (2018).
Boija, A. et al. Transcription factors activate genes through the phase-separation capacity of their activation domains. Cell 175, 1842–1855 (2018).
Chan, S. W. et al. A role for TAZ in migration, invasion, and tumorigenesis of breast cancer cells. Cancer Res. 68, 2592–2598 (2008).
Cordenonsi, M. et al. The Hippo transducer TAZ confers cancer stem cell-related traits on breast cancer cells. Cell 147, 759–772 (2011).
Plouffe, S. W. et al. The Hippo pathway effector proteins YAP and TAZ have both distinct and overlapping functions in the cell. J. Biol. Chem. 293, 11230–11240 (2018).
Xin, M. et al. Hippo pathway effector Yap promotes cardiac regeneration. Proc. Natl Acad. Sci. USA 110, 13839–13844 (2013).
Nishioka, N. et al. The Hippo signaling pathway components Lats and Yap pattern Tead4 activity to distinguish mouse trophectoderm from inner cell mass. Dev. Cell 16, 398–410 (2009).
Morin-Kensicki, E. M. et al. Defects in yolk sac vasculogenesis, chorioallantoic fusion, and embryonic axis elongation in mice with targeted disruption of Yap65. Mol. Cell. Biol. 26, 77–87 (2006).
Hossain, Z. et al. Glomerulocystic kidney disease in mice with a targeted inactivation of Wwtr1. Proc. Natl Acad. Sci. USA 104, 1631–1636 (2007).
Passaniti, A., Brusgard, J. L., Qiao, Y. T., Sudol, M. & Finch-Edmondson, M. Roles of RUNX in Hippo pathway signaling. Adv. Exp. Med. Biol. 962, 435–448 (2017).
Zhao, B. et al. TEAD mediates YAP-dependent gene induction and growth control. Gene Dev. 22, 1962–1971 (2008).
Zhao, B., Tumaneng, K. & Guan, K. L. The Hippo pathway in organ size control, tissue regeneration and stem cell self-renewal. Nat. Cell Biol. 13, 877–883 (2011).
Alberti, S., Gladfelter, A. & Mittag, T. Considerations and challenges in studying liquid-liquid phase separation and biomolecular condensates. Cell 176, 419–434 (2019).
Sabari, B. R. et al. Coactivator condensation at super-enhancers links phase separation and gene control. Science 361, eaar3958 (2018).
Lu, H., Liu, R. & Zhou, Q. Balanced between order and disorder: a new phase in transcription elongation control and beyond. Transcription 10, 157–163 (2019).
Kaan, H. Y. K. et al. Crystal structure of TAZ-TEAD complex reveals a distinct interaction mode from that of YAP-TEAD complex. Sci. Rep. 7, 2035 (2017).
Piccolo, S., Dupont, S. & Cordenonsi, M. The biology of Yap/Taz: Hippo signaling and beyond. Physiol. Rev. 94, 1287–1312 (2014).
Hyman, A. A., Weber, C. A. & Juelicher, F. Liquid-liquid phase separation in biology. Annu. Rev. Cell Dev. Biol. 30, 39–58 (2014).
Boeynaems, S. et al. Protein phase separation: a new phase in cell biology. Trends Cell Biol. 28, 420–435 (2018).
Cai, D. et al. Phase separation of YAP reorganizes genome topology for long-term YAP target gene expression. Nat. Cell Biol. 21, 1578–1589 (2019).
Henis, Y. I., Rotblat, B. & Kloog, Y. FRAP beam-size analysis to measure palmitoylation-dependent membrane association dynamics and microdomain partitioning of Ras proteins. Methods 40, 183–190 (2006).
Eisenberg, S., Giehl, K., Henis, Y. I. & Ehrlich, M. Differential interference of chlorpromazine with the membrane interactions of oncogenic K-Ras and its effects on cell growth. J. Biol. Chem. 283, 27279–27288 (2008).
Shvartsman, D. E. et al. Src kinase activity and SH2 domain regulate the dynamics of Src association with lipid and protein targets. J. Cell Biol. 178, 675–686 (2007).
Wolfenson, H. et al. A role for the juxtamembrane cytoplasm in the molecular dynamics of focal adhesions. PLoS ONE 4, e4304 (2009).
Molliex, A. et al. Phase separation by low complexity domains promotes stress granule assembly and drives pathological fibrillization. Cell 163, 123–133 (2015).
Nott, T. J. et al. Phase transition of a disordered nuage protein generates environmentally responsive membraneless organelles. Mol. Cell 57, 936–947 (2015).
Fan, R., Kim, N. G. & Gumbiner, B. M. Regulation of Hippo pathway by mitogenic growth factors via phosphoinositide 3-kinase and phosphoinositide-dependent kinase-1. Proc. Natl Acad. Sci. USA 110, 2569–2574 (2013).
Kim, N. G. & Gumbiner, B. M. Adhesion to fibronectin regulates Hippo signaling via the FAK-Src-PI3K pathway. J. Cell Biol. 210, 503–515 (2015).
Totaro, A., Panciera, T. & Piccolo, S. YAP/TAZ upstream signals and downstream responses. Nat. Cell Biol. 20, 888–899 (2018).
Yu, F. X. et al. Regulation of the Hippo-YAP pathway by G-protein-coupled receptor signaling. Cell 150, 780–791 (2012).
Panciera, T., Azzolin, L., Cordenonsi, M. & Piccolo, S. Mechanobiology of YAP and TAZ in physiology and disease. Nat. Rev. Mol. Cell Biol. 18, 758–770 (2017).
Hergovich, A. The roles of NDR protein kinases in Hippo signalling. Genes 7, 21 (2016).
Chan, S. W. et al. TEADs mediate nuclear retention of TAZ to promote oncogenic transformation. J. Biol. Chem. 284, 14347–14358 (2009).
Noland, C. L. et al. Palmitoylation of TEAD transcription factors is required for their stability and function in Hippo pathway signaling. Structure 24, 179–186 (2016).
Zhang, H. et al. TEAD transcription factors mediate the function of TAZ in cell growth and epithelial-mesenchymal transition. J. Biol. Chem. 284, 13355–13362 (2009).
Rausch, V. et al. The Hippo pathway regulates caveolae expression and mediates flow response via caveolae. Curr. Biol. 29, 242–255 (2019).
Misra, J. R. & Irvine, K. D. The Hippo signaling network and its biological functions. Annu. Rev. Genet. 52, 65–87 (2018).
Zhang, L., Yue, T. & Jiang, J. Hippo signaling pathway and organ size control. Fly 3, 68–73 (2009).
Gill, M. K. et al. A feed forward loop enforces YAP/TAZ signaling during tumorigenesis. Nat. Commun. 9, 3510 (2018).
Richardson, H. E. & Portela, M. Tissue growth and tumorigenesis in Drosophila: cell polarity and the Hippo pathway. Curr Opin. Cell Biol. 48, 1–9 (2017).
Zeng, M. et al. Phase transition in postsynaptic densities underlies formation of synaptic complexes and synaptic plasticity. Cell 166, 1163–1175 (2016).
Fang, X. et al. Arabidopsis FLL2 promotes liquid-liquid phase separation of polyadenylation complexes. Nature 569, 265–269 (2019).
Varelas, X. et al. TAZ controls Smad nucleocytoplasmic shuttling and regulates human embryonic stem-cell self-renewal. Nat. Cell Biol. 10, 837–848 (2008).
Zhang, J. M. et al. YAP-dependent induction of amphiregulin identifies a non-cell-autonomous component of the Hippo pathway. Nat. Cell Biol. 11, 1444–1450 (2009).
Kwon, I. et al. Phosphorylation-regulated binding of rna polymerase ii to fibrous polymers of low-complexity domains. Cell 155, 1049–1060 (2013).
Kato, M. et al. Cell-free formation of RNA granules: low complexity sequence domains form dynamic fibers within hydrogels. Cell 149, 753–767 (2012).
Hagenbeek, T. J. et al. The Hippo pathway effector TAZ induces TEAD-dependent liver inflammation and tumors. Sci. Signal. 11, eaaj1757 (2018).
Lu, H. et al. Phase-separation mechanism for C-terminal hyperphosphorylation of RNA polymerase II. Nature 558, 318–323 (2018).
Plouffe, S. W. et al. Characterization of Hippo pathway components by gene inactivation. Mol. Cell 64, 993–1008 (2016).
Zhu, Q. et al. SnoN antagonizes the Hippo kinase complex to promote TAZ signaling during breast carcinogenesis. Dev. Cell 37, 399–412 (2016).
Eisenberg, S. et al. Raft protein clustering alters N-Ras membrane interactions and activation pattern. Mol. Cell. Biol 31, 3938–3952 (2011).
Efron, B. & Tibshirani, R. in An Introduction to Bootstrap (eds Cox, D. R. et al.) 124–130 (Chapman & Hall, 1993).
Rashidian, J. et al. Ski regulates Hippo and TAZ signaling to suppress breast cancer progression. Sci. Signal. 8, ra14 (2015).
Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
Anders, S., Pyl, P. T. & Huber, W. HTSeq–a Python framework to work with high-throughput sequencing data. Bioinformatics 31, 166–169 (2015).
Acknowledgements
We thank K.-L. Guan and A. Mauviel for providing cDNAs of components of the Hippo pathway and H. Sasaki for the 8xGT-IIC-δ51LucII construct; J. He for technical assistance and Q. Zhu for helpful suggestions, discussions and help with experimental procedures; and D. Schichnes and S. Ruzin at the CNR biological imaging facility at the University of California, Berkeley for assistance with microscopy. This study was supported by DOD/US Army Medical Research And Materiel Command W81XWH-15-1-0068 (to K.L. and Q.Z.), a Tel Aviv University-University of California Berkeley collaborative research grant (to Y.I.H. and K.L.), and NIH R01AI41757 (to Q.Z.). Y.I.H. is an incumbent of the Zalman Weinberg Chair in Cell Biology. Y.L. is supported by the Berkeley Scholars program, and T.W. was supported by the China Scholarship Council.
Author information
Authors and Affiliations
Contributions
Y.L., T.W. and K.L. designed the research. Y.L. performed in vivo experiments. T.W. performed in vitro experiments. Y.I.H. designed and O.G. performed FRAP experiments. Y.L., T.W., H.L., Y.I.H., Q.Z. and K.L. analysed data and wrote the paper. K.L. conceived and directed the project. All of the authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Regulation of TAZ droplet formation in vitro and nuclear puncta formation in vivo.
a, GFP-TAZ purified from E. coil were analysed by SDS-PAGE and visualized by Coomassie blue staining. b. 50 μM GFP-TAZ were heated-inactivated (5 min at 95 °C and immediately put on ice for 5 min) or treated with 100 μg/ml Proteinase K for 30 min at 40 °C, and then subjected to droplet formation assay in vitro in the presence of 500 mM NaCl at room temperature. c, Ectopically expressed GFP-TAZ was expressed at a lower level than endogenous TAZ in MCF-10A cells as shown by western blotting. GAPDH was used as a loading control. d, Flag-TAZ formed nuclear puncta when transfected into the MCF-10A cells, as detected by immunofluorescence staining with anti-Flag. Scale bar, 10 μm. Experiments in a–d were repeated independently three times with similar results. Unprocessed blots are provided in Unprocessed Blots Extended Data Fig. 1.
Extended Data Fig. 2 YAP does not form droplets in vitro and in vivo in the absence of crowding agents.
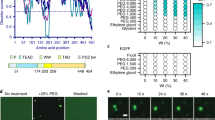
a, GFP-YAP purified from E. coil were analyzed by SDS-PAGE and visualized by Coomassie blue staining. b, GFP-YAP at varying concentrations was subjected to the droplet formation assay at room temperature and in the presence of 500 mM NaCl. c, 50 μM GFP-YAP was subjected to the droplet formation assay at room temperature in the presence of indicated salt concentrations. d, 50 μM GFP-YAP was subjected to droplet formation in the presence of 150 mM NaCl at 4 °C or 37 °C. e, Two YAP isoforms, GFP-YAP1–1β or GFP-YAP1–2α, did not form droplets (50 μM protein, 500 mM NaCl and room temperature). aa, amino acids. f, 50 μM GFP-YAP formed droplets in the presence of 10% PEG-8000, Ficoll or Dextran but not 10% glycerol or sucrose. Droplet formation assay was performed in the presence of 500 mM NaCl at room temperature. g, 50 μM GFP-YAP did not form droplets in the presence of BSA at varying concentrations. h, GFP-YAP did not form nuclear puncta in both HeLa cells and 293T cells. Scale bars, 10 μm. Experiments in a–h were repeated independently three times with similar results.
Extended Data Fig. 3 The CC and WW domains are required for TAZ to form nuclear puncta.
a, Domain structure of TAZ and TAZ truncations. The numbers above indicate the position of amino acid residues. b, Bacterially purified GFP-TAZ, ∆TB, ∆WW, ∆CC, and ∆WW+∆CC proteins were analyzed by SDS-PAGE and detected by Coomasssie blue staining. c, Localization of GFP-TAZ and various mutants in HeLa cells. d, Localization of GFP-TAZ and various TAZ/YAP chimera in HeLa cells. Scale bars, 10 μm. e, A GST pull-down assay was performed by incubating immobilized GST fusion proteins with lysates of cells expressing HA-tagged WT or mutant TAZ, and the associated TAZ proteins were detected by western blotting with anti-HA (upper). GST fusion proteins were assessed by western blotting with anti-GST, and HA-TAZ proteins in the cell lysates were measured by western blotting (lower). Experiments in b–e were repeated independently three times with similar results. Unprocessed blots are provided in Unprocessed Blots Extended Data Fig. 3.
Extended Data Fig. 4 TAZ CC domain enhances YAP phase separation in the presence of PEG.
a, Domain structure of YAP chimera. b, Substitution of the YAP CC and WW domains with that of TAZ is not sufficient to enable YAP to undergo LLPS in MCF10A cells in the absence of PEG. c, Coomasssie blue staining of various recombinant proteins purified from E. coil. d, 25 μM bacterially purified GFP-YAP chimera proteins were subjected to droplet formation assay in the presence of 10% PEG-8000. Quantification of the droplets is on the right. Scale bar, 10 μm. Data shown as the mean ± s.e.m. Statistical significance was evaluated using One-way ANOVA with Krusk-Wallis test. Droplets in n = 3 fields in each group were quantified. e, The TAZ CC and WW domains enhanced LLPS by GFP-YAP in transfected MCF10A cells in the presence of PEG as shown by confocal microscopy. Scale bar, 10 μm. Quantification of the percentage of cells that displayed nuclear puncta is shown on the right. Data shown as the mean ± s.e.m.. P value was determined by unpaired two-tailed Student’s t-test. 80 transfected cells in each group were quantified. n = 3 biologically independent samples. Experiments in b, c, e were repeated independently three times with similar results. Experiments in d were repeated twice with similar results. Statistical source data for d, e, are provided in Statistical Source Date Extended Data Fig. 4.
Extended Data Fig. 5 Hippo signaling negatively regulates TAZ phase separation in HeLa cells.
TAZ localization was examined by immunofluorescence staining with anti-TAZ (green) in HeLa cells that have been subjected to the following treatments: a, Serum-starved HeLa cells were treated with 1 μM LPA or 50 ng/ml EGF for 1 h. b, Serum-starved HeLa cells were seeded on fibronectin-coated coverslips for 10 min or 2 h in serum-free medium. c, HeLa cells were grown on fibronectin-coated polyacrylamide hydrogels of 1 kPa and 40 kPa stiffness. d, HeLa cells were treated with 1 μg/ml Latrunculin B for 1 h. Alexa Fluor 555-conjugated phalloidin (Red) staining was performed to detect F-actin in b-d. Scale bar, 10 µm. Experiments in a–d were repeated independently three times with similar results.
Extended Data Fig. 6 LATS2 regulates TAZ LLPS and recruitment of TEAD4 and BRD4.
a, MCF-10A cells transfected with GFP-TAZ-S89A and Flag-LATS2 were subjected to immunofluorescence staining with anti-Flag (Red). Scale bar, 10 µm. b, MCF-10A cells stably expressing siLATS1/2 were transfected with GFP-TAZ and Flag-TEAD4. TEAD localization at high cell density was detected by immunofluorescence staining with anti-Flag (Red). Scale bar, 10 μm. c, MCF-10A cells stably expressing siLATS1/2 were transfected with GFP-TAZ. Endogenous BRD4 localization was examined by immunofluorescence staining with anti-BRD4 (Red). Scale bar, 10 μm. All experiments were repeated independently three times with similar results.
Extended Data Fig. 7 TAZ nuclear condensates do not co-localize with the PML bodies, Cajal bodies or nucleoli.
The PML nuclear bodies, Cajal Bodies and nucleoli in MCF-10A cells expressing GFP-TAZ (green) were detected by immunofluorescence staining with antibodies targeting PML, Coilin and Fibrillarin, respectively (red). Scale bar, 10 μm. Experiments were repeated independently three times with similar results.
Extended Data Fig. 8 TAZ mutants lacking the CC domain still bind to LAST2 and TEAD4.
a, HA-tagged WT and mutant TAZ were co-transfected into 293T cells with Flag-LATS2. TAZ proteins associated with LATS2 were isolated by immunoprecipitation with anti-Flag and detected by western blotting with anti-HA antibodies (upper panels). The abundance of these proteins in the cell lysates was assessed by western blotting (lower panels). GAPDH was used as a loading control. b, Interaction of various TAZ mutants with Flag-TEAD4 was analyzed by co-IP assay as described in a. c, Interaction of various TAZ/YAP chimera with LATS2 was analyzed by co-IP as described in a. All experiments were repeated independently three times with similar results. Unprocessed blots are provided in Unprocessed Blots Extended Data Fig. 8.
Supplementary information
Source data
Source Data Fig. 1
Statistical source data.
Source Data Fig. 2
Statistical source data.
Source Data Fig. 3
Unprocessed blots.
Source Data Fig. 3
Statistical source data.
Source Data Fig. 4
Unprocessed blots.
Source Data Fig. 4
Statistical source data.
Source Data Fig. 6
Unprocessed blots.
Source Data Fig. 6
Statistical source data.
Source Data Fig. 7
Unprocessed blots.
Source Data Fig. 8
Unprocessed blots.
Source Data Fig. 8
Statistical source data.
Source Data Extended Data Fig. 1
Unprocessed blots.
Source Data Extended Data Fig. 3
Unprocessed blots.
Source Data Extended Data Fig. 4
Statistical source data.
Source Data Extended Data Fig. 8
Unprocessed blots.
Rights and permissions
About this article
Cite this article
Lu, Y., Wu, T., Gutman, O. et al. Phase separation of TAZ compartmentalizes the transcription machinery to promote gene expression. Nat Cell Biol 22, 453–464 (2020). https://doi.org/10.1038/s41556-020-0485-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41556-020-0485-0
- Springer Nature Limited
This article is cited by
-
Crosstalk between protein post-translational modifications and phase separation
Cell Communication and Signaling (2024)
-
Phase separation-mediated biomolecular condensates and their relationship to tumor
Cell Communication and Signaling (2024)
-
G-quadruplexes promote the motility in MAZ phase-separated condensates to activate CCND1 expression and contribute to hepatocarcinogenesis
Nature Communications (2024)
-
The nucleolar shell provides anchoring sites for DNA untwisting
Communications Biology (2024)
-
A chaperone-like function of FUS ensures TAZ condensate dynamics and transcriptional activation
Nature Cell Biology (2024)