Abstract
Hepatocellular carcinoma (HCC), one of the most malignant tumors, is characterized by its stubborn immunosuppressive microenvironment. As one of the main members of the tumor microenvironment (TME) of HCC, tumor-associated macrophages (TAMs) play a critical role in its occurrence and development, including stimulating angiogenesis, enhancing immunosuppression, and promoting the drug resistance and cancer metastasis. This review describes the origin as well as phenotypic heterogeneity of TAMs and their potential effects on the occurrence and development of HCC and also discusses about various adjuvant therapy based strategies that can be used for targeting TAMs. In addition, we have highlighted different treatment modalities for TAMs based on immunotherapy, including small molecular inhibitors, immune checkpoint inhibitors, antibodies, tumor vaccines, adoptive cellular immunotherapy, and nanocarriers for drug delivery, to explore novel combination therapies and provide feasible therapeutic options for clinically improving the prognosis and quality of life of HCC patients.
Similar content being viewed by others
Facts
-
Tumor-associated macrophages are one of the most abundant immune cells present in the tumor microenvironment of hepatocellular carcinoma, and have been implicated in both occurrence and development of hepatocellular carcinoma.
-
Targeting tumor-associated macrophages can serve as an important strategy for the clinical management of hepatocellular carcinoma in the future, and macrophage-based immunotherapy can gradually be incorporated into the clinical regimen.
-
Growing evidence suggest favorable prognosis and promising trends of tumor-associated macrophages as therapeutic targets.
Open questions
-
Where do the macrophages originate from in liver cancer?
-
What are the therapeutic strategies targeting tumor-associated macrophages?
-
What are the immunotherapeutic options based on macrophages?
Introduction
According to the global cancer data for 2020 recently released by the WHO, liver cancer ranks seventh in morbidity and third in mortality among different cancers [1]. Hepatocellular carcinoma (HCC), the most common type of liver cancer, accounting for ~80% of primary liver cancers, and remains one of the most common as well as leading causes of mortality worldwide [2, 3]. The intractable tumor microenvironment (TME) plays an vital role in the development and progression of HCC and constitutes one of the three key unsolved issues (cancer recurrence, fatal metastasis, and the refractory tumor microenvironment) that can obstruct effective cancer management in the clinical practice [4, 5]. The tumor microenvironment (TME) characterizes all the noncancerous components near the tumor cells, including fibroblasts, myelogenic suppressor cells (MDSCs), macrophages, lymphocytes, the extracellular matrix (ECM), and interwoven blood vessels generated by endothelial cells and pericytes, thereby creating a protective niche in which the tumor cells are immune to routine intervention, leading to the treatment failure [6]. Although the ‘seed and soil’ theory was initially proposed in 1889, only in recent years increasing attention have been paid to the role of the TME in promoting tumor development [6]. Over the past decade, cognitive expansion about how the TME can potentially interact with the tumor cells has led to exploration of a new tumor treatment model: targeting the tumor stromal cells. However, the design of novel therapies that can target the tumor stroma of HCC relies primarily on exhaustive comprehension of the mutual effects between the TME and HCC cells.
The TME is a complex multicellular system characterized by tumor cell-stromal cell-extracellular matrix interactions [6]. Tumor-associated macrophages (TAMs), the number of which in the TME accounts for 20% to 40% among all HCC patrolling and infiltrating lymphocytes and even more in some rare HCC subtypes, have been reported to be enriched in the TME of HCC and can coordinate with the tumor-associated inflammation [6,7,8,9,10,11,12,13]. In addition, tumor-associated macrophages (TAMs), which infiltrate most of the solid tumors in abundance, can contribute to tumor progression by providing a major barrier against antitumor immunity and by stimulating proliferation, angiogenesis, and metastasis [14]. Moreover, in terms of mechanism, TAMs can establish and reshape the structure of the extracellular matrix such that the tumor cells are able to invade the TME and interact with other tumor or stromal cells by secreting various cytokines such as interleukin, interferon, tumor necrosis factor superfamily, colony-stimulating factors, chemokines, and growth factors [15, 16]. It has been established that TAMs cannot not be regarded as a homogenous cell population, and these cells can either promote or inhibit tumors in different systems or even exhibit both the functions [16, 17]. In-depth molecular studies of TAMs in the human malignant tumors have expanded our understanding of their source complexity, phenotypic heterogeneity, and functional diversity. In principle, identification of the specific oncogenic TAM subtypes can pave the way for the development of novel as well as optimal TAM-targeted anticancer immunotherapy.
In this review, we have described the origin and heterogeneity of TAMs and discussed the role of TAMs in regulating the initiation, progression, metastasis, and drug resistance of HCC. We have also described in detail existing therapeutic strategies and feasible immunotherapy options for targeting TAMs in HCC to accelerate the leap from laboratory research to rapid clinical application.
Biology of TAMs in HCC
Origin of TAMs
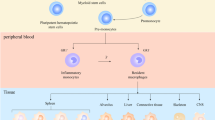
The source of TAMs is heterogeneous (Fig. 1). Tissue-resident macrophages (so-called Kupffer cells) in the liver and blood-recruited monocyte-derived macrophages have been reported to be involved in the formation of the TME [18, 19]. However, it has been hypothesized that TAMs are derived primarily from the circulating blood monocytes [20]. In the mice, TAMs are mainly derived from bone marrow monocytes (TAMs in human HCC arise from CCR2+ monocytes) that can recruited by inflammatory signals released by cancer cells in both the primary and metastatic tumors, and they can differentiate into TAMs under the action of chemokines and growth factors produced by the stromal cells and tumor cells, thus promoting tumor progression [20,21,22,23]. For example, an increase in TAMs induced by lysyl oxidase-like 4 (LOXL4: a copper-dependent monoamine oxidase in the extracellular matrix) in mice has been found mainly due to monocyte infiltration; LOXL4 can inhibit the proliferation of resident macrophages in the liver and nearly deplete the resident macrophages during the formation of HCC [24]. In addition, recent studies have indicated that tumor-dependent recruitment of monocyte-derived macrophages occurs in chronically damaged liver tissues compared with the tumors growing in healthy livers [25]. Furthermore, self-replication can serve as an important mechanism for facilitating accumulation of tumor-infiltrating macrophages in HCC tissues [26]. It is worth noting that evidence also suggest that macrophages in the liver tissues can be established by progenitor cells from the yolk sac and fetal liver and maintained by the self-proliferation and monocyte input [27,28,29]. For instance, Yu-Chen Ye et al. found that the dominating TAMs in orthotopic HCC in conditional disruption of the recombination signal binding protein Jκ (RBPj cKO) mice manifested properties of Kupffer cells (KCs), thus suggesting that TAMs (KC-like TAMs, F4/80+CD11bloLy6clo/− TAMs) in HCC in situ were more likely to originate from embryonic hematopoiesis–generated KCs and bone marrow (BM) monocyte-derived KCs, which are considered as the self-renewing tissue-resident macrophages [30,31,32]. However, it is not clear whether these KC-like TAMs can originate from true KCs, mononuclear cells derived from bone marrow or extramedullary origin, or even monocyte-derived TAMs (moTAMs). KC-like TAMs differentiate from true KCs or bone marrow-derived or extramedullary monocytes. However, KCs might account for only a small part of the total TAM pool of HCC [30]. These observations reinforce that the definition of TAMs in HCC should not only be used to identify bone marrow-derived macrophages that can infiltrate the tumors but should also be extended to all the macrophages that play a vital role in the TME.
YFP+ AA4.1+ Kit+ CD45lo EMPs (erythro-myeloid progenitor), which appeared initially in the yolk sac of a mouse embryo at E(day)8.5, have been identified as macrophage progenitor cells [135]. EMPs, which can exist as Kit+ progenitor cells can effectively migrate and proliferate in the fetal liver until E16.5 and produce the fetal liver monocytes (appearing at E12.5) until the late stage of fetal development [135]. Moreover, EMPs have been found to rapidly differentiate into premacrophages (pMacs), which can simultaneously colonize the whole embryo from E9.5 in a CX3CR1-dependent manner and differentiated into macrophages [136]. It has been reported that starting from E10.5, almost immediately after embryonic tissue colonization, YFP+F4/80bright fetal macrophages can appear in the fetal liver [135, 136]. Thereafter, around E14.5, fetal liver monocytes can replace early F4/80hi macrophages and constitute most of the liver-resident macrophages in the adult liver, that is, Kupffer cells (KCs) [135, 137]. During the postnatal bone formation, the hematopoietic function of the fetal liver decreases significantly and is replaced by BM [135, 138, 139]. BM-derived mononuclear phagocyte precursor MDPs (macrophage/dendritic cell progenitor cells) which can produce inflammatory monocytes (Ly6c+), giving rise to the patrol monocytes (Ly6c−) [138, 139]. Interestingly, both inflammatory monocytes (Ly6c+) and patrol monocytes (Ly6c-) can be recruited into the liver capsule and then differentiate into CD207-EGFPhi F4/80+ liver capsular macrophages (LCMs) [140, 141]. Inflammatory monocytes (Ly6c+) enter the liver and participate in the formation of the resident macrophages in a very small proportion of the tissues, namely, monocyte-derived liver macrophages (MoMFs) [31, 135, 139]. Both KCs and blood-recruited MoMFs have been found to be involved in the formation of the tumor-associated macrophages, but it is not clear whether LCMs are also involved [18, 19]. LCMs have been also rarely considered as a predecessor of TAMs because of their special anatomical location [140]. In addition, MDSCs can differentiate into immunosuppressive TAMs in the microenvironment of HCC in the presence of HIF-1α or hypoxia [142, 143]. Collectively, the precise origins of TAMs have not been fully elucidated.
TAMs can originate from the different sources and play diverse protumoral or sometimes antitumoral roles. Interestingly, each subpopulation has a characteristic transcriptional landscape and marker profile based on the type, stage, and immune composition of the tumors they can infiltrate [33]. Lineage tracing [34] is often used to track the origin of the tumor cells, and this technique has also been employed to identify the cellular origin of TAMs. Thus, a fascinating suggestion that monitoring the fate of TAMs during the evolution of HCC can lead to a more thorough understanding of the complex nature of the TME. Single-cell RNA sequencing technology [35, 36] can be used to reveal the TAM type at the single-cell level, and spatial transcriptomics [37] can aid to visualize the different proteins or expressed genes, the combination of which can provide a more theoretical basis to support the application for targeted therapy.
Phenotypic heterogeneity of TAMs
Macrophages have long been considered to have two different activation states: Macrophages with inflammatory functions are called M1 macrophages, and macrophages with anti-inflammatory functions are termed as M2 macrophages [38, 39]. Most TAMs exhibit M2 polarization (cell morphology tends to be more fusiform [40, 41]) and can promote progression of HCC by secreting the various protumor and angiogenic factors and inhibiting activation of tumor-infiltrating T cells. In addition, along with the transition from the M1 to the M2 state, macrophages acquire features that can effectively promote tumor invasion, metastasis, and immunosuppression with upregulated expression of genes such as MMP14 (matrix metalloproteinase 14), VEGFA (vascular endothelial growth factor A), and MRC1/CD206 (mannose receptor) [13]. However, macrophages can gradually obtain the characteristics of the M2 phenotype, but the M1 signature is not obviously reduced [13]. It has been reported that M2-like macrophages can maintain some antitumor properties, which supports the view that macrophage activation in the TME of HCC does not follow the classical polarization pattern. In the similar fashion, a study suggested that the two macrophage subsets (Mø_c2 and Mø_c3) are maintained in the transition state mentioned which can facilitate the transition from M1 polarization to M2 polarization of macrophages is gradual in the progression of HCC. The understanding of TAMs could be potentially extended to all macrophage subsets in the TME because each kind of macrophage inevitably can effectively exchange materials and information with the tumor cells, which is closely related to tumor progression, either promoting or inhibiting tumor progression.
The phenotypic heterogeneity of TAMs is primarily manifested by a wide range of biological markers with selective expression patterns in the context of a specific TME [42]. In mice, TAMs are identified in the tumors as F4/80+ and CD11b+. In humans, TAMs are identified as CD68+ by immunohistochemistry and CD14+ by flow cytometry. There are several other markers used to define HCC TAMs [43]. M1-phenotype TAMs polarized from the peripheral blood mononuclear cells (PBMCs) are marked by relatively higher expression of IL-1β, TNF-α, IL-6, IL-12, HLA-DR(HLA-DRα), CCR7, Type I IFNγ, CXCL1–3, CXCL-5, CXCL8–10, CCL10, inducible nitric oxide synthase (iNOS or NOS2), MHC II, CD11c, CD80, CD86 and CD16/32 in HCC [44,45,46,47,48,49,50,51,52,53,54,55]. Conversely, M2-phenotype TAMs polarized from PBMCs are marked by higher expression of CD209 (DC-SIGN), CD206 (MRC1), CD204 (MSR1/SR-A), CD163, CD115, IL4, IL10, Fizz1, p-STAT3 and Arg1 [41, 56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75] (Table 1). Moreover, similar to cancer-associated fibroblasts (CAFs) and T lymphocytes, TAMs comprise a group of distinct cell subsets that can respond differently to different interstitial stimuli, exhibit unique secretory phenotypes, and play specific biological roles in the TME. Thus, mastering reliable and specific cell surface markers is key to distinguishing TAM subsets. In addition, contradictory results obtained suggest that the pleiotropic function of TAMs can be attributed to the high heterogeneity of TAMs, and hence it is necessary to comprehensively describe the cell origin, surface markers, activation stages, and spatial distributions of TAMs to distinguish which TAM cell types are being studied in each experiment.
Etiological background of TAM in HCC
The etiological background of TAM in HCC is extremely complicated. It has been reported that the viral infection, long-term drinking and fat accumulation can cause liver damage, liver injury promotes the recruitment of Ly6Chi macrophages from the bone marrow, thus substantially increasing the number macrophages to the already-large number of liver-resident macrophages [76]. Moreover, liver-resident macrophages can act as promoters of inflammation and fibrosis in diseases such as viral hepatitis, alcoholic liver disease and non-alcoholic steatohepatitis. Interestingly, CCL2 can induce the recruitment of circulating CCR2+Ly6C+ monocytes into the liver [70]. Recruited monocytes are more pro-inflammatory than the resident Kupffer cells and can produce the various pro-inflammatory cytokines, such as TNFα and IL-1β that contribute to the development of NASH and fibrosis [70]. In the case of long-term inflammatory stimulation and fibrosis progression, HCC gradually formed. The existence of tumor not only can destroy the original spatial structure of the tissue to some extent, but also can damage the original niche of macrophages [77]. Macrophages, as immune cells, are originally involved in the regulation of immunological behavior against tumors, but after being modulated by the tumor cells, macrophages can be transformed into TAMs to promote the occurrence and development of tumors.
Strategies for targeting TAMs
Most of the available treatment options are only effective against small-sized HCC (2–3 cm in diameter); very few systemic chemotherapies have been shown to be consistently efficacious in treating HCC [78, 79]. Current modalities for HCC treatment include transarterial chemoembolization (TACE), surgical resection, radiotherapy, chemotherapy, local radiofrequency ablation, and systemic targeted therapy [80, 81]. Tumor resection, liver transplantation, and local radiofrequency ablation are the most effective treatments for the management of early HCC, but they are not suitable for most patients with HCC, many of whom are already at an advanced stage at the time of diagnosis [81]. Although sorafenib, lenvatinib, which is noninferior to sorafenib, and regorafenib can increase survival and are used as standard treatments in advanced HCC [81], their effect is still not very effective. Hopefully, targeting TAMs in HCC as a feasible target for adjuvant therapy of HCC might lead to a better prognosis for patients with HCC [30, 41, 56, 59, 63, 75, 82,83,84,85](Table 2). What’s more, a better understanding of the recruitment and functional tilt of TAMs can provide a sound basis for the development of macrophage-centered therapeutic strategies. The negative prognostic significance of macrophage infiltration has been evaluated by traditional immunohistochemistry, molecular markers, or single cell analysis, which has promoted the clinical evaluation of macrophage targeted therapy strategy.
Based on the consensus that “TAMs can generally promote tumors”, several kinds of preclinical or clinical experiments have been proposed and applied: depletion of TAMs (inhibition of the monocyte/macrophage recruitment), regulation of TAM polarization, and inhibition of TAM tumor-promoting functions (targeting the molecules with tumor-promoting functions). Regardless of the progress made, these strategies have displayed some major limitations. For example, as CSF1R inhibitors were unable to specifically consume TAMs and generally can target monocytes/macrophages, but they might also damage the body’s immune system over time [86]. The use of CCR2 antagonists alone cannot completely deplete TAMs in HCC because the tissue-resident macrophages were compensatively replenished [30]. For example, simply repolarizing M2 TAMs into M1 TAMs might not be as good strategy as once thought because M1 TAMs can also play a tumor-promoting role by aiding HCC escape [57, 87]. Fortunately, recent research has begun to supplement these deficiencies. For example, promoting combined application of Notch signaling and suppressing CCL2-CCR2 signaling can completely deplete TAMs in HCC. The combination of TAM M1 polarization and PD-L1/PD-1 checkpoint blockade can render M1 TAMs “politically firm” and completely kill the liver cancer cells [57, 88]. Both the limitations of previous targeting strategies and the improvement of treatment strategies suggest that relevant treatment modalities targeting TAMs should not be rigidly confined to one specific level. The TME of HCC is complex, dynamic, and heterogeneous. Thus, potential interactions between several types of cells and components in the TME of HCC is also complicated, providing a huge network to aid HCC survival and form an almost indestructible barrier. Therefore, novel treatment strategies for HCC need to build an equal or even larger treatment network.
Immunotherapy
Macrophages play a key role in regulating the actions of tumor chemotherapy, radiotherapy, antiangiogenic drugs, and hormone therapy [89]. Their effects are complex and dual, acting as amplifiers or inhibitors of anti-tumor activity. Although considerable progress has been made in dissecting the yin and yang of macrophages in traditional anti-tumor therapy, still a major challenge remains to translate deeper knowledge into more effective treatment. Due to the increasingly prominent limitations of the traditional cancer treatment methods, a variety of new cancer treatment drugs based on immunotherapy have emerged in recent years, including small molecular inhibitors, immune checkpoint inhibitors, antibodies, tumor vaccines, adoptive cellular immunotherapy, and nanocarriers for drug delivery. Although most of these drugs do not directly or initially target macrophages, but actions of macrophages contribute to the final treatment outcome (Fig. 2).
Thus combination of any two or more approaches between small molecular drugs, immune checkpoint inhibitors, antibodies, tumor vaccines, adoptive cellular immunotherapy, nanocarriers for drug delivery (including but not limited to) might be able to achieve effective tumor inhibition or even tumor elimination.
Small molecule drugs
Several small molecule drugs with the advantages of oral bioavailability, relatively low cost, ease of crossing physiological barriers, and entering intracellular targets have been used to target TAMs to achieve significant tumor inhibition [90]. For example, as a small molecule inhibitor, PLX3397, which is a highly specific competitive inhibitor of a CSF-1R tyrosine kinase, can change the polarization of TAMs from M2 to M1 [41]. RIPK3 (receptor-interacting protein kinase-3) deficiency in TAMs of HCC increased fatty acid oxidation (FAO) via the ROS-caspase1-PPAR (peroxisome proliferator-activated receptor) pathway, thus playing an essential role in accumulation and M2 polarization of TAMs in the TME and accelerating HCC growth [56]. Etomoxir (CPT1a inhibitor) and GW9662 (PPAR inhibitor), which are both small molecule drugs can effectively reverse accumulation of TAMs in HCC tissues, which was found to dramatically decrease Arg1 and increases iNOS in RIPK3 KO TAMs, and ablation of FAO can switch RIPK3 KO TAM polarization from M2 to M1 [56]. In addition, there is enormous potential for small molecule inhibitors to be used in combination with other tumor treatment strategies. A CCR5 antagonist (maraviroc [MVC]) was able to convert the phenotype of macrophages cocultured with irradiated liver cancer cells to M1, thereby enhancing their radiosensitivity and apoptosis [91].
Immune checkpoint inhibitors
A number of immune checkpoint blockade therapies have been gradually identified, but the most commonly used clinical treatments are anti-PD-1 and anti-PD-L1 therapy. Overall, tumor immunotherapy based on immunosuppressive checkpoints can significantly enhance the immune response while relieving immunosuppression. Blocking the PD-1/PD-L1 pathway with pharmacological inhibitors can enhance T-cell activity and cytotoxicity, which can significantly inhibit the tumor growth of HCC [92], but the therapeutic effect is still not optimistic. Both PD-L1 and PD-1 are expressed in TAMs [93, 94]. It is worth indicating that expression of PD-L1 on M1-like TAMs can lead to immune escape of HCC [87]; thus, the combination of PD-L1/PD-1 checkpoint blockade and M1 macrophage polarization therapy appears to be a promising and effective treatment strategy [57]. Kohei Shigeta et al. treated mice with established HCC (5–6 mm in diameter) with the anti-VEGFR-2 antibody DC101 at two different doses (AA-low, 10 mg/kg and standard AA, 40 mg/kg, both thrice a week), the anti-PD-1 antibody (ICB), or their combination [88]. Anti-PD-1 immune checkpoint blockade was found to not only attenuate immunosuppression but also induced M1 macrophage polarization [88], which further enhanced the antitumor effects. In addition to PD-1/PD-L1, CD47 has been reported to act as a checkpoint associated with macrophages as a poor prognostic factor for HCC [95]. The interaction of CD47 and SIRPα on macrophages can aid the tumor cells to escape the phagocytic clearance of macrophages, though blocking CD47 can reverse macrophage-mediated tumor inhibition [96,97,98]. Of course, macrophage-related checkpoints are not limited to the above, but the mechanisms of other related checkpoints in HCC needs to be further explored and revealed. Fortunately, inhibition of these immune checkpoints has been found to significantly improve the effectiveness of cancer immunotherapy.
Antibodies
The FDA has approved a number of monoclonal antibodies for the clinical management of cancers, including rituximab (for B-cell lymphoma) [99], trastuzumab (for breast cancer) [100], and immune checkpoint inhibitors. In HCC, bavituximab, a chimeric monoclonal antibody combined with sorafenib, has been reported to significantly reduce tumor microvessel density and M2 macrophage levels and increase the tumor endothelial cell apoptosis index and M1 macrophage frequency [101]. In the early stage of monoclonal antibody therapy, both the recruitment and infiltration of substantial number of M1 TAMs was considered as a sign of good prognosis [102].
In addition, known for their small size and their ease of penetrating tissue or blocking cell/protein function by binding to epitopes, single-domain antibodies might represent useful diagnostic and/or therapeutic tools that can be used as modules for various forms of antibody-based therapeutic molecules [103]. In a recent study, a bispecific single-domain antibody (BisCCL2/5i) was used in the treatment of malignant liver tumors, which could efficiently and specifically bind and neutralize CCL2 as well as CCL5, significantly induce the polarization of TAMs to the antitumor M1 phenotype, and reduce immunosuppression in the TME [104]. In addition, the combination of bispecific single domain antibody (BisCCL2/5i) and monoclonal antibody (PD-L1 inhibitor) enabled the mice to survive for a long time in liver metastasis models of the primary liver cancer, colorectal cancer, and pancreatic cancer [104].
Tumor vaccines
As classic preventive vaccines, the hepatitis B and hepatitis C vaccines can mainly induce specific adaptive immunity before the occurrence of hepatitis by activating humoral immunity to reduce the incidence of virus-induced cancer. Although effective preventive vaccination can result in eliciting adaptive immune response, impact of immediate innate immunity on the process of spontaneous cancer regression cannot be ignored [105]. In innate immunity, dendritic cells [106, 107] and macrophages [108] can activate T cells through antigen-presenting cells to achieve the therapeutic effect of the vaccine. For example, a whole-cell GM-CSF (granulocyte-macrophage colony-stimulating factor, also called CSF-2) vaccine in combination with low-dose cyclophosphamide was reported to enhance the antigen-presenting function of the dendritic cells, neutralize the immune regulation of inhibitory Tregs (regulatory T cells), and promote activation of tumor-specific CD8 + T cells [109]. It is worth noting that GM-CSF can also induce M1 polarization of macrophages, thus enabling tumor-associated macrophages to exert their antitumor effects [110]. A Listeria-based HCC vaccine could activate the NF-κB pathway in TAMs through modulating the TLR2 (Toll-like receptor 2) and MYD88 (myeloid differentiation primary response protein 88) pathways, recruit p62 to activate autophagy pathways, skew M2-polarized TAMs to M1-polarized TAMs and promote PD-L1 expression in HCC cells but cause resensitization of the local tumor T cells to PD-1 immunotherapy [63]. It is worth noting that as a rising star of gene therapy, mRNA vaccines have been also applied in malignant liver tumors [104], but their safety and stability still need to be further explored.
Adoptive cellular immunotherapy
Adoptive therapy of immune cells, which is considered as a promising method for the treatment of cancer, can induce the tumor progression inhibition or even tumor regression by transferring the specific immune cells from the host or from other donors to the tumor-bearing host. The method has been employed for the treatment of the various malignant diseases is adoptive transfer of T cells with engineered chimeric antigen receptor (CAR-Ts) [111] or genetically modified T-cell receptor (TCR-Ts) [112]. It is possible that evolutionarily conserved natural killer T (NKT) cells might be used as adoptive cell subsets to clear TAMs and liver cancer cells [113]. Nevertheless, the progress of adoptive T-cell therapy for solid tumors is relatively slow [114], which might be limited by the fact that T cells are unable fully infiltrate solid tumors such as HCC. Interestingly, M1-polarized TAMs can increase T-cell recruitment into HCC, activate T-cell cytotoxicity, and promote T-cell proliferation [82, 115]. Moreover, M1-polarized TAMs can enhance CAR T-cell activity by producing IL-12 [116]. The first generation of chimeric antigen receptors, which combine the scFv (single chain antibody fragment) of anti-CD19, anti-mesothelin, or anti-HER2 antibodies with a CD3 intracellular domain, was designed by Klichinsky et al., with further alterations as modified macrophages with the chimeric antigen receptor (CAR-Ms) for testing. These macrophages (M1-like) showed strong tumoricidal effects in various preclinical models, and even CAR-Ms maintained their antitumor activity in the presence of human M2 macrophages [117]. Although this study was not based conducted in liver tumors, it provides a practical basis for the application of macrophage adoptive therapy in HCC, which is also a solid tumor.
Nanocarriers for the drug delivery
Due to the rapid metabolism of some drugs, excretion from the body, or non-uniform distribution in the body to weaken efficacy, drug nanocarriers (including polymer nanoparticles, liposomes, micelles, dendrimers, and inorganic nanoparticles) that can effectively target diseased areas are needed [118]. For example, MC3 LNPs (Dlin-MC3-DMA-based lipid nanoparticles) based on the liver homing deliver mRNA encoding BisCCL2/5i to the malignant liver tumors, can reduce M2 TAM infiltration and increase the proportion of M1 TAMs, and immunotherapy-related adverse events (irAEs) associated with the common complications of the systemic administration of immunotherapy are significantly decreased [104]. In the HCC model, immunosuppressive macrophages in the liver are the main types of cells that can ingest nanoparticles (NPs) [119, 120]. Thus, by reducing hypoxia-induced M2 TAM hepatocellular carcinoma infiltration, promoting macrophage polarization to M1 TAMs, increasing the number of CD8 + cytotoxic T cells in HCC, and reprogramming the immunosuppressive TME, MnO2 NPs can effectively contribute to the therapeutic strategy for PD-1/PD-L1 immune checkpoints and the effect of whole-cell tumor vaccine immunotherapy [82]. Certainly, studies based on exosomes can provide a feasible basis for constructing suitable liposomes to specifically target TAMs in HCC. For example, studies of the premetastatic niche of tumors have revealed selective uptake of exosomes by the liver resident macrophages [121, 122], thereby suggesting that it is possible to construct exosomes, liposomes, and exosome-liposome hybrid nanoparticles to target TAMs. However, nanocarriers are not used alone, but they need to be combined with other drugs to achieve regulation of TAMs in HCC to exert a substantial antitumor effects. Furthermore, as with other targeted therapies for cancer, the identification of patient populations that are likely to benefit from macrophage-targeted therapies will be essential for improving cancer treatment strategies as well as the clinical outcomes.
Future perspectives and conclusion
TAMs have emerged as an interesting candidate for innovative anti-tumor therapy, and several new treatments have been tested to reduce the population of TAMs in the tumors. However, so far, the effect is rather limited. Recently, reprogramming M2-like TAMs into immunostimulatory and anti-tumor M1-like cells has become an attractive strategy for cancer treatment, with encouraging preclinical and preliminary clinical data. For example, some clinical trials based on the solid tumors have revealed efficacy of the targeted therapy for TAMs (including NCT02829723,NCT03447314,NCT03007732,NCT02216409, etc.). However, while tumors display the characteristics of convergent evolution, there are also some uniqueness and some differences among the solid tumors, especially with respect to TAMs populations. For instance, in the lung cancer model, bone marrow-derived macrophages promote the spread of the metastatic tumors, whereas the tissue resident macrophages support the proliferation of the cancer cells at the primary tumor site [123]. In murine ovarian cancer, a population of self-renewing CD163+Tim4+TRMs in the omentum can promote the metastatic spread by generating a protecting niche for cancer stem cells [124]. In the murine glioma, TAMs are mostly derived from the resident microglia, which can promote murine glioblastoma through regulating mTOR-mediated immunosuppression [125, 126]. In the breast cancer mouse model, the number of macrophages in the tissue decreased over time, but the number of TAMs produced by the monocytes derived from bone marrow increased [127]. In this case, the ablation of tissue resident macrophages was not found to affect the growth of the tumor, but the ablation of the circulating monocytes led to the decrease of tumor size [127]. In contrast, in the mouse model of pancreatic cancer, TRMs were amplified during the tumor development and obtained a transcriptional spectrum of a typical fibrogenic program beneficial to the pancreatic cancer, which was not destroyed by the depletion of BM-derived macrophages, but was reversed by the depletion of TRMs [128]. Interestingly, in HCC model, when the tumor formed, the resident macrophages in the liver were depleted, and some of the bone marrow-derived macrophages were effectively transformed into KCs-like TAMs after entering the original liver resident macrophage niche [24, 77]. Moreover, the simple depletion of BM-derived macrophages or liver resident macrophages was not able to achieve TAMs clearance in HCC [24, 30]. The differences among TAMs function in different tumors are primarily caused by the differences of various sources, different proportions, and various locations. Therefore, future studies related to development of the targeted therapy targeting TAMs should focus on a certain kind of tumor, such as HCC.
At present, there are only few clinical trials in HCC based on targeting TAMs, and the efficacy of these modalities need to be further evaluated (Table 3), which could be affected by the therapeutic limitations caused by high heterogeneity of macrophages. In recent years, gradually mature and perfect techniques such as single cell sequencing, metabonomic and digital spatial mapping can reveal multiple targets that can be used to specifically target TAMs from the perspectives of their functional phenotype and corresponding markers, metabolic capacity, and spatial distribution. At present, clinical trials of TAMs imaging and targeted therapy based on biological agents and macrophage markers have begun. Although these studies do not target HCC, they might provide some ideas for future targeted therapy against TAMs present in HCC. For instance, a phase I/IIa clinical trial has been launched to evaluate both the tolerance and safety of Ga [68]-labeled anti-MMR(CD206) Nb(nanobody) for PET/CT scans in cancer patients with melanoma, breast cancer, and head and neck cancer (NCT04168528). In addition, in a phase II clinical trial, inoperable solid cancer patients will undergo 68GA-anti-MMR Nb imaging before and after treatment, including ICIS (NCT04758650), to evaluate the prognostic value of MMR imaging. One of the key problems that need to be solved is the specificity of the targeted markers, which should be as specific as possible for identifying TAMs. Unfortunately, although markers such as CD206 and SIRPα can be used for treatment, they are not specific towards TAMs and peripheral toxicities are inevitable [129, 130].
A basic understanding of macrophages has led to unfolding of novel ideas for potential application of adoptive engineered macrophages, which exhibit limited phenotypes or enhanced therapeutic functions. On the other hand, macrophages display natural tumor site homing characteristics in response to the cytokines / chemokines released from TME [131]. This enables macrophages to emerge as potential therapeutic agents or diagnostic reagents to transfer to the tumor site. Therefore, three different kinds of genetic engineering therapy based on macrophages have been gradually developed: genetically engineered macrophages with enhanced therapeutic effects, macrophages as delivery tools, and macrophage derivatives as therapeutic carriers [117, 132,133,134].Although adoptive cell therapy using engineered macrophages has shown promising potential for the clinical application, however, mass production with strict quality control and potential biosafety problems are still two major challenges that must be overcome in the transition to the clinical application in the future. In addition, in-depth study of macrophage-mediated pharmacokinetics, pharmacodynamics, and phenotypic transformation could be of great significance to markedly improve the therapeutic efficacy and reduce side effects.
In the future, the treatment of tumor will not be limited to single or few drugs, and the combined use of multi-drugs and multi-pathways will be the inevitable trend for tumor therapy. Of course, there are still many complex problems to be solved in the dimension of TAMs. It is not clear whether the depletion of TAMs can be limited to the tumor without affecting monocytes/macrophages in the normal tissues and blood circulation? In addition, macrophage transcription factors that play a key role in promoting tumor immunosuppression and immune activation need to be identified? Moreover, it is not known whether the long-term maintenance of anti-tumor phenotype of macrophages can be regulated. Furthermore, it needs to be examined if CAR-M can infiltrate the tumors well through depletion of TAMs. In addition, although the traditional “M1/M2” dichotomy is still used in this paper, future studies should give sufficient functional and phenotypic markers on the basis of this classification, such as F4/80+CD11bloLy6clo/−TAMs, which should be able to more clearly reveal the different subsets of TAMs present in TME and pave the way for the future development of multi-targeted drugs.
Data availability
All data generated or analyzed in this study are included in this published article.
References
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global cancer statistics 2020: GLOBOCAN estimates of Incidence and mortality worldwide for 36 cancers in 185 countries. CA: Cancer J Clin. 2021;71:209–49.
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A, et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: Cancer J Clin. 2018;68:394–424.
Ahmed F, Perz JF, Kwong S, Jamison PM, Friedman C, Bell BP. National trends and disparities in the incidence of hepatocellular carcinoma, 1998–2003. Prev Chronic Dis. 2008;5:A74.
Zhou BB, Zhang H, Damelin M, Geles KG, Grindley JC, Dirks PB. Tumour-initiating cells: challenges and opportunities for anticancer drug discovery. Nat Rev Drug Discov. 2009;8:806–23.
Kalluri R. The biology and function of fibroblasts in cancer. Nat Rev Cancer. 2016;16:582–98.
Chen X, Song E. Turning foes to friends: targeting cancer-associated fibroblasts. Nat Rev Drug Discov. 2019;18:99–115.
Kim JH, Oh SH, Kim EJ, Park SJ, Hong SP, Cheon JH, et al. The role of myofibroblasts in upregulation of S100A8 and S100A9 and the differentiation of myeloid cells in the colorectal cancer microenvironment. Biochem Biophys Res Commun. 2012;423:60–6.
Qian BZ, Pollard JW. Macrophage diversity enhances tumor progression and metastasis. Cell. 2010;141:39–51.
Charoentong P, Finotello F, Angelova M, Mayer C, Efremova M, Rieder D, et al. Pan-cancer immunogenomic analyses reveal genotype-immunophenotype relationships and predictors of response to checkpoint blockade. Cell Rep. 2017;18:248–62.
Zhao Y, Shuen TWH, Toh TB, Chan XY, Liu M, Tan SY, et al. Development of a new patient-derived xenograft humanised mouse model to study human-specific tumour microenvironment and immunotherapy. Gut. 2018;67:1845–54.
Kim YJ, Rhee H, Yoo JE, Alves VAF, Kim GJ, Kim HM, et al. Tumour epithelial and stromal characteristics of hepatocellular carcinomas with abundant fibrous stroma: fibrolamellar versus scirrhous hepatocellular carcinoma. Histopathology. 2017;71:217–26.
Zhang Q, He Y, Luo N, Patel SJ, Han Y, Gao R, et al. Landscape and dynamics of single immune cells in hepatocellular carcinoma. Cell. 2019;179:829–84.e820.
Song G, Shi Y, Zhang M, Goswami S, Afridi S, Meng L, et al. Global immune characterization of HBV/HCV-related hepatocellular carcinoma identifies macrophage and T-cell subsets associated with disease progression. Cell Discov. 2020;6:90.
Li X, Liu R, Su X, Pan Y, Han X, Shao C, et al. Harnessing tumor-associated macrophages as aids for cancer immunotherapy. Mol Cancer. 2019;18:177.
Noy R, Pollard JW. Tumor-associated macrophages: from mechanisms to therapy. Immunity. 2014;41:49–61.
Mantovani A, Marchesi F, Malesci A, Laghi L, Allavena P. Tumour-associated macrophages as treatment targets in oncology. Nat Rev Clin Oncol. 2017;14:399–416.
Pathria P, Louis TL, Varner JA. Targeting tumor-associated macrophages in cancer. Trends Immunol. 2019;40:310–27.
Lahmar Q, Keirsse J, Laoui D, Movahedi K, Van Overmeire E, Van Ginderachter JA. Tissue-resident versus monocyte-derived macrophages in the tumor microenvironment. Biochim et Biophys Acta. 2016;1865:23–34.
Sharma A, Seow JJW, Dutertre CA, Pai R, Blériot C, Mishra A, et al. Onco-fetal reprogramming of endothelial cells drives immunosuppressive macrophages in hepatocellular carcinoma. Cell. 2020;183:377–94.e321.
Yang L, Zhang Y. Tumor-associated macrophages: from basic research to clinical application. J Hematol Oncol. 2017;10:58.
Arwert EN, Harney AS, Entenberg D, Wang Y, Sahai E, Pollard JW, et al. A unidirectional transition from migratory to perivascular macrophage is required for tumor cell intravasation. Cell Rep. 2018;23:1239–48. https://doi.org/10.1016/j.celrep.2018.04.007
Qian BZ, Li J, Zhang H, Kitamura T, Zhang J, Campion LR, et al. CCL2 recruits inflammatory monocytes to facilitate breast-tumour metastasis. Nature. 2011;475:222–5.
Li X, Yao W, Yuan Y, Chen P, Li B, Li J, et al. Targeting of tumour-infiltrating macrophages via CCL2/CCR2 signalling as a therapeutic strategy against hepatocellular carcinoma. Gut. 2017;66:157–67.
Tan HY, Wang N, Zhang C, Chan YT, Yuen MF, Feng Y. Lysyl oxidase-like 4 fosters an immunosuppressive microenvironment during hepatocarcinogenesis. Hepatology. 2021;73:2326–41.
Delire B, Henriet P, Lemoine P, Leclercq IA, Stärkel P. Chronic liver injury promotes hepatocarcinoma cell seeding and growth, associated with infiltration by macrophages. Cancer Sci. 2018;109:2141–52.
Wang J, Wang Y, Chu Y, Li Z, Yu X, Huang Z, et al. Tumor-derived adenosine promotes macrophage proliferation in human hepatocellular carcinoma. J Hepatol. 2021;74:627–37.
Hettinger J, Richards DM, Hansson J, Barra MM, Joschko AC, Krijgsveld J, et al. Origin of monocytes and macrophages in a committed progenitor. Nat Immunol. 2013;14:821–30.
van de Laar L, Saelens W, De Prijck S, Martens L, Scott CL, Van Isterdael G, et al. Yolk Sac macrophages, fetal liver, and adult monocytes can colonize an empty niche and develop into functional tissue-resident macrophages. Immunity. 2016;44:755–68.
Sieweke MH, Allen JE. Beyond stem cells: self-renewal of differentiated macrophages. Science. 2013;342:1242974.
Ye YC, Zhao JL, Lu YT, Gao CC, Yang Y, Liang SQ, et al. NOTCH signaling via WNT regulates the proliferation of alternative, CCR2-independent tumor-associated macrophages in hepatocellular carcinoma. Cancer Res. 2019;79:4160–72.
Scott CL, Zheng F, De Baetselier P, Martens L, Saeys Y, De Prijck S, et al. Bone marrow-derived monocytes give rise to self-renewing and fully differentiated Kupffer cells. Nat Commun. 2016;7:10321.
Krenkel O, Tacke F. Liver macrophages in tissue homeostasis and disease. Nat Rev Immunol. 2017;17:306–21.
Cassetta L, Pollard JW. Tumor-associated macrophages. Curr Biol: CB. 2020;30:R246–8.
Kretzschmar K, Watt FM. Lineage tracing. Cell. 2012;148:33–45.
Mereu E, Lafzi A, Moutinho C, Ziegenhain C, McCarthy DJ, Álvarez-Varela A, et al. Benchmarking single-cell RNA-sequencing protocols for cell atlas projects. Nat Biotechnol. 2020;38:747–55.
Ho DW, Tsui YM, Chan LK, Sze KM, Zhang X, Cheu JW, et al. Single-cell RNA sequencing shows the immunosuppressive landscape and tumor heterogeneity of HBV-associated hepatocellular carcinoma. Nat Commun. 2021;12:3684.
Ståhl PL, Salmén F, Vickovic S, Lundmark A, Navarro JF, Magnusson J, et al. Visualization and analysis of gene expression in tissue sections by spatial transcriptomics. Science. 2016;353:78–82.
Komohara Y, Fujiwara Y, Ohnishi K, Takeya M. Tumor-associated macrophages: potential therapeutic targets for anti-cancer therapy. Adv Drug Deliv Rev. 2016;99:180–5.
Edholm ES, Rhoo KH, Robert J. Evolutionary aspects of macrophages polarization. Results Probl Cell Differ. 2017;62:3–22.
Sprinzl MF, Puschnik A, Schlitter AM, Schad A, Ackermann K, Esposito I, et al. Sorafenib inhibits macrophage-induced growth of hepatoma cells by interference with insulin-like growth factor-1 secretion. J Hepatol. 2015;62:863–70.
Ao JY, Zhu XD, Chai ZT, Cai H, Zhang YY, Zhang KZ, et al. Colony-stimulating factor 1 receptor blockade inhibits tumor growth by altering the polarization of tumor-associated macrophages in hepatocellular carcinoma. Mol Cancer Ther. 2017;16:1544–54.
Duan Z, Luo Y. Targeting macrophages in cancer immunotherapy. Signal Transduct Target Ther. 2021;6:127.
Wan S, Kuo N, Kryczek I, Zou W, Welling TH. Myeloid cells in hepatocellular carcinoma. Hepatology. 2015;62:1304–12.
Perry CJ, Muñoz-Rojas AR, Meeth KM, Kellman LN, Amezquita RA, Thakral D, et al. Myeloid-targeted immunotherapies act in synergy to induce inflammation and antitumor immunity. J Exp Med. 2018;215:877–93.
He G, Dhar D, Nakagawa H, Font-Burgada J, Ogata H, Jiang Y, et al. Identification of liver cancer progenitors whose malignant progression depends on autocrine IL-6 signaling. Cell. 2013;155:384–96.
Ishtiaq SM, Arshad MI, Khan JA. PPARγ signaling in hepatocarcinogenesis: mechanistic insights for cellular reprogramming and therapeutic implications. Pharmacol Ther. 2022;240:108298.
Mantovani A, Barajon I, Garlanda C. IL-1 and IL-1 regulatory pathways in cancer progression and therapy. Immunol Rev. 2018;281:57–61.
van Kooten C, Banchereau J. CD40-CD40 ligand. J Leukoc Biol. 2000;67:2–17.
Zhang X, Zeng Y, Qu Q, Zhu J, Liu Z, Ning W, et al. PD-L1 induced by IFN-γ from tumor-associated macrophages via the JAK/STAT3 and PI3K/AKT signaling pathways promoted progression of lung cancer. Int J Clin Oncol. 2017;22:1026–33.
Qiao Y, Giannopoulou EG, Chan CH, Park SH, Gong S, Chen J, et al. Synergistic activation of inflammatory cytokine genes by interferon-γ-induced chromatin remodeling and toll-like receptor signaling. Immunity. 2013;39:454–69.
Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer. 2012;12:252–64.
Sica A, Mantovani A. Macrophage plasticity and polarization: in vivo veritas. J Clin Investig. 2012;122:787–95.
Hoch T, Schulz D, Eling N, Gómez JM, Levesque MP, Bodenmiller B. Multiplexed imaging mass cytometry of the chemokine milieus in melanoma characterizes features of the response to immunotherapy. Sci Immunol. 2022;7:eabk1692.
House IG, Savas P, Lai J, Chen AXY, Oliver AJ, Teo ZL, et al. Macrophage-derived CXCL9 and CXCL10 are required for antitumor immune responses following immune checkpoint blockade. Clin Cancer Res. 2020;26:487–504.
Chow MT, Ozga AJ, Servis RL, Frederick DT, Lo JA, Fisher DE, et al. Intratumoral activity of the CXCR3 chemokine system is required for the efficacy of anti-PD-1 therapy. Immunity. 2019;50:1498–151.e1495.
Wu L, Zhang X, Zheng L, Zhao H, Yan G, Zhang Q, et al. RIPK3 orchestrates fatty acid metabolism in ttumor-associated macrophages and hepatocarcinogenesis. Cancer Immunol Res. 2020;8:710–21.
Zong Z, Zou J, Mao R, Ma C, Li N, Wang J, et al. M1 macrophages induce PD-L1 expression in hepatocellular carcinoma cells through IL-1β signaling. Front Immunol. 2019;10:1643.
Tian X, Wu Y, Yang Y, Wang J, Niu M, Gao S, et al. Long noncoding RNA LINC00662 promotes M2 macrophage polarization and hepatocellular carcinoma progression via activating Wnt/β-catenin signaling. Mol Oncol. 2020;14:462–83.
Zhang YL, Li Q, Yang XM, Fang F, Li J, Wang YH, et al. SPON2 promotes M1-like macrophage recruitment and inhibits hepatocellular carcinoma metastasis by distinct integrin-Rho GTPase-Hippo pathways. Cancer Res. 2018;78:2305–17.
Dong N, Shi X, Wang S, Gao Y, Kuang Z, Xie Q, et al. M2 macrophages mediate sorafenib resistance by secreting HGF in a feed-forward manner in hepatocellular carcinoma. Br J Cancer. 2019;121:22–33.
Li Z, Li H, Zhao ZB, Zhu W, Feng PP, Zhu XW, et al. SIRT4 silencing in tumor-associated macrophages promotes HCC development via PPARδ signalling-mediated alternative activation of macrophages. J Exp Clin Cancer Res: CR. 2019;38:469.
Wang N, Liang H, Zen K. Molecular mechanisms that influence the macrophage m1-m2 polarization balance. Front Immunol. 2014;5:614.
Xu G, Feng D, Yao Y, Li P, Sun H, Yang H, et al. Listeria-based hepatocellular carcinoma vaccine facilitates anti-PD-1 therapy by regulating macrophage polarization. Oncogene. 2020;39:1429–44.
Ng TH, Britton GJ, Hill EV, Verhagen J, Burton BR, Wraith DC, et al. Regulation of adaptive immunity; the role of interleukin-10. Front Immunol. 2013;4:129.
Savage ND, de Boer T, Walburg KV, Joosten SA, van Meijgaarden K, Geluk A, et al. Human anti-inflammatory macrophages induce Foxp3+ GITR+ CD25+ regulatory T cells, which suppress via membrane-bound TGFbeta-1. J Immunol. 2008;181:2220–6.
Pollard JW. Tumour-educated macrophages promote tumour progression and metastasis. Nat Rev Cancer. 2004;4:71–8.
Cao W, Liu Y, Zhang R, Zhang B, Wang T, Zhu X, et al. Homoharringtonine induces apoptosis and inhibits STAT3 via IL-6/JAK1/STAT3 signal pathway in Gefitinib-resistant lung cancer cells. Sci Rep. 2015;5:8477.
Oh SA, Li MO. TGF-β: guardian of T cell function. J Immunol. 2013;191:3973–9.
Maller O, Drain AP, Barrett AS, Borgquist S, Ruffell B, Zakharevich I, et al. Tumour-associated macrophages drive stromal cell-dependent collagen crosslinking and stiffening to promote breast cancer aggression. Nat Mater. 2021;20:548–59.
Miura K, Yang L, van Rooijen N, Ohnishi H, Seki E. Hepatic recruitment of macrophages promotes nonalcoholic steatohepatitis through CCR2. Am J Physiol Gastrointest Liver Physiol. 2012;302:G1310–21.
Guan B, Li H, Yao J, Guo J, Yu F, Li G, et al. CCL3-CCR5 axis promotes cell migration and invasion of colon adenocarcinoma via Akt signaling pathway. Environ Toxicol. 2023;38:172–84.
Takai H, Ashihara M, Ishiguro T, Terashima H, Watanabe T, Kato A, et al. Involvement of glypican-3 in the recruitment of M2-polarized tumor-associated macrophages in hepatocellular carcinoma. Cancer Biol Ther. 2009;8:2329–38.
Zhang A, Xu Y, Xu H, Ren J, Meng T, Ni Y, et al. Lactate-induced M2 polarization of tumor-associated macrophages promotes the invasion of pituitary adenoma by secreting CCL17. Theranostics. 2021;11:3839–52.
Wang D, Yang L, Yue D, Cao L, Li L, Wang D, et al. Macrophage-derived CCL22 promotes an immunosuppressive tumor microenvironment via IL-8 in malignant pleural effusion. Cancer Lett. 2019;452:244–53.
Chen Y, Yang D, Li S, Gao Y, Jiang R, Deng L, et al. Development of a Listeria monocytogenes-based vaccine against hepatocellular carcinoma. Oncogene. 2012;17:2140–52.
Duffield JS, Forbes SJ, Constandinou CM, Clay S, Partolina M, Vuthoori S, et al. Selective depletion of macrophages reveals distinct, opposing roles during liver injury and repair. J Clin Investig. 2005;115:56–65.
Guilliams M, Thierry GR, Bonnardel J, Bajenoff M. Establishment and Maintenance of the Macrophage Niche. Immunity. 2020;52:434–51.
El-Serag HB. Hepatocellular carcinoma. N. Engl J Med. 2011;365:1118–27.
Heimbach JK, Kulik LM, Finn RS, Sirlin CB, Abecassis MM, Roberts LR, et al. AASLD guidelines for the treatment of hepatocellular carcinoma. Hepatology. 2018;67:358–80.
Llovet JM, Zucman-Rossi J, Pikarsky E, Sangro B, Schwartz M, Sherman M, et al. Hepatocellular carcinoma. Nat Rev Dis Prim. 2016;2:16018.
Forner A, Reig M, Bruix J. Hepatocellular carcinoma. Lancet. 2018;391:1301–14.
Chang CC, Dinh TK, Lee YA, Wang FN, Sung YC, Yu PL, et al. Nanoparticle delivery of MnO(2) and antiangiogenic therapy to overcome hypoxia-driven tumor escape and suppress hepatocellular carcinoma. ACS Appl Mater Interfaces. 2020;12:44407–19.
Li L, Sun P, Zhang C, Li Z, Zhou W. MiR-98 suppresses the effects of tumor-associated macrophages on promoting migration and invasion of hepatocellular carcinoma cells by regulating IL-10. Biochimie. 2018;150:23–30.
Chai ZT, Zhu XD, Ao JY, Wang WQ, Gao DM, Kong J, et al. microRNA-26a suppresses recruitment of macrophages by downregulating macrophage colony-stimulating factor expression through the PI3K/Akt pathway in hepatocellular carcinoma. J Hematol Oncol. 2015;8:56.
Man K, Ng KT, Xu A, Cheng Q, Lo CM, Xiao JW, et al. Suppression of liver tumor growth and metastasis by adiponectin in nude mice through inhibition of tumor angiogenesis and downregulation of Rho kinase/IFN-inducible protein 10/matrix metalloproteinase 9 signaling. Clin Cancer Res. 2010;16:967–77.
Cannarile MA, Weisser M, Jacob W, Jegg AM, Ries CH, Rüttinger D. Colony-stimulating factor 1 receptor (CSF1R) inhibitors in cancer therapy. J Immunother Cancer. 2017;5:53.
Kuang DM, Zhao Q, Peng C, Xu J, Zhang JP, Wu C, et al. Activated monocytes in peritumoral stroma of hepatocellular carcinoma foster immune privilege and disease progression through PD-L1. J Exp Med. 2009;206:1327–37.
Shigeta K, Datta M, Hato T, Kitahara S, Chen IX, Matsui A, et al. Dual programmed death receptor-1 and vascular endothelial growth factor receptor-2 blockade promotes vascular normalization and enhances antitumor immune responses in hepatocellular carcinoma. Hepatology. 2020;71:1247–61.
Formenti SC, Demaria S. Systemic effects of local radiotherapy. Lancet Oncol. 2009;10:718–26.
Adams JL, Smothers J, Srinivasan R, Hoos A. Big opportunities for small molecules in immuno-oncology. Nat Rev Drug Discov. 2015;14:603–22.
Zhuang Y, Zhao X, Yuan B, Zeng Z, Chen Y. Blocking the CCL5-CCR5 axis using maraviroc promotes M1 polarization of macrophages cocultured with irradiated hepatoma cells. J Hepatocell Carcinoma. 2021;8:599–611.
Zhu Y, Yang J, Xu D, Gao XM, Zhang Z, Hsu JL, et al. Disruption of tumour-associated macrophage trafficking by the osteopontin-induced colony-stimulating factor-1 signalling sensitises hepatocellular carcinoma to anti-PD-L1 blockade. Gut. 2019;68:1653–66.
Ma J, Zheng B, Goswami S, Meng L, Zhang D, Cao C, et al. PD1(Hi) CD8(+) T cells correlate with exhausted signature and poor clinical outcome in hepatocellular carcinoma. J Immunother cancer. 2019;7:331.
Petty, AJ, Dai, R, Lapalombella, R, Baiocchi, RA, Benson, DM, et al. Hedgehog-induced PD-L1 on tumor-associated macrophages is critical for suppression of tumor-infiltrating CD8+ T cell function. JCI Insight. 2021. https://doi.org/10.1172/jci.insight.146707.
Chen J, Zheng DX, Yu XJ, Sun HW, Xu YT, Zhang YJ, et al. Macrophages induce CD47 upregulation via IL-6 and correlate with poor survival in hepatocellular carcinoma patients. Oncoimmunology. 2019;8:e1652540.
Xiao Z, Chung H, Banan B, Manning PT, Ott KC, Lin S, et al. Antibody mediated therapy targeting CD47 inhibits tumor progression of hepatocellular carcinoma. Cancer Lett. 2015;360:302–9.
Willingham SB, Volkmer JP, Gentles AJ, Sahoo D, Dalerba P, Mitra SS, et al. The CD47-signal regulatory protein alpha (SIRPa) interaction is a therapeutic target for human solid tumors. Proc Natl Acad Sci USA. 2012;109:6662–7.
Du K, Li Y, Liu J, Chen W, Wei Z, Luo Y, et al. A bispecific antibody targeting GPC3 and CD47 induced enhanced antitumor efficacy against dual antigen-expressing HCC. Mol Ther: J Am Soc Gene Ther. 2021;29:1572–84.
Bouwstra R, He Y, de Boer J, Kooistra H, Cendrowicz E, Fehrmann RSN, et al. CD47 expression defines efficacy of Rituximab with CHOP in Non-Germinal Center B-cell (Non-GCB) diffuse large B-cell lymphoma patients (DLBCL), but Not in GCB DLBCL. Cancer Immunol Res. 2019;7:1663–71.
Shi Y, Fan X, Deng H, Brezski RJ, Rycyzyn M, Jordan RE, et al. Trastuzumab triggers phagocytic killing of high HER2 cancer cells in vitro and in vivo by interaction with Fcγ receptors on macrophages. J Immunol. 2015;194:4379–86.
Cheng X, Li L, Thorpe PE, Yopp AC, Brekken RA, Huang X. Antibody-mediated blockade of phosphatidylserine enhances the antitumor effect of sorafenib in hepatocellular carcinomas xenografts. Ann Surgical Oncol. 2016;23:583–91.
Gauttier V, Judor JP, Le Guen V, Cany J, Ferry N, Conchon S. Agonistic anti-CD137 antibody treatment leads to antitumor response in mice with liver cancer. Int J Cancer. 2014;135:2857–67.
Teillaud JL. From whole monoclonal antibodies to single domain antibodies: think small. Methods Mol Biol. 2012;911:3–13.
Wang Y, Tiruthani K, Li S, Hu M, Zhong G, Tang Y, et al. mRNA delivery of a bispecific single-domain antibody to polarize tumor-associated macrophages and synergize immunotherapy against liver malignancies. Adv Mater. 2021;33:e2007603.
Thomas JA, Badini M. The role of innate immunity in spontaneous regression of cancer. Indian J Cancer. 2011;48:246–51.
Palucka K, Banchereau J. Dendritic-cell-based therapeutic cancer vaccines. Immunity. 2013;39:38–48.
Santos PM, Butterfield LH. Dendritic cell-based cancer vaccines. J Immunol. 2018;200:443–9.
Woo SR, Corrales L, Gajewski TF. Innate immune recognition of cancer. Annu Rev Immunol. 2015;33:445–74.
Chen C, Hou J, Lin Z, Yao M, Jiang R, Wang Y, et al. A bystander cell-based GM-CSF secreting vaccine synergized with a low dose of cyclophosphamide presents therapeutic immune responses against murine hepatocellular carcinoma. Cell Mol Immunol. 2013;10:349–59.
Feng R, Morine Y, Ikemoto T, Imura S, Iwahashi S, Saito Y, et al. Nrf2 activation drive macrophages polarization and cancer cell epithelial-mesenchymal transition during interaction. Cell Commun Signal: CCS. 2018;16:54.
June CH, Sadelain M. Chimeric antigen receptor therapy. N. Engl J Med. 2018;379:64–73.
Manfredi F, Cianciotti BC, Potenza A, Tassi E, Noviello M, Biondi A, et al. TCR redirected T cells for cancer treatment: achievements, hurdles, and goals. Front Immunol. 2020;11:1689.
Song L, Asgharzadeh S, Salo J, Engell K, Wu HW, Sposto R, et al. Valpha24-invariant NKT cells mediate antitumor activity via killing of tumor-associated macrophages. J Clin Investig. 2009;119:1524–36.
Bagley SJ, O’Rourke DM. Clinical investigation of CAR T cells for solid tumors: lessons learned and future directions. Pharmacol Ther. 2020;205:107419.
Yang Y, Sun M, Yao W, Wang F, Li X, Wang W, et al. Compound kushen injection relieves tumor-associated macrophage-mediated immunosuppression through TNFR1 and sensitizes hepatocellular carcinoma to sorafenib. J Immunother Cancer. 2020. https://doi.org/10.1136/jitc-2019-000317.
Wu X, Luo H, Shi B, Di S, Sun R, Su J, et al. Combined antitumor effects of sorafenib and GPC3-CAR T cells in mouse models of hepatocellular carcinoma. Mol Ther: J Am Soc Gene Ther. 2019;27:1483–94.
Klichinsky M, Ruella M, Shestova O, Lu XM, Best A, Zeeman M, et al. Human chimeric antigen receptor macrophages for cancer immunotherapy. Nat Biotechnol. 2020;38:947–53.
Mura S, Nicolas J, Couvreur P. Stimuli-responsive nanocarriers for drug delivery. Nat Mater. 2013;12:991–1003.
Liu LY, Ma XZ, Ouyang B, Ings DP, Marwah S, Liu J, et al. Nanoparticle uptake in a spontaneous and immunocompetent woodchuck liver cancer model. ACS Nano. 2020;14:4698–715.
Fernández-Varo G, Perramón M, Carvajal S, Oró D, Casals E, Boix L, et al. Bespoken nanoceria: an effective treatment in experimental hepatocellular carcinoma. Hepatology. 2020;72:1267–82.
Hoshino A, Costa-Silva B, Shen TL, Rodrigues G, Hashimoto A, Tesic Mark M, et al. Tumour exosome integrins determine organotropic metastasis. Nature. 2015;527:329–35.
Costa-Silva B, Aiello NM, Ocean AJ, Singh S, Zhang H, Thakur BK, et al. Pancreatic cancer exosomes initiate pre-metastatic niche formation in the liver. Nat cell Biol. 2015;17:816–26.
Loyher PL, Hamon P, Laviron M, Meghraoui-Kheddar A, Goncalves E, Deng Z, et al. Macrophages of distinct origins contribute to tumor development in the lung. J Exp Med. 2018;215:2536–53.
Etzerodt A, Moulin M, Doktor TK, Delfini M, Mossadegh-Keller N, Bajenoff M, et al. Tissue-resident macrophages in omentum promote metastatic spread of ovarian cancer. J Exp Med. 2020. https://doi.org/10.1084/jem.20191869.
Müller A, Brandenburg S, Turkowski K, Müller S, Vajkoczy P. Resident microglia, and not peripheral macrophages, are the main source of brain tumor mononuclear cells. Int J Cancer. 2015;137:278–88.
Dumas AA, Pomella N, Rosser G, Guglielmi L, Vinel C, Millner TO, et al. Microglia promote glioblastoma via mTOR-mediated immunosuppression of the tumour microenvironment. EMBO J. 2020;39:e103790.
Franklin RA, Liao W, Sarkar A, Kim MV, Bivona MR, Liu K, et al. The cellular and molecular origin of tumor-associated macrophages. Science. 2014;344:921–5.
Zhu Y, Herndon JM, Sojka DK, Kim KW, Knolhoff BL, Zuo C, et al. Tissue-resident macrophages in pancreatic ductal adenocarcinoma originate from embryonic hematopoiesis and promote tumor progression. Immunity. 2017;47:323–33.e326.
Bolli E, Scherger M, Arnouk SM, Pombo Antunes AR, Straßburger D, Urschbach M, et al. Targeted repolarization of tumor-associated macrophages via imidazoquinoline-linked nanobodies. Adv Sci. 2021;8:2004574.
De Vlaminck K, Romão E, Puttemans J, Pombo Antunes AR, Kancheva D, Scheyltjens I, et al. Imaging of glioblastoma tumor-associated myeloid cells using nanobodies targeting signal regulatory protein alpha. Front Immunol. 2021;12:777524.
Bottazzi B, Polentarutti N, Acero R, Balsari A, Boraschi D, Ghezzi P, et al. Regulation of the macrophage content of neoplasms by chemoattractants. Science. 1983;220:210–2.
Zhang Y, Zhang Y, Guo Q, Guo Z, Chen X, Liu L, et al. Trained macrophage bioreactor for penetrating delivery of fused antitumor protein. ACS Appl Mater Interfaces. 2019;11:23018–25.
Pang L, Zhu Y, Qin J, Zhao W, Wang J. Primary M1 macrophages as multifunctional carrier combined with PLGA nanoparticle delivering anticancer drug for efficient glioma therapy. Drug Deliv. 2018;25:1922–31.
Wang P, Wang H, Huang Q, Peng C, Yao L, Chen H, et al. Exosomes from M1-polarized macrophages enhance paclitaxel antitumor activity by activating macrophages-mediated inflammation. Theranostics. 2019;9:1714–27.
Gomez Perdiguero E, Klapproth K, Schulz C, Busch K, Azzoni E, Crozet L, et al. Tissue-resident macrophages originate from yolk-sac-derived erythro-myeloid progenitors. Nature. 2015;518:547–51.
Mass E, Ballesteros I, Farlik M, Halbritter F, Günther P, Crozet L, et al. Specification of tissue-resident macrophages during organogenesis. Science. 2016. https://doi.org/10.1126/science.aaf4238.
Schulz C, Gomez Perdiguero E, Chorro L, Szabo-Rogers H, Cagnard N, Kierdorf K, et al. A lineage of myeloid cells independent of Myb and hematopoietic stem cells. Science. 2012;336:86–90.
Wynn TA, Chawla A, Pollard JW. Macrophage biology in development, homeostasis and disease. Nature. 2013;496:445–55.
Yona S, Kim KW, Wolf Y, Mildner A, Varol D, Breker M, et al. Fate mapping reveals origins and dynamics of monocytes and tissue macrophages under homeostasis. Immunity. 2013;38:79–91.
Sierro F, Evrard M, Rizzetto S, Melino M, Mitchell AJ, Florido M, et al. A liver capsular network of monocyte-derived macrophages restricts hepatic dissemination of intraperitoneal bacteria by neutrophil recruitment. Immunity. 2017;47:374–38.e376.
Jakubzick CV, Randolph GJ, Henson PM. Monocyte differentiation and antigen-presenting functions. Nat Rev Immunol. 2017;17:349–62.
Corzo CA, Condamine T, Lu L, Cotter MJ, Youn JI, Cheng P, et al. HIF-1α regulates function and differentiation of myeloid-derived suppressor cells in the tumor microenvironment. J Exp Med. 2010;207:2439–53.
Kapanadze T, Gamrekelashvili J, Ma C, Chan C, Zhao F, Hewitt S, et al. Regulation of accumulation and function of myeloid derived suppressor cells in different murine models of hepatocellular carcinoma. J Hepatol. 2013;59:1007–13.
Acknowledgements
This study was supported by grants from the National Natural Science Foundation of China (No. 81472302/No. 82003040), Natural Science Foundation of Liaoning Province (2020-BS-103), China Postdoctoral Science Foundation (2020M681016), Xingliaoyingcaijihua Project of Liaoning Province (XLYC1902050), Qingnianyingcaijihua Project of China Medical University, Science Foundation of Education Department of Liaoning Province (LJKZ0770; LJKZ0768), China Medical University General Project of National Natural Science Foundation of China (3111210512), and Shenyang Science and Technology Innovation Platform Construction Project (21-104-0-04).
Author information
Authors and Affiliations
Contributions
HZ and XP drafted this review and designed the tables and figures. LY gave some valuable suggestions. HL provided guidance and revised the manuscript. LY and HL gave the final approval of the version to be submitted and obtained the funding. All authors approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zheng, H., Peng, X., Yang, S. et al. Targeting tumor-associated macrophages in hepatocellular carcinoma: biology, strategy, and immunotherapy. Cell Death Discov. 9, 65 (2023). https://doi.org/10.1038/s41420-023-01356-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41420-023-01356-7
- Springer Nature Limited
We’re sorry, something doesn't seem to be working properly.
Please try refreshing the page. If that doesn't work, please contact support so we can address the problem.