Abstract
Progesterone (P4) is required for the preparation of the endometrium for a successful pregnancy. P4 resistance is a leading cause of the pathogenesis of endometrial disorders like endometriosis, often leading to infertility; however, the underlying epigenetic cause remains unclear. Here we demonstrate that CFP1, a regulator of H3K4me3, is required for maintaining epigenetic landscapes of P4-progesterone receptor (PGR) signaling networks in the mouse uterus. Cfp1f/f;Pgr-Cre (Cfp1d/d) mice showed impaired P4 responses, leading to complete failure of embryo implantation. mRNA and chromatin immunoprecipitation sequencing analyses showed that CFP1 regulates uterine mRNA profiles not only in H3K4me3-dependent but also in H3K4me3-independent manners. CFP1 directly regulates important P4 response genes, including Gata2, Sox17, and Ihh, which activate smoothened signaling pathway in the uterus. In a mouse model of endometriosis, Cfp1d/d ectopic lesions showed P4 resistance, which was rescued by a smoothened agonist. In human endometriosis, CFP1 was significantly downregulated, and expression levels between CFP1 and these P4 targets are positively related regardless of PGR levels. In brief, our study provides that CFP1 intervenes in the P4-epigenome-transcriptome networks for uterine receptivity for embryo implantation and the pathogenesis of endometriosis.
Similar content being viewed by others
Introduction
CXXC finger protein 1 (CFP1) is an important player in the epigenetic regulation of genes by inducing trimethylation at histone H3 lysine 4 (H3K4me3) with SETD1, a histone methyltransferase1. H3K4me3 is mainly found in active promoters and turns chromatin into transcriptionally active euchromatin in the transcription start site (TSS) and CpG island (CGI)2. When CFP1 binds to unmethylated CpG, it recruits SETD1A/B to trigger H3K4me3 in the promoters of target genes to increase gene expression1,3. The deletion of CFP1, SETD1A, or SETD1B in mice caused embryonic lethality during or after gastrulation, suggesting the roles of SETD1-CFP1 complexes in early mammalian development4,5. Furthermore, the conditional deletion of CFP1 highlighted that CFP1-associated H3K4me3 has fundamental roles in various biological processes. CFP1-deficient embryonic stem cells failed to differentiate in vitro because of aberrant H3K4me3 at non-methylated CGI promoters, leading to transcriptional disturbance2,4,6. CFP1 plays an important role in intrathymic T-cell development and differentiation program of TH17 cells7,8. Recent studies have also demonstrated that CFP1 is required for epigenetic modification in non-replicative cells, such as germ cells, in mice9,10,11.
The endometrium is a highly dynamic organ, and its function and cyclicity are mainly regulated by ovarian progesterone (P4) and estrogen (E2). Imbalances between P4 and E2 may cause various gynecological disorders such as endometriosis, repeated implantation failure, and endometrial cancer12. P4 and E2 activate their nuclear receptors, progesterone receptors (PGRs), and estrogen receptors (ESRs) to control the expression of local factors for uterine functions. PGR-dependent signaling networks for epithelium–stroma interaction are necessary to promote uterine receptivity for embryo implantation and decidualization in the uterus13. The fine-tuning of epigenetic modulation is required for phase-specific transcriptional networks during the reproductive cycle, embryo implantation, and subsequent pregnancy14,15,16. Dynamic changes in histone modifications occur during decidualization17,18. Silencing and overexpression of EZH2, a histone methyltransferase for H3K27me3, a repressive histone mark, disturb the expression of decidualization markers, such as IGFBP1 and PRL17. For successful parturition, biphasic modes of H3K27me3 dynamics in decidual stromal cells dictate the regulated gene silencing in the uterine adaptation to pregnancy19. The acetylation and methylation patterns of H3 and H4 in women with endometriotic lesions are distinct from those of disease-free women20. However, the physiological significance of epigenetic regulatory machineries, such as histone modification during early pregnancy and the pathogenesis of uterine disorders, remains largely unexplored.
Endometriosis is a disorder in which endometrial cells grow abnormally outside the uterus. The surgical removal of the ectopic endometrial lesion with hormonal suppression is the current standard of care; however, these therapies have a high incidence of relapse and various side effects21,22. Endometriosis affects 10%–15% of women of reproductive age and is one of the leading causes of female infertility21,22,23,24. Leading causes of endometriosis include increased E2 response, P4 resistance (decreased P4 response), and/or abnormal epigenetic regulation25,26,27,28. Although decreased P4 response caused by reduced PGR expression is considered a main cause of endometriosis26,29, the underlying cause of the decreased P4 response, even with normal P4 secretion and PGR expression, has not yet been elucidated.
Altered DNA methylation and histone modification on the genes for balanced hormone responses were mainly proposed to affect the endometrial function and the development of endometriosis9,12. The loss of histone deacetylase 3 (HDAC3) is a result of failures in embryo implantation and decidualization and caused fibrosis in the endometrium, one of the symptoms of human endometriosis with reduced HDAC3 expression25. However, epigenetic causes of how uterine cells grow outside the uterus in patients with endometriosis have not been clearly identified. Using a combination of genetic and pharmacologic tools and various analyses, we demonstrated that CFP1-dependent epigenetic regulation is necessary to maintain uterine transcriptional landscapes for P4 response for a successful pregnancy and prevent endometriosis with P4 resistance. Thus, this study provides fundamental insight into understanding the complex interplay between the P4-PGR signaling pathway and uterine epigenome–transcriptome under physiologic and pathophysiologic conditions, such as endometriosis.
Results
Loss of Cfp1 leads to infertility with multiple failures in oviductal embryo transport, P4 uterine responses, embryo implantation, and decidualization in mice
CFP1 is expressed in many cells of female reproductive organs, such as the ovary, oviduct, and uterus (Supplementary Fig. 1). During early pregnancy, uterine CFP1 expression gradually increased from day 1 of pregnancy (Day 1) to Day 3. It peaked on Day 4, with the highest expression in the luminal epithelium. However, its expression is not directly affected by the regulation of E2 and/or P4 in the mouse uterus (Supplementary Fig. 1). While Cfp1 is deleted in most cell types in the female reproductive tract of adult Cfp1f/f;Pgrcre/+ (Cfp1d/d) mice (Supplementary Fig. 2), they showed normal architectures in the gross morphology and histology of the reproductive tract with regular estrous cycle (Supplementary Fig. 3a–c). Furthermore, serum levels of E2 and P4 on Day 4 in Cfp1d/d mice were comparable to those of Cfp1f/f mice. We also found that Cfp1d/d mice ovulate similar numbers of oocytes that can fertilize normally, and the fertilized embryos develop to the blastocyst stage without any aberrations in vitro (Supplementary Fig. 3d–f). However, implantation sites (IS), blue bands along the uterus, were not observed in Cfp1d/d female mice on Day 5 (Fig. 1a). In addition, Cfp1d/d uteri did not show decidual responses to artificial stimuli, such as oil (Fig. 1b, c). Accordingly, Cfp1d/d female mice did not produce any pups (Fig. 1d). Interestingly, all blastocysts were found in the oviduct but not in the uterus of Cfp1d/d mice on Day 4, whereas they were found in the uterus of Cfp1f/f mice as expected (Fig. 1e–g).
a Representative photographs of uteri with IS (black arrowheads) in Cfp1f/f and Cfp1d/d mice on Day 5. n = 4 to 5 biologically independent samples per genotype. Data are presented as mean values with SD. Statistical analyses were performed using the unpaired Student’s t-tests. **p < 0.01. b Artificial decidualization responses in hormone primed Cfp1f/f and Cfp1d/d OVX mice. The decidual response was determined by the uterine weight of the oil-injected (black arrowheads)/non-injected uterine horn. n = 3 to 4 biologically independent samples per genotype. Data are presented as mean values with SD. Statistical analyses were performed using the unpaired Student’s t-tests. **p < 0.01. c Microscopic images of alkaline phosphatase staining of artificially decidualized Cfp1f/f and Cfp1d/d uteri. Scale bar, 100 µm. d Litter size of Cfp1f/f and Cfp1d/d female mice that were mated with fertile male mice for 8–10 weeks. The numbers above the bars indicate the number of mice with litter/total number of mice examined in each group. Data are presented as mean values with SD. Statistical analyses were performed using the unpaired Student’s t-tests. **p < 0.01. e–h Impairment of the embryo transport from the oviduct to the uterus in Cfp1d/d female mice. Percentage graph (e) and microscopic images (g) of embryos recovered from uteri and/or oviducts in Cfp1f/f and Cfp1d/d mice on Day 4. Scale bar, 50 µm in (f). g Schematic image of Cfp1d/d oviduct in the morning of Day 4. Representative histological images of Cfp1d/d oviduct (ampulla and isthmus) where blastocysts were found even in the morning of Day 4. Scale bar, 50 µm. Figure was created with BioRender.com. h Experimental scheme of embryo transfer. i Representative photographs of uteri with IS (black arrowheads) in pseudopregnant Cfp1f/f and Cfp1d/d recipients after transferring wildtype blastocysts. Scale bars, 50 µm. Figure was created with BioRender.com. j, k Immunofluorescent staining of KI67 to examine uterine cell proliferation in Cfp1f/f and Cfp1d/d mice on Day 4. Scale bar, 50 µm. Bl blastocyst, S stroma, M muscle cells, E epithelium, GE glandular epithelium, LE luminal epithelium. n = 6 to 7 biologically independent samples per genotype. Data are presented as mean values with SD. Statistical analyses were performed using the unpaired Student’s t-tests. **p < 0.01.
To examine embryo implantation in the uterus of Cfp1d/d mice with defective oviductal embryo transport, wildtype blastocysts were transferred to the uteri of Cfp1f/f and Cfp1d/d mice on day 4 of pseudopregnancy. Distinct IS were observed in Cfp1f/f but not in Cfp1d/d recipients, and unimplanted blastocysts were retrieved from Cfp1d/d recipients 24 h after embryo transfer (Fig. 1h, i and Table 1), indicating that CFP1-deficient uterine environments do not support embryo implantation. In this aspect, cell proliferation profiles are aberrant in the uterus of Cfp1d/d mice on Day 4 (Fig. 1j, k), whereas the levels of E2 and P4 (Supplementary Fig. 3d) and their receptors (Supplementary Fig. 4) are comparable between Cfp1f/f and Cfp1d/d mice. Nevertheless, the uterine epithelium persistently proliferates, and the stroma showed less proliferation potential in the uterus of Cfp1d/d mice, suggesting that CFP1, as an epigenetic regulator, is required for proper hormone responses in the uterus for a successful pregnancy.
CFP1 regulates uterine mRNA landscapes in both H3K4me3-dependent and H3K4me3-independent manners
To understand the CFP1-dependent epigenetic regulation on transcriptional landscapes in the uterus, we performed chromatin immunoprecipitation sequencing (ChIP-seq) and mRNA sequencing (mRNA-seq) with the uteri of Cfp1f/f and/or Cfp1d/d mice on Day 4 (Fig. 2). ChIP-seq with antibodies for CFP1 and H3K4me3 provided evidence that CFP1-binding sites are highly enriched in TSS and CGI, and H3K4me3 in TSS and CGI was generally reduced in the uterus of Cfp1d/d mouse (Fig. 2a). De novo motif analysis for CFP1 ChIP-seq data showed that CFP1 recognizes the CCGG motifs, including CGG and its reverse complement CCG (Fig. 2b), which are consistent with the CFP1-binding motifs found in humans30,31,32. CFP1 binding was noticeably enriched in extended gene bodies, including promoters (9%), exons (4%), and introns (38%) in mouse uterus, considering that the mouse genome consists of promoters (2%), exons (2%), introns (20%), and intergenic factors (76%) (Fig. 2c). When the mRNA-seq and H3K4me3 ChIP-seq data in ±2 kilo base pairs (Kbp) of TSS were analyzed together, 40.3% of differentially expressed genes (DEGs) had a reduction in both gene expression and H3K4me3 levels in Cfp1d/d mice (Fig. 2d) as expected from the known actions of CFP1 on H3K4me3. Generally, the lower gene expression levels are, the lower the H3K4me3 levels in the +2Kbp TSS region are (Supplementary Fig. 5). However, a significant portion of DEGs (18.2%) was made up of genes with decreased expression levels and increased H3K4me3 levels in Cfp1d/d mouse uterus on Day 4, suggesting that CFP1 could promote gene expression in the uterus in H3K4me3-independent manner.
a Tag density of CFP1 binding and H3K4me3 peaks were calculated on ±3 Kbp window centered on TTS and CGI regions of all RefSeq (mm10) genes in Cfp1f/f and/or Cfp1d/d mouse uteri on Day 4 for heatmap and graph data. b CFP1-binding sequence logo of the top 10 motifs identified using de novo motif discovery. c Distribution of the genetic features across the mouse genome and CFP1-binding peaks in Cfp1f/f mouse uterus on Day 4. d Correlation between H3K4me3 promoter (TSS ± 2 Kbp) enrichment conditions and gene expression in the mouse uterus on Day 4 (Cfp1d/d versus Cfp1f/f). Each dot represents a differentially expressed gene with statistical significance (p < 0.05, normalized data average, log2 > 3). e Volcano plot to compare expression profiles from the Cfp1f/f vs. Cfp1d/d in the uterus on Day 4. f Pie chart summarizing upregulated or GO term in Cfp1f/f versus Cfp1d/d mouse uterus on Day 4. g GSEA to identify downregulated GO term and curated gene sets in Cfp1f/f versus Cfp1d/d mouse uterus on Day 4. Gene sets with an FDR q-value of <0.25 (red dotted line) were considered significant. h GSEA enrichment plot and heatmap of the “GO Regulation of smoothened signaling” gene set from RNA-seq data of Cfp1f/f and Cfp1d/d mouse uterus on Day 4. The color spectrum from blue to red indicates low to high expression. i RT-PCR and Real-time RT-PCR analyses for IHH-dependent SSP genes (red colored in h). n = 8 biologically independent samples per genotype. Data are presented as mean values with SD. Statistical analyses were performed using the unpaired Student’s t-tests. **p < 0.01.
Although CFP1 binds to unmethylated CpG and induces H3K4me3 to increase gene expression1,3, the number of significantly downregulated (3329) and upregulated (2829) genes in Cfp1d/d mice was comparable (54% vs. 46%) in DEGs from mRNA-seq (Fig. 2e and Supplementary Fig. 6a). However, gene ontology (GO) analyses showed that most GO terms (126/132, 95.5%) with false discovery rate (FDR) of <0.25 were reduced in the uteri of Cfp1d/d mice on Day 4 (Fig. 2f and Supplementary Fig. 6b). Gene set enrichment analyses (GSEA) showed that various gene sets associated with P4 response, hedgehog signaling, cancers, ion channel activities, and stromal cell stimulation were significantly downregulated in Cfp1d/d uterus (Fig. 2g). Interestingly, the Indian hedgehog (IHH)-dependent smoothened signaling pathway (SSP) is a well-known P4 downstream pathway that inhibits E2-dependent epithelial proliferation and further stimulates stromal proliferation in the uterus33. A heatmap, reverse-transcriptase polymerase chain reaction (RT-PCR), and real-time RT-PCR analyses for the SSP gene set validated that IHH-dependent SSP is significantly downregulated in Cfp1d/d mouse uterus on Day 4 (Fig. 2h, i).
CFP1 epigenetically regulates P4 responses through the IHH-dependent SSP in mouse uterus
To further investigate the epigenetic actions of CFP1 in the uterus, we tried to identify CFP1 direct target genes by analyzing all sequencing data together. The CFP1 direct target gene candidates should have CFP1-binding site(s) and be downregulated in Cfp1d/d uteri. We found that the putative direct target genes of CFP1 are regulated not only in H3K4me3-dependent (673 genes) but also inH3K4me3-independent (423 genes) manner (Fig. 3a and Supplementary Data 1–2). Interestingly, the upstream and downstream genes of IHH-dependent SSP were included in the list; Ihh, Gli3, and Gata2 as H3K4me3-dependent and Ptch1, Sox17, and Nr2f2 as H3K4me3-independent target genes (Fig. 3b and Supplementary Data 2). The expression of these genes was reduced with statistical significance in Cfp1d/d mice (Figs. 2i and 3c). The visualization of sequencing data for Gata2, Ihh, and Sox17 using Integrative Genomics Viewer demonstrated that they all have CFP1 binding sites, and their expression levels were significantly reduced in Cfp1d/d uteri. However, H3K4me3 levels were decreased in extended gene bodies of Gata2 and Ihh, but not in Sox17 (Fig. 3d). Real-time ChIP PCR reinforced that SETD1 and H3K4me3 were enriched in the promoter regions of Gata2 and Ihh but not in Sox17, whereas CFP1 is enriched in all of them in Cfp1f/f uterus (Fig. 3e and Supplementary Table 1). In summary, CFP1 works with SETD1 to increase H3K4me3 in Gata2 and Ihh promoters but not for Sox17 promoter for their expression in the uterus.
a Venn diagram of identifying genes that overlapped in datasets between CFP1-binding sites on their extended gene bodies (2 Kbp upstream and 200 bp downstream of TSS and CGI), downregulated gene in Cfp1d/d mice (>1.5 folds), and downregulated H3K4me3 sites in Cfp1d/d mice (>1.5 folds) on Day 4. b Schematic cartoon to show the epithelium–stroma crosstalk mediated by SSP. c Real-time RT-PCR analyses of upstream and downstream genes in IHH-dependent SSP (Gata2, Sox17, Nr2f2, Hand2, Fgf2, and Fgf18). n = 8 biologically independent samples per genotype. Data are presented as mean values with SD. Statistical analyses were performed using the unpaired Student’s t-tests. *p < 0.05, **p < 0.01. d Integrative Genomics Viewer screenshots that show the distribution of CFP1 binding, H3K4me3 site, and RNA expression intensity in Gata2, Sox17, and Ihh of Cfp1f/f and Cfp1d/d mouse uterus on Day 4. e Real-time ChIP PCR for detecting CFP1, SETD1, and H3K4me3, binding on Gata2, Sox17, and Ihh in the uterus of Cfp1f/f and Cfp1d/d mice on Day 4. n = 3 biologically independent samples per genotype. Data are presented as mean values with SD. Statistical analyses were performed using the unpaired Student’s t-tests. *p < 0.05, **p < 0.01. f Immunofluorescent staining of KI67 in the uterus of the OVX Cfp1f/f and Cfp1d/d mice treated with E2 and/or P4 for 24 h. At 3 h after E2 or E2 + P4, one uterine horn was injected with SAG and the other one with PBS. Green and red indicate KI-67 and the nucleus, respectively. S stroma, GE glandular epithelium, LE luminal epithelium. Scale bar, 50 µm. g Percentages of KI67-positive cells in (f). n = 4 biologically independent samples per genotype. Data are presented as mean values with SD. Statistical analyses were performed using the unpaired Student’s t-tests. *p < 0.05, **p < 0.01. h Real-time RT-PCR analyses of SSP genes (Gli1, Gli2, Nr2f2, and Hand2) 3 h after SAG injection. n = 8 biologically independent samples per genotype. Data are presented as mean values with SD. Statistical analyses were performed using the multiple comparisons. *p < 0.05, **p < 0.01.
P4-PGR induces stromal cell proliferation and inhibits epithelial cell proliferation via the activation of SSP downstream of Gata2, Sox17, and Ihh in mouse uterus on Day 4 (Fig. 3b). However, P4 could not inhibit E2-dependent epithelial proliferation and facilitate stromal cell proliferation in the uteri of ovariectomized (OVX) Cfp1d/d mice (Fig. 3f, g) in line with the results on Day 4 (Fig. 1k). Since defective SSP could cause aberrant cell proliferation in Cfp1d/d uteri, we tried to rescue this phenotype with SAG, a smoothened agonist, in these mice. An intrauterine delivery of SAG successfully rescued aberrant epithelial proliferation in the uteri of OVX Cfp1d/d mice treated with E2 and P4 (Fig. 3f and 3g). Furthermore, SAG significantly restored the decreased expression levels of genes in SSP and its downstream genes, such as Gli1, Gli2, Nr2f2, and Hand2, in Cfp1d/d uteri to the levels in Cfp1f/f uteri (Fig. 3h). When our mRNA-seq datasets were compared with other P4-related transcriptomic data (GSE118264, GSE40661, and GSE178541)13,34,35, comparative analyses showed that significant numbers of DEGs in Cfp1d/d uterus overlap with DEGs in other P4-related datasets (Supplementary Fig. 7). Collectively, these results suggest that CFP1 loss disturbs the epigenetic maintenance of P4-PGR signaling pathways in the uterus.
CFP1 is required for P4 function to inhibit the growth of ectopic endometriotic lesions in mice
Endometriosis can occur possibly via a decreased P4 response, and some patients with endometriosis show P4 resistance even with normal PGR expression36,37,38,39. To further evaluate CFP1 function for uterine P4 responses, we established a mouse model of endometriosis with some modifications from previous reports25,26. When P4 was given with E2, the size of ectopic lesions was significantly smaller than with E2 alone, although the number of ectopic lesions was not different (Supplementary Fig. 8a, b). Ectopic lesions were mainly observed on organs with highly developed blood vessels, such as the small intestine, kidney, uterus, and fat pad (Supplementary Fig. 8c). When small pieces of Cfp1f/f and Cfp1d/d uterus as endometriotic lesions were transplanted to wildtype recipients (Fig. 4a), P4 effectively suppressed the E2-induced growth of Cfp1f/f but not Cfp1d/d ectopic lesions even if the number of ectopic lesions was not affected by Cfp1 genotypes (Fig. 4b–e). The mRNA expression of Gata2, Sox17, and Ihh was also significantly downregulated in Cfp1d/d ectopic uterine lesions (Fig. 4f). When SAG was administered to rescue P4 resistance and/or insensitivity in Cfp1d/d ectopic uterine lesions, P4 suppressed the size of Cfp1d/d ectopic lesions in the endometriosis model (Fig. 4b–e), suggesting that CFP1 is required for proper P4 responses to suppress ectopic growth of uterine tissues in mice.
a Schematic protocol to induce an experimental model of human endometriosis with transplantation of Cfp1f/f or Cfp1d/d uterine fragments. b–e Representative photographs to show the in vivo locations (b) and size (c) of ectopic lesions (dashed circles) harvested 15 days after transplantation of small pieces of Cfp1f/f and Cfp1d/d uterine tissues in recipients treated with E2, E2 + P4 + PBS, or E2 + P4 + SAG. Scale bar, 5 mm. Average volumes (d) and numbers (e) of ectopic lesions collected from recipients. n = 5 to 6 biologically independent samples per group. Statistical analyses were performed using the multiple comparisons. *p < 0.05, **p < 0.01. f Real-time RT-PCR analyses of Cfp1, Gata2, Sox17, and Ihh expression in Cfp1f/f and Cfp1d/d ectopic uterine lesions in recipients treated with E2 and P4. n = 3 biologically independent samples per genotype. Data are presented as mean values with SD. Statistical analyses were performed using the unpaired Student’s t-tests. *p < 0.05, **p < 0.01.
Downregulation of the epigenetic factor CFP1 may be associated with endometriosis in humans
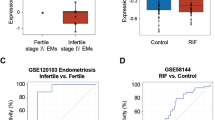
To further evaluate the potential actions of CFP1 on the pathogenesis of endometriosis in humans, we analyzed datasets (GSE51981) of the endometria of patients with endometriosis (endometriosis group, n = 77) and healthy women (control group, n = 71) from a previous study40. While no correlation exists between the expression levels of CFP1 and PGR mRNAs, GATA2, SOX17, and IHH mRNA expression levels were positively correlated with that of CFP1 (Fig. 5a) and PGR mRNA (Supplementary Fig. 9a) regardless of endometrial pathologic conditions. We further analyzed a subset of GSE51981, i.e., endometria of women with mild and severe endometriosis in the mid-secretory phase where P4 is dominant (Fig. 5b). In patients with mild endometriosis, the expression profiles of PGR, CFP1, and P4 target genes are not different from those of the control group. However, there was a statistically significant reduction in their expression levels in patients with severe endometriosis (Supplementary Fig. 9b). Among patients with severe endometriosis, some had a comparable level of PGR but a low level of CFP1 (circled red, Fig. 5b). In these patients, the expression patterns of GATA2, SOX17, and IHH mRNAs were significantly decreased. It suggests that aberrantly reduced expression of CFP1 may be associated with endometriosis via abnormal P4 response in patients with normal PGR expression. We also performed immunohistochemistry and real-time RT-PCR for these genes in endometrial samples from patients with severe endometriosis and disease-free control women in the secretory phase. The eutopic and ectopic endometria from patients with endometriosis showed decreased expression levels of CFP1, GATA2, SOX17, and IHH mRNAs compared with the endometrium from the control group (Fig. 5c, d), suggesting that reduced CFP1 expression may disturb epigenetic landscapes that mediate P4-PGR signaling pathways in human endometrium, often leading to endometriosis.
a Scatterplot of the positive correlations between mRNA expression levels of CFP1 and important P4-responsive genes, GATA2, SOX17, and IHH, in human endometrium of healthy women (control, blue dots) and patients with mild (green) or severe (red) endometriosis (data from GSE51981). b Positive correlations were observed between the expression levels of CFP1 and GATA2, SOX17, and IHH in the human endometrium in the med-secretory phase in (a). Note that a subgroup of patients with severe endometriosis had lower expression of CFP1 and important P4-responsive genes without PGR reduction (circled red dots). r indicates Pearson correlation coefficient. c–d Immunohistochemical staining (c) and real-time RT-PCR (d) of CFP1, GATA2, SOX17, and/or IHH in the human endometrium of healthy women (control) and patients with severe endometriosis (eutopic and ectopic) on the secretory phase. n = 4 to 5 biologically independent samples per group. Scale bar, 100 µm. Data are presented as mean values with SD. Statistical analyses were performed using the multiple comparisons. *p < 0.05, **p < 0.01.
Discussion
Many gynecological diseases are caused by abnormally reduced P4 response; however, the underlying epigenetic aberration for the imbalanced steroid hormone responses has not been clearly elucidated. We demonstrate that CFP1-associated epigenetic regulation is required for maintaining appropriate P4 responses for embryo implantation and decidualization in the uterus and inhibiting the ectopic growth of endometrial lesions outside the uterus. Cfp1d/d mice exhibit a wide spectrum of infertility, including defective oviductal embryo transport, abnormal uterine cell proliferation, and complete failure of embryo implantation and decidualization (Fig. 1). These phenotypes suggest that epigenetic regulation through H3K4me3 is involved in the sequential events of female reproduction. A recent study supported those spatiotemporal dynamics of H3K4me3 in the uterine genome are needed to be well controlled for early pregnancy. In mice without MENIN (Men1d/d mice), a member of the H3K4 methyltransferase complex, defective decidualization compromised fertility, whereas embryo implantation normally occurs41. Impaired decidualization in Men1d/d mice was caused by reduced Bmp2 expression by abnormally increased FGF2 signaling41. In Cfp1d/d uterus on Day 4, Fgf2 expression aberrantly increased because of a decrease in Ihh-dependent SSP that antagonizes FGF2 signaling, although the Bmp2 expression did not decrease (Fig. 3). While MLL1/2 complexes that contain MENIN catalyze H3K4 methylation in a gene- and cell-specific manner, SETD1–CFP1 complexes are the leading H3K4 methyltransferases among the six histone methyltransferases, SETD1A/B, MLL1/2, and MLL3/49,41. This is consistent with the fact that Cfp1d/d mice show a wider spectrum of infertile phenotypes than Men1d/d mice, suggesting that CFP1-dependent epigenetic regulation may work on the wider area of the genome. For example, we found an impairment of the oviductal embryo transport in Cfp1d/d mice, which was not observed in Men1d/d mice. Embryo transport in the oviduct could be interrupted by abnormalities in cilia movement, fluid secretion, and smooth muscle contraction42,43. Since Cfp1 was mainly deleted in non-ciliated epithelial cells of the isthmus, but not in ciliated ones, which are positive for acetylated tubulin in the ampulla of Cfp1d/d oviduct by Pgr-Cre (Supplementary Fig. 2e, f), embryo retainment in the oviduct could result from abnormalities in fluid secretion and smooth muscle contraction44, but not from cilia movement. While the epigenetic changes for fluid secretion are largely unknown, histone marks to promote transcription, such as H3K4me3 and H3K27 acetylation (H3K27ac), are enriched at promoters of genes driving muscle contraction on the advance of labor onset in the myometrium45.
CFP1-dependent epigenetic regulation has been investigated in various biological events. The conditional deletion of Cfp1 in mouse hematopoietic cells resulted in severe defects during hematopoiesis with complete loss of lineage-committed progenitors and mature cells46. CFP1 is also required for thymocyte survival, the balanced differentiation between Th17 and Treg cells7,8, and the phagocytic and bactericidal activity of macrophages47. Since Pgr is expressed in various immune cells, including macrophages48, dendritic cells49, and T cells50, Cfp1 was supposed to be deleted in immune cells as well as uterine cells in Cfp1d/d mice. Immune cells play important roles during pregnancy51. However, immune-related gene sets were not significantly altered in Cfp1d/d mouse uterus on Day 4 (Fig. 2g). Furthermore, normal fertility was observed in mice (Cfp1f/f;LysMcre/+) in which Cfp1 is deleted in the myeloid lineage cells, such as monocytes and macrophages, using LysM-Cre (Supplementary Table 2), suggesting that phenotypes observed in Cfp1d/d mice are not directly associated with CFP1 functions in immune cells. Recently, the function of CFP1–SETD1 complexes for epigenetic reprogramming during germ cell development has been investigated. CFP1-mediated H3K4me3 is required for maintaining chromatin accessibility for transcriptional activities during oocyte development, and oocyte-specific deletion of Cfp1 caused reduced H3K4 methylation levels and globally downregulated transcription activities, in turn, leading to multiple defects in the meiotic division and maternal–to–zygotic transition10. Furthermore, CFP1 participates in regulating the expression of paracrine factors for communication between the oocyte and surrounding granulosa cells for follicle growth and ovulation52. The conditional deletion of Setd1b in the oocyte also caused the dysregulation of transcription factors for oogenesis, including Obox transcription factors, leading to oocyte maturation defects and infertility53. CFP1 deletion before the onset of meiosis with Stra8-Cre in male mice caused complete infertility with the spermatogenic arrest at the MII stage11, suggesting that CFP1-mediated H3K4me3 plays a role in meiosis and cell fate decision.
P4 antagonizes E2 action on epithelial proliferation and promotes stromal cell proliferation to prepare uterine receptivity for embryo implantation and decidualization13,54. P4 increased the expression of Gata2, Sox17, and Ihh in epithelial cells to trigger SSP in the stroma on Day 4 in mice13,33,34, all of which were downregulated in Cfp1d/d mice (Figs. 2 and 3). As a result, uterine epithelial cells aberrantly proliferated and failed to prepare embryo implantation on Day 4 in Cfp1d/d mice (Fig. 1). The expression of gene sets related to P4 response, IHH-dependent SSP, and stromal cell stimulation was significantly reduced in Cfp1d/d mice (Fig. 2g). Furthermore, SAG successfully rescued abnormal uterine cell proliferation in Cfp1d/d mouse uterus (Fig. 3). They all indicate that disturbed P4–PGR–SSP signaling pathway is the main cause of complete failure of embryo implantation and decidualization in Cfp1d/d mice. In fact, mice without Gata2, Sox17, or Ihh phenocopied all these uterine defects observed in Cfp1d/d mice13,33,34. In addition, SAG restored decidualization and abnormal uterine cell proliferation in Ihh cKO mice33. PGR is decreased in Gata2-deficient uteri, but it is necessary to mention that mice deficient in Sox17, Ihh, or Cfp1 had normal PGR expression. Although reduced PGR expression appears to be the main cause of P4 resistance that contributes to the pathogenesis of endometriosis29,38,55, some studies have reported comparable levels of PGR in eutopic36,38 or ectopic56 endometria of women with endometriosis. This suggests that the epigenetic aberration in PGR downstream pathways could be involved in the pathogenesis of endometriosis57. Essentially, the activation of PGR downstream pathways with SAG rescued the abnormal epithelial proliferation in Cfp1d/d uterus (Fig. 3) and suppressed the ectopic growth of Cfp1d/d uterine lesions with P4 resistance in the endometriosis model (Fig. 4). Aberrant epithelial proliferation was also found in the endometrium of patients with endometriosis25,58. Considering impaired P4-PGR signaling without PGR reduction (Supplementary Fig. 4) in Cfp1d/d mice reflects P4 resistance, disturbed CFP1-associated H3K4me3 could contribute to P4 resistance that often leads to endometriosis in humans.
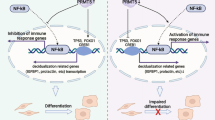
CFP1 mainly works to increase the expression of target genes through H3K4me3-dependent manners7,52. Accordingly, 40.3% of DEGs in Cfp1d/d uteri were downregulated with reduced H3K4me3 levels (Fig. 2d). However, a substantial portion of downregulated DEGs had even higher H3K4me3 levels (18.2%). Although CFP1 is an evolutionally conserved epigenetic regulator to work with SETDs for H3K4me3 from yeast to mammals, recent studies have suggested that CFP1 could collaborate with epigenetic modulators other than COMPASS (complex associated with SET1) complexes, such as DNA methyltransferases (DNMTs) and HDACs, in a context-dependent manner. When CFP1/SETD1 or MLL1/2 binds to DNA, they hamper DNA methylation by blocking the access of DNMT3A59. CFP1 interacts with and recruits DNMT1 to suppress aberrant transcription re-initiation or silence specific genes2,60. The expression of Dnmt1 and Dnmt3a increased gradually in the mouse uterus during early pregnancy61. They suggest that the H3K4me3-independent gene expression in Cfp1d/d uteri could be associated with reduced DNA methylation. In addition to DNMTs, CFP1 could interact with HDAC1/2 complexes to regulate fertility and development in C. elegans. CFP1 recruits the HDAC complex to H3K4me3-rich promoter regions to deacetylate chromatin62. CFP1-dependent H3K4me3 is necessary to recruit histone acetylase(s) for H3K9ac dynamics in mouse embryonic stem cells6, suggesting that CFP1-associated H3K4me3 cross-talks with histone acetylation. Interestingly, higher P4 levels in in vitro fertilization cycles on the day of hCG administration altered various epigenetic marks, including H3K9ac, H3K9me2, and H3K27me3, in the endometrium63, suggesting that histone modifications and P4-PGR signaling influence each other in the endometrium. The epithelial cells in endometriotic lesions expressed a higher level of EZH2, the enzyme responsible for a repressive mark H3K27me3, which P4 upregulates64. MLL1 is directly regulated by P4–PGR signaling in the uterus and MLL1 and H3K4me3 both decreased in the eutopic endometrium of patients with endometriosis65. Furthermore, the expression of HDAC3, one of HDACs that reduce gene expression, was significantly lower in the endometrium of patients with endometriosis. The loss of HDAC3 in mice leads to infertility that results from embryo implantation failures with defective decidualization possibly through the aberrant activation of Col1a1 and Col1a2 genes that promote fibrosis with decreased ESR and PGR in the uterus25. In summary, our results suggest that aberrant epigenetic regulation in CFP1-deficient mice provides a uterine environment with P4 resistance that leads to infertility caused by multiple failures and endometriosis (Fig. 6). This study is of great significance to provide an underlying epigenetic mechanism of P4 resistance in the endometrium that could lead to endometriosis in humans.
Methods
Animals
All mice used in this study were maintained in accordance with the policies of the CHA University Institutional Animal Care and Use Committee (No150083). Adult (8–10 weeks of age) C57BL/6 mice, provided by Orient Bio (Gapyeong, Gyeonggi, Korea), were housed under temperature- and light-controlled conditions with the light on for 12 h daily and fed ad libitum. Cfp1f/f mice were kindly provided by Dr. David G. Shkolnik’s laboratory1. First, Cfp1f/f mice were mated to Pgrcre/+ mice to generate Cfp1f/+;Pgrcre/+ mice66. Then, these mice were crossed to generate Cfp1d/d and Cfp1f/f mice. Genotyping PCR was performed by genomic DNA extracts from tail biopsies (Supplementary Table 3).
Vaginal smear analysis and fertility test
Estrous cyclicity was evaluated in mature Cfp1f/f and Cfp1d/d female mice by daily analysis of vaginal smears over 2 weeks between 8:00 and 9:00 AM. The estrous cycle stage (proestrus, estrus, metestrus, and diestrus) was determined based on the presence of vaginal cornified epithelial cells and nucleated epithelial cells/total vaginal cells. To evaluate reproductive performance, mature Cfp1f/f and Cfp1d/d female mice (n = 4 per genotype) were individually bred to wildtype males with proven fertility. The numbers of litters and pups were recorded for 2 months.
Serum E2 and P4 level measurement
Blood samples were collected on Day 4 (9:00 AM) from the heart of Cfp1f/f and Cfp1d/d female mice (n = 5 to 9 per genotype). First, the mouse blood was sampled without anticoagulant and transferred to a sterile empty tube. Next, the mouse blood was centrifuged at 1500 × g for 15 min at 4 °C. Separated serum was transferred into a new sterile empty tube. Serum levels of mouse E2 and P4 were measured by radioimmunoassay67.
Ovulation, fertilization, and preimplantation embryo development
Cfp1f/f and Cfp1d/d female mice were bred to wildtype males with known fertility. The morning of the vaginal plug observation was designated as Day 1. The mice were sacrificed on Day 2, and their oviducts were flushed to evaluate the number and fertilization potential of ovulated oocytes (n = 7 per genotype). In addition, fertilized 2-cell embryos were cultured up to the blastocyst stage in 20 ml droplets of KSOM (Millipore, Danvers, MA, USA) covered with oil (SAGE In-Vitro Fertilization, Inc., Trumbulla, CT, USA) in a petri dish.
Hormone treatments
To investigate time-dependent actions of E2 or P4 on the expression of Cfp1 in C57BL/6 mice uterus, adult (8–10 weeks of age) female mice were OVX, rested for 14 days, and then subcutaneously injected with either vehicle (sesame oil, 0.1 mL/mouse; Acros, NJ, USA), E2 (100 ng/mouse, Sigma–Aldrich, St. Louis, MO, USA) or E2 + P4 (2 mg/mouse, Sigma–Aldrich). After hormone injection, the mice were sacrificed at various time points (0–24 h) and the uterus was collected for real-time RT-PCR (n = 4 to 6 per each group).
Tissue preparation
Female reproductive organs under various conditions, such as early pregnancy, ovarian steroid hormone treatment, and artificial decidualization, were dissected and then fixed in 4% paraformaldehyde. Fixed tissues were washed, dehydrated, and embedded in paraplast (Merck KGaA, Darmstadt, Germany). Paraffin-embedded tissues were sectioned to 5 μm thickness using a microtome, stained with hematoxylin and eosin (Sigma–Aldrich), and observed by light microscopy.
Early pregnancy and embryo implantation
Pregnancy was evaluated by the presence of a vaginal plug on the next morning after breeding with a fertile male. The pregnant mice were sacrificed on various days of pregnancy from Day 1 to 5, and their uterine horns were collected and processed for the following experiments (n = 4 to 6 per each group). IS were detected by intravenous (i.v.) injection of 1% Chicago Sky Blue (in saline) on the morning of Day 5, and the number of IS demarcated by blue spots was recorded68.
Embryo transfer
Embryo transfer was performed as previously described with some modifications69. Four-week-old C57BL/6 mice were given intraperitoneal (i.p.) injections of 5 IU PMSG (Sigma–Aldrich) followed by i.p. injections with 5 IU hCG (Sigma–Aldrich) and then mated with fertile male mice to obtain the embryos for embryo transfers. The blastocysts were transferred to the uteri of either pseudopregnant day 4 Cfp1f/f and Cfp1d/d recipient mice, which were mated with vasectomized C57BL/6 male mice. Embryo implantation was evaluated by an i.v. injection (0.1 mL/mouse) of Chicago Sky Blue (1% in saline, Sigma–Aldrich) 24 h after embryo transfer.
Artificial decidualization
Artificial decidualization was experimentally induced, as previously described, with some modifications69. Adult (8–10 weeks of age) Cfp1f/f and Cfp1d/d female mice were OVX and rested for 14 days and then received daily injections of 100 ng E2 for 3 days (n = 3 to 4 per genotype). After 2 days of resting, the mice were then treated with daily injections of 1 mg P4 and 10 ng E2 for 3 days. At 6 h after the last injection, one uterine horn was traumatized by the injection of 50 μL of sesame oil. Mice were given daily injections of P4 (1 mg) + E2 (10 ng) following trauma. The weight of stimulated and non-stimulated uterine horns was recorded 4 days after the oil infusion. The fold increase in uterine weights and alkaline phosphatase staining served as an index of decidualization.
Intrauterine delivery of SAG
SAG (Abcam, Cambridge, UK), a smoothened agonist, was applied to rescue aberrant uterine cell proliferation and gene expression in Cfp1d/d female mice33. Adult (8–10 weeks of age) Cfp1f/f and Cfp1d/d female mice were OVX and rested for 14 days. Each OVX mouse was treated with 2 mg P4 for 2 days and then 2 mg P4 and E2 60 ng/mouse on the third day. At 3 h after E2 and P4 injection, 10 μL of vehicle (PBS) and SAG (1000 nM) in PBS were injected intraluminally in each horn of the uterus. Mice were sacrificed at 3 or 21 h after SAG injection depending on experimental conditions, and the uterus was collected for real-time RT-PCR and immunofluorescence staining (n = 8 per each group).
RNA extraction, RT-PCR, and real-time RT-PCR
Total RNA was extracted from mouse uteri under various conditions (n = 3 to 8 per each group) using Trizol Reagent (Invitrogen Life Technologies, San Diego, CA, USA) according to the manufacturer’s protocols. The first-strand cDNA was synthesized from 1 µg of total RNA using M–MLV reverse transcriptase (Promega, Madison, WI, USA) and RNasin Ribonuclease inhibitor (Promega). Synthesized cDNA (10 ng) was utilized for PCR with specific primers at optimized cycles (Supplementary Table 4). To quantify expression levels, real-time RT-PCR was performed using the SYBR green dye (iQ SYBR Green Supermix, Bio-Rad, Hercules, CA, USA), as previously described68. To compare transcript levels between samples, a standard curve of cycle thresholds for several serial dilutions of a cDNA sample was established and then used to calculate the relative abundance of each gene. Then, values were normalized to the relative amounts of Rpl7 cDNA. All reactions were performed in duplicates.
Western blotting
Uterus tissues (n = 4 to 6 per each group) were lysed in lysis buffer including PRO-PREP (iNtRON, Seongnam, Korea) solution and 1X phosphatase inhibitor (Roche Applied Sciences, Indianapolis, IN, USA). The protein samples (20 μg/lane) were then separated by 8% SDS-PAGE, transferred onto nitrocellulose membrane (Bio-Rad), and blocked with 5% non-fat milk (Bio-Rad) in TBS (Bio-Rad) containing 0.1% Tween 20 (Sigma–Aldrich). Antibodies used for Western blotting, immunostaining, and/or ChIP were summarized in Supplementary Tables 5 and 6. The signals were developed using an ECL Western blotting substrate kit (Bio-Rad) and detected using a Chemidoc XRS+ (Bio-Rad) with Image Lab software.
Immunohistochemistry and immunofluorescence analysis
Paraffin-embedded tissues were sectioned to 5 µm thickness using a microtome. Uterine sections were deparaffinized and rehydrated. Endogenous peroxidase was inactivated with 3% H2O2. Sections were subjected to antigen retrieval in 0.01 M sodium citrate buffer (pH 6.0). Nonspecific staining was blocked using protein block serum (Dako, Carpinteria, CA, USA) for 1 h. Sections were incubated with primary antibodies at 4 °C overnight. On the following day, sections were incubated with appropriate secondary antibodies for 1 h at room temperature. Sections were counterstained using Topro-3-iodide (TOPRO; Life Technologies, Carlsbad, CA, USA) and mounted. For immunohistochemistry, DAB reagent (Vector Laboratories, Inc., Burlingame, CA, USA) was applied to visualize signals. Images were observed under a microscope (Carl Zeiss, Oberkochen, Germany) and analyzed using ZEN software (Carl Zeiss).
mRNA-seq and data analysis
Libraries were prepared from 2 μg of total RNA in Cfp1f/f and Cfp1d/d mouse uterus on Day 4 using the SMARTer stranded mRNA-Seq kit (Clontech Laboratories, Inc., USA). mRNAs were used for the cDNA synthesis and shearing, following the manufacturer’s instructions (n = 3 pools: 3 mice per each pool). The Illumina indexes were used, and the enrichment step was conducted with PCR. The RT (read count) data were processed based on the quantile normalization method using EdgeR within R70 utilizing bioconductor. The alignment files were also used for assembling transcripts, estimating their abundances, and detecting differential expression of genes or isoforms using cufflinks. These are performed using Bowtie2 software. We also used the fragments per kilobase of exon per million (FPKM) fragments to determine the expression levels of the gene regions. Gene classification was based on searches made by GSEA, and heatmaps of the unsupervised hierarchical clustering and DEGs were produced using MeV software.
CFP1 immunoprecipitation
Immunoprecipitation of CFP1 protein using CFP1 antibodies was performed with the manufacturer’s instructions using immunoprecipitation kit (Abcam). In addition, CFP1 Western blotting following immunoprecipitation was performed to evaluate the specificity of CFP1 antibodies used for CFP1 ChIP-seq for Cfp1f/f uterine samples (Supplementary Fig. 10). On Day 4, uterine horns of Cfp1f/f and Cfp1d/d mice were cut vertically, and epithelial and stromal cells were separated from the smooth muscles (n = 4 to 5 per each group). Then, tissues were incubated in a lysis buffer with protease inhibitors and mixed on a rocker at 4 °C for 1 h. The tissue extracts were transferred to a fresh tube, and a predetermined amount of antibodies was added on a rocker at 4 °C for 4 h. After antibody binding, protein A/G Sepharose beads were added on a rocker at 4 °C for 1 h. Next, protein A/G Sepharose beads were collected, washed, and eluted. The protein of interest was purified by low-speed centrifugation at 4 °C and used for Western blotting.
ChIP and real-time ChIP PCR
ChIP analysis was performed with a slight modification of the manufacturer’s instructions using the ChIP-IT Express Enzymatic kit (Active Motif, Carlsbad, CA, USA). On Day 4, Cfp1f/f and Cfp1d/d mouse uterus horns were cut, and epithelial and stromal cells were separated from the uterine smooth muscles (n = 3 pools: 3 mice per each pool). Then, epithelial and stromal cells were fixed in DMEM high-glucose media containing 1% formaldehyde and then made into single cell slurry through an electric homogenizer. DNA fragments between 150 and 500 bp were obtained by enzymatic shearing cocktail after lysis cell and nuclei isolation through lysis buffer and Dounce homogenizer. Antibodies were added in sheared chromatin, and immunoprecipitation was performed overnight at 4 °C. Immunoprecipitated DNA was utilized for real-time ChIP PCR with specific primers (Supplementary Table 1). To quantify the enrichment level in ChIP-seq data, real-time ChIP PCR was performed with iQTM SYBR Green Supermix (Bio-Rad) on BIO-RAD iCycler using immunoprecipitated DNA. All PCR reactions (10% input, CFP1, SETD1, H3K4me3, and normal IgG) were duplicated (n = 3 to 4 per each group).
ChIP-seq and data analysis
The library was constructed using NEBNext® UltraTM DNA Library Prep Kit for Illumina (New England Biolabs, UK) according to the manufacturer’s instructions. Briefly, the chopped DNA was ligated with adaptors. After purification, the PCR reaction was conducted with adaptor-ligated DNA and index primer for multiplexing sequencing. The library was purified using magnetic beads to remove all reaction components. The library size was assessed by Agilent 2100 bioanalyzer (Agilent Technologies, Amstelveen, Netherlands). High-throughput sequencing was performed as paired-end 100 sequencing using HigSeq 2500 (Illumina, Inc. USA). ChIP-seq metaplots and heatmaps were analyzed using DeepTools software.
Experimentally induced mouse model of endometriosis
Recipient (wild type) and donor (Cfp1f/f and Cfp1d/d) female mice (8–10 weeks of age) were primed with 5 IU PMSG (Sigma–Aldrich) for 48 h to stimulate uterine growth. After inhalation anesthesia, the uterine horn was collected from donor mice and vertically opened with scissors in a petri dish containing warmed Dulbecco’s phosphate-buffered saline (37 °C). The uterine horn was cut into small pieces of approximately 1 mm2 and injected into the peritoneum of the OVX recipient mice. After transplantation of uterine tissues in recipient mice, E2 (100 ng/mouse), P4 (2 mg/mouse), PBS, and/or SAG were injected every 3 days. At 15 days after transplantation, the volume and number of ectopic lesions were measured, and ectopic lesions were collected for real-time RT-PCR.
Human endometrial sampling
Control (n = 7) and endometriotic endometrial tissues (n = 5) were collected from patients who underwent hysteroscopy–laparoscopy surgery to evaluate endometrial abnormalities, including endometriosis in the Department of Obstetrics and Gynecology of the First Affiliated Hospital of Xiamen University. The sample collection and studies reported here were approved by the ethics committee of Hospital of Xiamen University (XMYY-2021KYSB044), and all participants signed the informed consent. Endometriotic lesions were obtained from women (aged 25–35 years) suffering from ovarian endometriosis, confirmed by laparoscopy and histopathology. These patients had regular menstrual cycles and were recruited without hormone treatment for at least 3 months before surgery. All samples were collected during the luteal phase of the menstrual cycle.
Statistical analysis
GraphPad Prism version 8 software (GraphPad Software, La Jolla, CA, USA) was used for statistical analyses. All values are represented as mean ± standard deviation. Statistical analyses were performed using the unpaired Student’s t-tests, and p < 0.05 was considered statistically significant.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
Raw data files are deposited in the NCBI Gene Expression Omnibus database under Super Series accession number GSE219104. The Supplementary Fig. 7 data re-analyzed in this study are available in the GSE118264 database13, GSE40661 database34, and GSE178541 database35. All other data supporting the findings of this study are available within the paper and its Supplementary Information. Source data are provided with this paper.
References
Voo, K. S., Carlone, D. L., Jacobsen, B. M., Flodin, A. & Skalnik, D. G. Cloning of a mammalian transcriptional activator that binds unmethylated CpG motifs and shares a CXXC domain with DNA methyltransferase, human trithorax, and methyl-CpG binding domain protein 1. Mol. Cell. Biol. 20, 2108–2121 (2000).
Yang, Y., Yang, Y., Chan, K. & Couture, J. F. Analyzing the impact of CFP1 mutational landscape on epigenetic signaling. FASEB J. 35, e21790 (2021).
Thomson, J. P. et al. CpG islands influence chromatin structure via the CpG-binding protein Cfp1. Nature 464, 1082–1086 (2010).
Carlone, D. L. et al. Reduced genomic cytosine methylation and defective cellular differentiation in embryonic stem cells lacking CpG binding protein. Mol. Cell. Biol. 25, 4881–4891 (2005).
Bledau, A. S. et al. The H3K4 methyltransferase Setd1a is first required at the epiblast stage, whereas Setd1b becomes essential after gastrulation. Development 141, 1022–1035 (2014).
Clouaire, T., Webb, S. & Bird, A. Cfp1 is required for gene expression-dependent H3K4 trimethylation and H3K9 acetylation in embryonic stem cells. Genome Biol. 15, 451 (2014).
Lin, F. et al. Epigenetic initiation of the TH17 differentiation program is promoted by Cxxc finger protein 1. Sci. Adv. 5, eaax1608 (2019).
Cao, W. et al. CXXC finger protein 1 is critical for T-cell intrathymic development through regulating H3K4 trimethylation. Nat. Commun. 7, 11687 (2016).
Sha, Q. Q. et al. CFP1 coordinates histone H3 lysine-4 trimethylation and meiotic cell cycle progression in mouse oocytes. Nat. Commun. 9, 3477 (2018).
Yu, C. et al. CFP1 regulates histone H3K4 trimethylation and developmental potential in mouse oocytes. Cell Rep. 20, 1161–1172 (2017).
Jiang, Y. et al. CXXC finger protein 1-mediated histone H3 lysine-4 trimethylation is essential for proper meiotic crossover formation in mice. Development 147, dev183764 (2020).
Brown, S. B. & Hankinson, S. E. Endogenous estrogens and the risk of breast, endometrial, and ovarian cancers. Steroids 99, 8–10 (2015).
Wang, X. et al. SOX17 regulates uterine epithelial-stromal cross-talk acting via a distal enhancer upstream of Ihh. Nat. Commun. 9, 4421 (2018).
Kong, S. et al. Epigenetic control of embryo-uterine crosstalk at peri-implantation. Cell Mol. Life Sci. 76, 4813–4828 (2019).
Retis-Resendiz, A. M. et al. The role of epigenetic mechanisms in the regulation of gene expression in the cyclical endometrium. Clin. Epigenet. 13, 116 (2021).
Bunkar, N., Pathak, N., Lohiya, N. K. & Mishra, P. K. Epigenetics: a key paradigm in reproductive health. Clin. Exp. Reprod. Med. 43, 59–81 (2016).
Grimaldi, G. et al. Down-regulation of the histone methyltransferase EZH2 contributes to the epigenetic programming of decidualizing human endometrial stromal cells. Mol. Endocrinol. 25, 1892–1903 (2011).
Tamura, I. et al. Genome-wide analysis of histone modifications in human endometrial stromal cells. Mol. Endocrinol. 28, 1656–1669 (2014).
Nancy, P. et al. H3K27me3 dynamics dictate evolving uterine states in pregnancy and parturition. J. Clin. Invest. 128, 233–247 (2018).
Monteiro, J. B. et al. Endometriosis is characterized by a distinct pattern of histone 3 and histone 4 lysine modifications. Reprod. Sci. 21, 305–318 (2014).
Zondervan, K. T., Becker, C. M. & Missmer, S. A. Endometriosis. N. Engl. J. Med. 382, 1244–1256 (2020).
Vannuccini, S., Clemenza, S., Rossi, M. & Petraglia, F. Hormonal treatments for endometriosis: the endocrine background. Rev. Endocr. Metab. Disord. 23, 333–355 (2022).
Saunders, P. T. K. & Horne, A. W. Endometriosis: etiology, pathobiology, and therapeutic prospects. Cell 184, 2807–2824 (2021).
Lee, D., Kim, S. K., Lee, J. R. & Jee, B. C. Management of endometriosis-related infertility: Considerations and treatment options. Clin. Exp. Reprod. Med. 47, 1–11 (2020).
Kim, T. H. et al. Loss of HDAC3 results in nonreceptive endometrium and female infertility. Sci. Transl. Med. 11, eaaf7533 (2019).
Li, Y. et al. Progesterone alleviates endometriosis via inhibition of uterine cell proliferation, inflammation and angiogenesis in an immunocompetent mouse model. PLoS ONE 11, e0165347 (2016).
Dizerega, G. S., Barber, D. L. & Hodgen, G. D. Endometriosis: role of ovarian steroids in initiation, maintenance, and suppression. Fertil. Steril. 33, 649–653 (1980).
Kitawaki, J. et al. Endometriosis: the pathophysiology as an estrogen-dependent disease. J. Steroid Biochem. Mol. Biol. 83, 149–155 (2002).
Marquardt, R. M., Kim, T. H., Shin, J. H. & Jeong, J. W. Progesterone and estrogen signaling in the endometrium: what goes wrong in endometriosis? Int. J. Mol. Sci. 20, 3822 (2019).
Liu, K. & Min, J. Structural basis for the recognition of non-methylated DNA by the CXXC domain. J. Mol. Biol. 432, 1674–1686 (2020).
Xu, C. et al. DNA sequence recognition of human CXXC domains and their structural determinants. Structure 26, 85–95.e83 (2018).
Xu, C., Bian, C., Lam, R., Dong, A. & Min, J. The structural basis for selective binding of non-methylated CpG islands by the CFP1 CXXC domain. Nat. Commun. 2, 227 (2011).
Lee, K. et al. Indian hedgehog is a major mediator of progesterone signaling in the mouse uterus. Nat. Genet. 38, 1204–1209 (2006).
Rubel, C. A. et al. A Gata2-dependent transcription network regulates uterine progesterone responsiveness and endometrial function. Cell Rep. 17, 1414–1425 (2016).
Li, R. et al. The role of epithelial progesterone receptor isoforms in embryo implantation. iScience 24, 103487 (2021).
Prentice, A. et al. Ovarian steroid receptor expression in endometriosis and in two potential parent epithelia: endometrium and peritoneal mesothelium. Hum. Reprod. 7, 1318–1325 (1992).
McKinnon, B., Mueller, M. & Montgomery, G. Progesterone resistance in endometriosis: an acquired property? Trends Endocrinol. Metab. 29, 535–548 (2018).
Patel, B. G., Rudnicki, M., Yu, J., Shu, Y. & Taylor, R. N. Progesterone resistance in endometriosis: origins, consequences and interventions. Acta Obstet. Gynecol. Scand. 96, 623–632 (2017).
Szukiewicz, D. et al. Estrogen- and progesterone (P4)-mediated epigenetic modifications of endometrial stromal cells (EnSCs) and/or mesenchymal stem/stromal cells (MSCs) in the etiopathogenesis of endometriosis. Stem Cell Rev. Rep. 17, 1174–1193 (2021).
Tamaresis, J. S. et al. Molecular classification of endometriosis and disease stage using high-dimensional genomic data. Endocrinology 155, 4986–4999 (2014).
Liu, M. et al. Menin directs regionalized decidual transformation through epigenetically setting PTX3 to balance FGF and BMP signaling. Nat. Commun. 13, 1006 (2022).
Shi, D. et al. Celsr1 is required for the generation of polarity at multiple levels of the mouse oviduct. Development 141, 4558–4568 (2014).
Wang, H. et al. Aberrant cannabinoid signaling impairs oviductal transport of embryos. Nat. Med. 10, 1074–1080 (2004).
Li, S. & Winuthayanon, W. Oviduct: roles in fertilization and early embryo development. J. Endocrinol. 232, R1–R26 (2017).
Shchuka, V. M. et al. The pregnant myometrium is epigenetically activated at contractility-driving gene loci prior to the onset of labor in mice. PLoS Biol. 18, e3000710 (2020).
Chun, K. T. et al. The epigenetic regulator CXXC finger protein 1 is essential for murine hematopoiesis. PLoS ONE 9, e113745 (2014).
Hui, Z. et al. Cxxc finger protein 1 positively regulates GM-CSF-derived macrophage phagocytosis through Csf2ralpha-mediated signaling. Front. Immunol. 9, 1885 (2018).
Khan, K. N. et al. Estrogen and progesterone receptor expression in macrophages and regulation of hepatocyte growth factor by ovarian steroids in women with endometriosis. Hum. Reprod. 20, 2004–2013 (2005).
Butts, C. L. et al. Inhibitory effects of progesterone differ in dendritic cells from female and male rodents. Gend. Med. 5, 434–447 (2008).
Hughes, G. C., Clark, E. A. & Wong, A. H. The intracellular progesterone receptor regulates CD4+ T cells and T cell-dependent antibody responses. J. Leukoc. Biol. 93, 369–375 (2013).
Lee, J. Y., Lee, M. & Lee, S. K. Role of endometrial immune cells in implantation. Clin. Exp. Reprod. Med. 38, 119–125 (2011).
Sha, Q. Q. et al. CFP1-dependent histone H3K4 trimethylation in murine oocytes facilitates ovarian follicle recruitment and ovulation in a cell-nonautonomous manner. Cell Mol. Life Sci. 77, 2997–3012 (2020).
Brici, D. et al. Setd1b, encoding a histone 3 lysine 4 methyltransferase, is a maternal effect gene required for the oogenic gene expression program. Development 144, 2606–2617 (2017).
Li, Q. et al. The antiproliferative action of progesterone in uterine epithelium is mediated by Hand2. Science 331, 912–916 (2011).
Al-Sabbagh, M., Lam, E. W. & Brosens, J. J. Mechanisms of endometrial progesterone resistance. Mol. Cell Endocrinol. 358, 208–215 (2012).
Zanatta, A. et al. The relationship among HOXA10, estrogen receptor alpha, progesterone receptor, and progesterone receptor B proteins in rectosigmoid endometriosis: a tissue microarray study. Reprod. Sci. 22, 31–37 (2015).
Zubrzycka, A., Zubrzycki, M., Perdas, E. & Zubrzycka, M. Genetic, epigenetic, and steroidogenic modulation mechanisms in endometriosis. J. Clin. Med. 9, 1309 (2020).
Lessey, B. A. & Kim, J. J. Endometrial receptivity in the eutopic endometrium of women with endometriosis: it is affected, and let me show you why. Fertil. Steril. 108, 19–27 (2017).
Smith, Z. D. & Meissner, A. DNA methylation: roles in mammalian development. Nat. Rev. Genet. 14, 204–220 (2013).
Butler, J. S., Lee, J. H. & Skalnik, D. G. CFP1 interacts with DNMT1 independently of association with the Setd1 Histone H3K4 methyltransferase complexes. DNA Cell Biol. 27, 533–543 (2008).
Gao, F. et al. Epigenetic changes through DNA methylation contribute to uterine stromal cell decidualization. Endocrinology 153, 6078–6090 (2012).
Pokhrel, B., Chen, Y. & Biro, J. J. CFP-1 interacts with HDAC1/2 complexes in C. elegans development. FEBS J. 286, 2490–2504 (2019).
Xiong, Y. et al. Effects of high progesterone level on the day of human chorionic gonadotrophin administration in in vitro fertilization cycles on epigenetic modification of endometrium in the peri-implantation period. Fertil. Steril. 108, 269–276.e261 (2017).
Colon-Caraballo, M., Monteiro, J. B. & Flores, I. H3K27me3 is an epigenetic mark of relevance in endometriosis. Reprod. Sci. 22, 1134–1142 (2015).
Wen, X. et al. Decreased mixed lineage leukemia 1 is involved in endometriosis-related infertility. J. Mol. Endocrinol. 66, 45–57 (2021).
Soyal, S. M. et al. Cre-mediated recombination in cell lineages that express the progesterone receptor. Genesis 41, 58–66 (2005).
Zhang, S. et al. Uterine Rbpj is required for embryonic-uterine orientation and decidual remodeling via Notch pathway-independent and -dependent mechanisms. Cell Res. 24, 925–942 (2014).
Park, M. et al. Perivascular stem cell-derived cyclophilin A improves uterine environment with Asherman’s Syndrome via HIF1alpha-dependent angiogenesis. Mol. Ther. 28, 1818–1832 (2020).
Kim, H. R. et al. Estrogen induces EGR1 to fine-tune its actions on uterine epithelium by controlling PR signaling for successful embryo implantation. FASEB J. 32, 1184–1195 (2018).
Nguefack-Tsague, G., Zucchini, W. & Fotso, S. Frequentist model averaging and applications to bernoulli trials. Open J. Stat. 6, 545 (2016).
Acknowledgements
This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (NRF- 2019R1A6A1A03032888 to H.S.), by the NRF grant funded by the Korea government (the Ministry of Science and ICT) (NRF-2015R1A2A2A01006714 to H.S., NRF- 2020R1C1C1008317 to H.R.K., NRF-2020R1A2C2005012 to H.S., and NRF-2020R1A6A3A01100338 to M.P.), by Bio & Medical Technology Development Program of the NRF and funded by the Korean government (MSIT) (NRF-2022M3A9E4016936 to S.-H.H.).
Author information
Authors and Affiliations
Contributions
S.C.Y. and H.S. conceived and designed the experiments. S.C.Y., M.P., H.L., P.W., C.P., G.L., Q.C., S.H.P., and Y.S.K. performed experiments. S.C.Y., M.P., K.-H.H., Y.C., F.J.D., J.P.L., D.G.S., H.J.L., S.-H.H., H.-R.K. and H.S. analyzed the results. S.C.Y., M.P., and H.S. discussed the results and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yang, S.C., Park, M., Hong, KH. et al. CFP1 governs uterine epigenetic landscapes to intervene in progesterone responses for uterine physiology and suppression of endometriosis. Nat Commun 14, 3220 (2023). https://doi.org/10.1038/s41467-023-39008-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-023-39008-0
- Springer Nature Limited