Abstract
Multiple myeloma (MM) patients typically receive several lines of combination therapy and first-line treatment commonly includes lenalidomide. As patients age, they become less tolerant to treatment, requiring convenient/tolerable/lenalidomide-free options. Carfilzomib and/or bortezomib-exposed/intolerant, lenalidomide-refractory MM patients with ≥2 prior lines of therapy were randomized 3:2 to ixazomib-dexamethasone (ixa-dex) (n = 73) or pomalidomide-dexamethasone (pom-dex) (n = 49) until progression/toxicity. Median progression-free survival (mPFS) was 7.1 vs 4.8 months with ixa-dex vs pom-dex (HR 0.847, 95% CI 0.535–1.341, P = 0.477; median follow-up: 15.3 vs 17.3 months); there was no statistically significant difference between arms. In patients with 2 and ≥3 prior lines of therapy, respectively, mPFS was 11.0 vs 5.7 months (HR 1.083, 95% CI 0.547–2.144) and 5.7 vs 3.7 months (HR 0.686, 95% CI 0.368–1.279). Among ixa-dex vs pom-dex patients, 69% vs 81% had Grade ≥3 treatment-emergent adverse events (TEAEs), 51% vs 53% had serious TEAEs, 39% vs 36% had TEAEs leading to drug discontinuation, 44% vs 32% had TEAEs leading to dose reduction, and 13% vs 13% died on study. Quality of life was similar between arms and maintained during treatment. Ixa-dex represents an important lenalidomide-free, oral option for this heavily pretreated, lenalidomide-refractory, proteasome inhibitor-exposed population.
Trial registration: ClinicalTrials.gov number, NCT03170882.
Similar content being viewed by others
Introduction
For most patients with multiple myeloma (MM), the development of relapsed and refractory MM (RRMM) is inevitable, requiring several lines of therapy with multiple drug combinations throughout the course of treatment [1,2,3]. Consequently, the disease may become refractory to more than one agent, and patients receiving their third or later line of therapy are likely to have poorer responses than in earlier lines, making treatment more challenging [4,5,6,7,8,9,10].
RRMM is a heterogeneous disease, and no single treatment regimen is effective for all patients [1, 2, 11]. Since lenalidomide-containing regimens are common as first-line therapy in MM [1, 2, 12], lenalidomide-free options for subsequent lines are needed as patients become refractory to this agent [13,14,15]. Several lenalidomide-free triplet regimens are approved in this setting [14,15,16,17,18,19,20,21,22,23]. These combinations, however, are parenterally administered, increasing the treatment burden for the already heavily pretreated patient, suggesting a need for options that can limit hospital or clinic visits [24]. Residual comorbidities or impairments from previous therapies are common in this population [25], and, as patients have aged several years since therapy onset, they are likely to be frailer and potentially less able to tolerate treatment toxicity [10, 12, 25], notably in the context of standard-of-care triplet regimens [19, 26,27,28,29]. Therefore, there is a need for more convenient regimens, such as all-oral doublets, that are active, have manageable toxicity, and do not adversely impact quality of life (QoL).
Dexamethasone-based doublets have been shown to be effective and tolerable in patients with RRMM [30, 31], and pomalidomide-dexamethasone (pom-dex) is indicated for use in adults with RRMM who have received ≥2 prior treatment regimens, including both lenalidomide and bortezomib, and have progressed on their last therapy [30]. However, treatment with pom-dex is associated with Grade ≥3 hematological toxicities and infections [18, 32]. In addition, for a patient population previously exposed to pom-dex-based triplets or those who have become refractory to pomalidomide, alternative and effective all-oral doublet options may be required.
Ixazomib, the first oral proteasome inhibitor (PI), is approved in the United States and European Union in combination with lenalidomide-dexamethasone for MM patients who have received ≥1 prior line of therapy [33, 34]. Ixazomib is well tolerated as long-term therapy, with predictable and manageable toxicities [26]. A Phase 2 study in which RRMM patients with limited or no prior exposure to bortezomib were treated with ixazomib 4 or 5.5 mg weekly for 3 of 4 weeks in combination with dexamethasone 40 mg weekly reported overall response rates (ORRs) of 31% for the ixazomib 4 mg arm and 54% for the 5.5 mg arm, with both arms demonstrating manageable toxicities. In that study, the authors suggest that ixazomib-dexamethasone (ixa-dex), at either dose of ixazomib, may provide greater clinical benefit compared with pom-dex in this population of patients with limited prior exposure to a PI [35, 36]. These findings provided the rationale for the present global, multicenter, open-label, randomized, Phase 2 study of ixa-dex vs pom-dex in adult patients with lenalidomide-refractory, PI-exposed MM.
Methods
Patients
Eligible patients had a confirmed diagnosis of MM per International Myeloma Working Group (IMWG) criteria, an Eastern Cooperative Oncology Group performance score of 0–2, had relapsed or progressed after ≥2 prior lines of therapy, and were lenalidomide-refractory. Patients had to have achieved ≥partial response (PR) to carfilzomib or bortezomib, or had to have discontinued treatment with either PI due to intolerance (see Supplementary Table 1 for detailed eligibility criteria).
Study design
In this international, multicenter, open-label Phase 2 study, patients were centrally randomized (3:2) via interactive response technology to receive either ixa-dex or pom-dex in 28-day cycles. Patients on the ixa-dex arm received oral ixazomib 4 mg on Days 1, 8, and 15, and oral dexamethasone 20 mg (10 mg in patients aged ≥75 years at randomization) on Days 1, 2, 8, 9, 15, 16, 22, and 23. The starting dose of ixazomib 4 mg was selected based on the dose used in TOURMALINE-MM1 [26], with escalation to 5.5 mg at Cycle 2 if the 4 mg dose was well tolerated [35, 36]. Patients on pom-dex received oral pomalidomide 4 mg on Days 1–21 and oral dexamethasone 40 mg (20 mg in patients aged ≥75 years at randomization) on Days 1, 8, 15, and 22 as per license. Patients continued treatment until progressive disease (PD) or unacceptable toxicity. Randomization was stratified by age (<65 vs ≥65 years), International Staging System (ISS) disease stage at study entry (I or II vs III), and number of prior lines of therapy (2 vs ≥3).
The primary endpoint was progression-free survival (PFS), defined as time from randomization to first documentation of PD as assessed by the investigator per IMWG criteria [37], or death from any cause, whichever occurred first. Secondary endpoints were overall survival (OS), ORR (≥ PR), duration of response (DOR), time to response (TTR), time to progression (TTP), health-related QoL (HRQoL), healthcare resource utilization (HRU), and safety.
The trial was conducted in accordance with the Declaration of Helsinki, International Conference on Harmonization Good Clinical Practice guideline, and appropriate regulatory requirements. Local ethics committees or institutional review boards approved the protocol and all patients provided written informed consent.
Assessments
Response assessments were performed every cycle until PD or every 4 weeks in patients who discontinued treatment prior to PD. Response and disease progression assessments were based on central laboratory results and IMWG 2011 criteria [38]. A single bone marrow (BM) aspirate or biopsy disease assessment was performed locally at screening and repeated if the patient showed evidence of complete response (CR), or to investigate suspected PD. To confirm suspected CR, BM immunohistochemistry or immunofluorescence for kappa/lambda ratio was performed. Following PD, patients were followed up every 12 weeks for subsequent therapy and OS.
Safety was assessed throughout the study, and treatment-emergent adverse events (TEAEs) were graded using the National Cancer Institute’s Common Terminology Criteria for Adverse Events, version 4.03. If peripheral neuropathy (PN) occurred, each subsequent monthly evaluation recorded the grade of PN at that visit. PN was followed monthly until either resolution, initiation of a subsequent line of therapy, or 6 months after PD, whichever occurred first.
HRQoL was evaluated through patient self-reported instruments including the European Organization for Research and Treatment of Cancer (EORTC) Quality of Life Questionnaire (QLQ) Core-30 (C30) and myeloma-specific (MY-20) instruments, and the 5-level classification system of the EuroQol 5-Dimensional Health Questionnaire (EQ-5D-5L). These assessments were obtained at screening and at every cycle until PD. In patients who discontinued treatment prior to PD, assessments were made every 4 weeks, and the EQ-5D-5L was completed every 12 weeks. HRU data included medical visits (number and rates, reasons for, and length of stay): hospitalizations, emergency room visits, and outpatient visits.
Statistical analysis
The analyses of the primary endpoint, PFS, and the secondary endpoints of TTP and OS were conducted in the intent-to-treat (ITT; all randomized patients) population. Kaplan–Meier methodology was used to estimate PFS, and OS distributions in each treatment arm, as well as TTP distributions. DOR was summarized descriptively for responders using the Kaplan–Meier method; TTR was compared in the ITT population and summarized descriptively for the responders. Patients without documentation of PD were censored at the date of the last response assessment (≥stable disease on PFS, TTP and DOR analyses), and patients without documentation of death at the time of OS analysis were censored at the date last known to be alive. Kaplan–Meier medians, plus 95% confidence intervals (CIs), were determined, if estimable. An unadjusted, unstratified Cox model was used to estimate the hazard ratio (HR) and 95% CI for the treatment effect, and a 2-sided, unstratified log-rank test was used to compare treatment groups for each endpoint. Subgroup analyses were conducted for PFS and OS relative to baseline stratification factors, demographic data, and disease characteristics.
A sample size of ~120 patients was determined in order to give ~81 events for the PFS analysis, which provided 80% power at a 2-sided alpha of 0.20 to detect a difference between arms; this was based on an assumption of a HR of 0.62 and a median PFS of 7.3 months for ixa-dex vs 4.5 months for pom-dex (estimated based on previous published data in similar populations [36, 39]). OS was to be tested at 2-sided alpha of 0.20; the study was not powered for OS comparisons.
ORR was defined as the proportion of patients who achieved ≥PR in the ITT population. A logistic regression model was used to estimate the treatment effect in terms of odds ratio (OR) and its 95% CIs.
This report represents the final analysis of the study. A Phase 3 portion in a larger sample size was planned, but this was removed following a protocol amendment in response to slower-than-projected accumulation of PFS events and contemporaneous advances in standard-of-care approaches in the RRMM treatment paradigm.
Results
Patients
Between February 28, 2018, and October 3, 2019, 122 patients from 51 sites in 13 countries in Europe and the Middle East, and 3 sites in Australia were randomized: 73 to the ixa-dex arm and 49 to the pom-dex arm (Fig. 1). Baseline demographics and disease characteristics were generally well-balanced between the two treatment arms (Table 1). Median age in patients treated with ixa-dex vs pom-dex was 72 vs 68 years (75% vs 73% were aged ≥65 years and 36% vs 18% were aged ≥75 years), 25% vs 22% of patients had ISS stage III MM, and 52% vs 53% had received ≥3 prior lines of therapy per stratification (prior lines of therapy are detailed in Supplementary Table 2).
At data cutoff (May 31, 2020), 19% ixa-dex vs 20% pom-dex patients were ongoing on study treatment (Fig. 1). Study treatment had been discontinued in 79% vs 76% patients; primary reasons for discontinuation were PD in 47% vs 57%, and adverse events (AEs) in 23% vs 12% of patients (Fig. 1).
Efficacy
With 46 (63%) and 34 (69%) PFS events in the ixa-dex and pom-dex arms, respectively, median PFS was 7.1 months (95% CI 3.9–11.1) vs 4.8 months (95% CI 3.7–8.5), and the HR was 0.847 (95% CI 0.535–1.341; P = 0.477; Fig. 2A). Median PFS with ixa-dex vs pom-dex in patients with 2 prior lines of therapy was 11.0 vs 5.7 months (HR 1.083, 95% CI 0.547–2.144); in patients with ≥3 prior lines, median PFS was 5.7 vs 3.7 months (HR 0.686, 95% CI 0.368–1.279). Figure 2B shows PFS with ixa-dex vs pom-dex in all prespecified subgroups based on patient and disease characteristics.
A Kaplan–Meier analysis of PFS and (B) Forest plot of PFS in prespecified subgroups based on patient and disease characteristics. ALT alanine aminotransferase, AST aspartate aminotransferase, CI confidence interval, dex dexamethasone, ECOG Eastern Cooperative Oncology Group, HR hazard ratio, ISS International Staging System, ITT intent-to-treat, ixa ixazomib, NE not estimable, PFS progression-free survival, pom pomalidomide, ULN upper limit of normal.
Although age (<65 vs ≥65 years) was a stratification factor, an imbalance in the number of elderly patients was observed in the ixa-dex vs pom-dex arms (32% vs 18% of patients were aged ≥75 to ≤84 years, and 4% vs 0% were aged ≥85 years). Consequently, an ad-hoc analysis of PFS with ixa-dex vs pom-dex in patients aged <75 years (median 6.5 vs 4.8 months, HR 0.843, 95% CI 0.491–1.448) and ≥75 years (median 8.0 vs 4.7 months, HR 0.890, 95% CI 0.309–2.562) is provided in Supplementary Fig. 1.
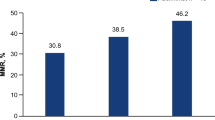
Overall, 28 vs 20 patients had ≥PR to treatment with ixa-dex vs pom-dex for an ORR of 38% vs 41% (OR 0.90, 95% CI 0.43–1.90) with ≥very good PR (VGPR) rates of 7% vs 16% (Table 2). Among responding patients, median TTR was 2.0 vs 1.1 months (HR 0.556, 95% CI 0.288–1.073) and median DOR was 14.8 vs 14.3 months (Supplementary Fig. 2).
Median TTP was 8.4 vs 5.1 months (HR 0.830, 95% CI 0.506–1.361) with ixa-dex vs pom-dex (Fig. 3A). After a median follow-up of 15.3 and 17.3 months, and with 29 (40%) and 15 (31%) patients having died in the ixa-dex and pom-dex arms, respectively, median OS was 18.8 months vs not reached (HR 1.427, 95% CI 0.761–2.677; Fig. 3B). OS in patients aged <75 and ≥75 years is shown in Supplementary Fig. 1, and the forest plot of OS in prespecified patient subgroups is shown in Supplementary Fig. 3.
Overall, 30 (42%) vs 23 (49%) of patients in the ixa-dex vs pom-dex arms had started subsequent lines of therapy at data cutoff for this analysis (Supplementary Table 3). Subsequent therapy containing immunomodulatory drugs was received by 32% vs 9% of patients, including 28% vs 4% who received pom; by contrast, 10% vs 36% of patients received PIs, and 19% vs 28% received monoclonal antibodies.
Treatment exposure and safety
The safety population included 72 and 47 patients in the ixa-dex and pom-dex arms, respectively (Fig. 1). Patients received a median of 6 cycles in both the ixa-dex (range 1–25) and pom-dex (range 1–27) arms, with 7% and 15%, respectively, completing ≥19 cycles (Table 3). Mean relative dose intensities were numerically lower with ixa-dex vs pom-dex. Overall, 64% of patients receiving ixa-dex escalated to a 5.5 mg dose of ixazomib (Table 3).
Safety profiles are summarized in Table 3. In total, 69% vs 81% of patients had Grade ≥3 TEAEs with ixa-dex vs pom-dex, 51% vs 53% had serious TEAEs, 39% vs 36% had a TEAE leading to drug discontinuation, 44% vs 32% had a TEAE leading to dose reduction, and 13% of patients in each arm died while on study.
The most common any-grade and Grade ≥3 TEAEs with ixa-dex and pom-dex are summarized in Table 4. TEAEs occurring with a higher incidence (≥10% rate difference) with ixa-dex vs pom-dex were diarrhea (40% vs 28%), PN (29% vs 6%), insomnia (22% vs 11%), and peripheral edema (14% vs 4%). TEAEs occurring with a higher incidence (≥10% rate difference) with pom-dex vs ixa-dex were neutropenia (45% vs 3%), anemia (38% vs 18%), pruritus (11% vs 0), asthenia (17% vs 6%), and pyrexia (15% vs 4%). No Grade ≥3 TEAEs occurred at a higher incidence (≥10% rate difference) in the ixa-dex arm than the pom-dex arm. Grade ≥3 TEAEs occurring at a ≥ 10% higher rate with pom-dex vs ixa-dex were anemia (15% vs 30%) and neutropenia (3% vs 45%). The most common serious TEAE in both arms was pneumonia, reported in 9 (13%) ixa-dex vs 10 (21%) pom-dex patients. TEAEs that resulted in study drug discontinuation in ≥5% of patients were thrombocytopenia in 5 patients (7%) and PN in 4 patients (6%) in the ixa-dex arm vs pneumonia in 4 patients (9%) and neutropenia in 3 patients (6%) in the pom-dex arm. On-study deaths were not considered related to study treatment except for 1 patient in the ixa-dex arm who died due to a treatment-related AE (gastrointestinal disorder).
Patient-reported outcomes and healthcare resource utilization
HRQoL, as assessed by mean domain scores on the patient-reported EORTC QLQ-C30 and MY-20 instruments, was generally similar at baseline between the 2 arms and was maintained over time. On the EORTC QLQ-C30 instrument, functional subscale scores were generally high at baseline, indicating good HRQoL, whereas symptom scale scores for fatigue, pain, and nausea/vomiting were high or very high (Supplementary Table 4). Significant differences between treatment arms were seen at multiple time points for the cognitive functioning (Cycles 4, 8, 15, and 16), insomnia (Cycles 2, 9, 10, and 14), and diarrhea (Cycles 7, 8, 9, and 14) domains. For these 3 scales, mean changes from baseline in the ixa-dex arm were somewhat higher compared with those in the pom-dex arm, indicating better cognitive functioning but worse insomnia and diarrhea symptoms (Fig. 4). Mean baseline scores for the EORTC QLQ-MY20 subscales were high and similar between arms, and remained stable and similar over time (Supplementary Fig. 4).
Significant differences between arms were seen at multiple time points: (A) cognitive functioning, (B) insomnia, and (C) diarrhea in patients on the ixa-dex and pom-dex arms. C Cycle, D1 Day 1, dex dexamethasone, CI confidence interval, ixa ixazomib, LS least squares, pom pomalidomide. * Indicates P ≤ 0.05.
HRU during treatment is summarized in Supplementary Table 5. Mean rates of hospitalizations and emergency room stays were similar in the ixa-dex and pom-dex arms, whereas the rate of outpatient visits per patient-year exposure was higher in the pom-dex arm.
Discussion
This Phase 2 study demonstrated that PFS was comparable with ixa-dex and pom-dex in this population of heavily pretreated, carfilzomib and/or bortezomib-exposed and/or intolerant, lenalidomide-refractory patients (with medians of 7.1 and 4.8 months, respectively, and a HR of 0.847); there was no statistically significant difference between arms. These PFS data for ixa-dex appear generally consistent with median event-free survival findings of 7.8–11.5 months reported across dosing regimens in a previous Phase 2 study [35, 36], allowing for the higher rate of lenalidomide-refractory patients in the present study. Median PFS with pom-dex appeared similar or slightly higher than the 4.0- and 4.6-month medians seen in previous Phase 3/3b studies of pom-dex in refractory populations [39, 40], but slightly lower than the 6.9 months reported in the APOLLO Phase 3 study in less heavily pretreated patients [23].
Subgroup analyses of PFS showed numerically lower HRs in patients with ≥3 prior lines of therapy (HR 0.686) and in patients with creatinine clearance <60 mL/min (HR 0.467) compared to the ITT analysis (HR 0.847). The PFS HR appeared comparable in patients aged <75 (HR 0.843) and ≥75 (HR 0.890) years suggesting that, although there was an imbalance in the proportions of elderly patients between arms, this was unlikely to have materially impacted PFS findings in the ITT population.
Secondary efficacy endpoints appeared comparable between treatment arms, including ORR (38% vs 41%), TTR (median 2.0 vs 1.1 months), and DOR (median 14.8 vs 14.3 months). The TTP HR (0.830) was similar to that of PFS.
OS data were not mature at data cutoff, with only 36% of patients having died, and the study was not powered for OS comparisons. Nevertheless, preliminary outcomes exceeded expectations for this patient population in both arms, as previous studies report medians of 4–5 months and ~12–15 months for PFS and OS, respectively [39,40,41]. Differential OS findings were seen in patients aged <75 and ≥75 years. Given the advanced age of some patients, the overall OS findings may have been impacted and rendered inconclusive by the age imbalance between arms, including a higher rate of early censoring in the ixa-dex arm. The OS findings may also be partly explained by the differences in subsequent therapies received in each arm. More patients in the pom-dex arm received subsequent therapies, which may have been because they progressed sooner on study treatment, and were younger, and thus more likely to be able to receive subsequent therapies. Patients on the pom-dex arm also received subsequent daratumumab (28% vs 15%) and PIs (carfilzomib 28% vs 7%, bortezomib 13% vs 1%) more frequently than those on the ixa-dex arm who, by contrast, received pomalidomide (28% vs 4%) more frequently. Greater use of these alternative active regimens following treatment in this study may have contributed to the OS seen in the pom-dex arm, with a high proportion of patients censored at >12 months on the Kaplan–Meier analysis.
The data showed ixa-dex and pom-dex to be similarly tolerable. Notably, ixazomib was sufficiently well tolerated at the starting dose of 4 mg (based on the dose used in TOURMALINE-MM1 [26]) to enable 64% of patients to escalate to the 5.5 mg dose. This dose escalation of ixazomib was based on the previous Phase 2 study of ixa-dex, in which greater efficacy was seen with an ixazomib dose of 5.5 mg vs 4 mg (ORR 54% vs 31%), albeit with higher rates of TEAEs [35, 36]. In the current study, median duration of treatment with ixazomib 5.5 mg was 1.8 months (equating to ~2 cycles), suggesting that this was a challenging dose for patients and that subsequent dose reductions were required for continued treatment with ixazomib. Nonetheless, rates of discontinuations due to TEAEs were similar between the 2 arms. Thus, the oral PI-based doublet of ixa-dex appears tolerable in this treatment setting. While other approved PIs (bortezomib and carfilzomib) have demonstrated efficacy benefits in RRMM patients, these PIs require parenteral administration, a procedure with greater impact on these RRMM patients, who are less tolerant of visits to the hospital or clinic as their disease progresses [24].
There were no consistent differences between ixa-dex and pom-dex in terms of overall safety profile. Differential rates of specific TEAEs associated more commonly with ixazomib or pomalidomide were seen, such as diarrhea and thrombocytopenia with ixazomib, and anemia and pneumonia with pomalidomide [39, 40, 42]. Of note, the overall rate of PN, a TEAE of clinical importance with bortezomib-based therapy [43], was higher with ixa-dex vs pom-dex (29% vs 6%); however, this rate is consistent with that reported for ixazomib (4 mg) in combination with lenalidomide-dexamethasone in the Phase 3 TOURMALINE-MM1 trial (27%) [26], despite the higher ixazomib dose used in some patients in the current study. Overall rates of serious TEAEs in both arms were also consistent with rates of grade 3/4 treatment-related TEAEs in previous studies of ixa-dex [35, 36] and of serious TEAEs in previous studies of ixa-dex plus lenalidomide [26] and pom-dex [39, 40] in RRMM.
Rates of cardiovascular and renal toxicities, which can be associated with carfilzomib-based treatment [44], were notably low with ixa-dex suggesting that this PI-based doublet regimen may be preferred for patients at risk of those toxicities.
The tolerable safety profiles of ixa-dex and pom-dex were reflected in the similar patient-reported QoL and HRU data between arms. HRQoL scores were maintained from study entry in both arms and were generally similar throughout the study, indicating no relative adverse impact on HRQoL of one regimen vs the other.
This study had several limitations. The patient population was generally older than anticipated, and thus the age cutoff for stratification (65 years) was too low to balance the treatment arms. Consequently, there was an imbalance of 36% vs 18% in the proportions of patients aged ≥75 years in the ixa-dex vs pom-dex arms. Data on cytogenetic abnormalities were not collected routinely in this study, and so the impact of high-risk cytogenetics on outcomes could not be evaluated. However, additional stratification according to cytogenetic risk would not have been feasible due to the limited sample size, as stratification group sizes would have been too small due to the use of multiple stratification factors. Finally, as noted, OS data were not mature at data cutoff, and may have been impacted by imbalances in subsequent therapies, confounding interpretation of the findings.
In conclusion, these results suggest ixa-dex can be effective in heavily pretreated, lenalidomide-refractory, PI-exposed RRMM patients, a population with considerable unmet medical needs. Ixa-dex was tolerable, with a HRQoL comparable with pom-dex in this population. RRMM is a heterogeneous disease, and no single treatment benefits all patients. Ixa-dex is an appropriate option for patients who cannot tolerate triplet combinations, or who are not able or prefer not to receive parenteral treatment administration, or who require a non-immunomodulatory drug-based treatment approach.
Data availability
The datasets, including the redacted study protocol, redacted statistical analysis plan, and individual participants’ data supporting the results reported in this article will be available 3 months from initial request to researchers who provide a methodologically sound proposal. The data will be provided after its de-identification, in compliance with applicable privacy laws, data protection, and requirements for consent and anonymization.
References
Goldschmidt H, Ashcroft J, Szabo Z, Garderet L. Navigating the treatment landscape in multiple myeloma: which combinations to use and when? Ann Hematol. 2019;98:1–18.
Rajkumar SV, Kumar S. Multiple myeloma current treatment algorithms. Blood Cancer J. 2020;10:94.
Terpos E, Suzan F, Goldschmidt H. Going the distance: are we losing patients along the multiple myeloma treatment pathway? Crit Rev Oncol Hematol. 2018;126:19–23.
Kumar SK, Therneau TM, Gertz MA, Lacy MQ, Dispenzieri A, Rajkumar SV, et al. Clinical course of patients with relapsed multiple myeloma. Mayo Clin Proc. 2004;79:867–74.
Willenbacher E, Weger R, Rochau U, Siebert U, Willenbacher W, Austrian Myeloma R. Real-world use of 3rd line therapy for multiple myeloma in Austria: an Austrian Myeloma Registry (AMR) analysis of the therapeutic landscape and clinical outcomes prior to the use of next generation myeloma therapeutics. PLoS One. 2016;11:e0147381.
Kumar SK, Lee JH, Lahuerta JJ, Morgan G, Richardson PG, Crowley J, et al. Risk of progression and survival in multiple myeloma relapsing after therapy with IMiDs and bortezomib: a multicenter international myeloma working group study. Leukemia. 2012;26:149–57.
Jagannath S, Roy A, Kish J, Lunacsek O, Globe D, Eaddy M, et al. Real-world treatment patterns and associated progression-free survival in relapsed/refractory multiple myeloma among US community oncology practices. Expert Rev Hematol. 2016;9:707–17.
Venner CP, Bahlis NJ, Neri P, Sandhu I, Duggan P, Belch A, et al. In multiple myeloma progression free and overall survival in the relapsed setting remains poor with early exposure to novel agents: experience from a real-world cohort. Blood. 2015;126:4261–4261.
Kumar SK, Lee JH, Dimopoulos MA, Terpos E, Kastritis E, Chng WJ, et al. Outcomes after initial relapse of multiple myeloma: an international myeloma working group study. Blood. 2015;126:4201–4201.
Yong K, Delforge M, Driessen C, Fink L, Flinois A, Gonzalez-McQuire S, et al. Multiple myeloma: patient outcomes in real-world practice. Br J Haematol. 2016;175:252–64.
Barlogie B, Mitchell A, van Rhee F, Epstein J, Morgan GJ, Crowley J. Curing myeloma at last: defining criteria and providing the evidence. Blood. 2014;124:3043–51.
Kumar SK, Callander NS, Adekola K, Anderson L, Baljevic M, Campagnaro E, et al. Multiple Myeloma, Version 3.2021, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2020;18:1685–717.
van de Donk N. Sequencing multiple myeloma therapies with and after antibody therapies. Hematol Am Soc Hematol Educ Program. 2020;2020:248–58.
Dimopoulos MA, Moreau P, Terpos E, Mateos MV, Zweegman S, Cook G, et al. Multiple myeloma: EHA-ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up(dagger). Ann Oncol. 2021;32:309–22.
Moreau P, Kumar SK, San Miguel J, Davies F, Zamagni E, Bahlis N, et al. Treatment of relapsed and refractory multiple myeloma: recommendations from the International Myeloma Working Group. Lancet Oncol. 2021;22:e105–e118.
Dimopoulos M, Quach H, Mateos MV, Landgren O, Leleu X, Siegel D, et al. Carfilzomib, dexamethasone, and daratumumab versus carfilzomib and dexamethasone for patients with relapsed or refractory multiple myeloma (CANDOR): results from a randomised, multicentre, open-label, phase 3 study. Lancet. 2020;396:186–97.
Chari A, Suvannasankha A, Fay JW, Arnulf B, Kaufman JL, Ifthikharuddin JJ, et al. Daratumumab plus pomalidomide and dexamethasone in relapsed and/or refractory multiple myeloma. Blood. 2017;130:974–81.
Attal M, Richardson PG, Rajkumar SV, San-Miguel J, Beksac M, Spicka I, et al. Isatuximab plus pomalidomide and low-dose dexamethasone versus pomalidomide and low-dose dexamethasone in patients with relapsed and refractory multiple myeloma (ICARIA-MM): a randomised, multicentre, open-label, phase 3 study. Lancet. 2019;394:2096–107.
Palumbo A, Chanan-Khan A, Weisel K, Nooka AK, Masszi T, Beksac M, et al. Daratumumab, bortezomib, and dexamethasone for multiple myeloma. N Engl J Med. 2016;375:754–66.
Dimopoulos MA, Dytfeld D, Grosicki S, Moreau P, Takezako N, Hori M, et al. Elotuzumab plus pomalidomide and dexamethasone for multiple myeloma. N Engl J Med. 2018;379:1811–22.
Richardson PG, Oriol A, Beksac M, Liberati AM, Galli M, Schjesvold F, et al. Pomalidomide, bortezomib, and dexamethasone for patients with relapsed or refractory multiple myeloma previously treated with lenalidomide (OPTIMISMM): a randomised, open-label, phase 3 trial. Lancet Oncol. 2019;20:781–94.
Moreau P, Dimopoulos M-A, Mikhael J, Yong K, Capra M, Facon T, et al., editors. Isatuximab plus carfilzomib and dexamethasone vs carfilzomib and dexamethasone in relapsed/refractory multiple myeloma (IKEMA): Interim analysis of a phase 3, randomized, open-label study [abstract]. European Hematology Association; 2020 11-21 June; EHA, EHA25 Virtual.
Dimopoulos MA, Terpos E, Boccadoro M, Delimpasi S, Beksac M, Katodritou E, et al. Apollo: phase 3 randomized study of subcutaneous daratumumab plus pomalidomide and dexamethasone (D-Pd) versus pomalidomide and dexamethasone (Pd) alone in patients (Pts) with relapsed/refractory multiple myeloma (RRMM). Blood. 2020;136:5–6. Supplement 1
Chari A, Romanus D, DasMahapatra P, Hoole M, Lowe M, Curran C, et al. Patient-reported factors in treatment satisfaction in patients with relapsed/refractory multiple myeloma (RRMM). Oncologist. 2019;24:1479–87.
Snowden JA, Greenfield DM, Bird JM, Boland E, Bowcock S, Fisher A, et al. Guidelines for screening and management of late and long-term consequences of myeloma and its treatment. Br J Haematol. 2017;176:888–907.
Moreau P, Masszi T, Grzasko N, Bahlis NJ, Hansson M, Pour L, et al. Oral ixazomib, lenalidomide, and dexamethasone for multiple myeloma. N Engl J Med. 2016;374:1621–34.
Dimopoulos MA, Lonial S, White D, Moreau P, Palumbo A, San-Miguel J, et al. Elotuzumab plus lenalidomide/dexamethasone for relapsed or refractory multiple myeloma: ELOQUENT-2 follow-up and post-hoc analyses on progression-free survival and tumour growth. Br J Haematol. 2017;178:896–905.
Dimopoulos MA, Oriol A, Nahi H, San-Miguel J, Bahlis NJ, Usmani SZ, et al. Daratumumab, lenalidomide, and dexamethasone for multiple myeloma. N Engl J Med. 2016;375:1319–31.
Stewart AK, Rajkumar SV, Dimopoulos MA, Masszi T, Spicka I, Oriol A, et al. Carfilzomib, lenalidomide, and dexamethasone for relapsed multiple myeloma. N Engl J Med. 2015;372:142–52.
Hanaizi Z, Flores B, Hemmings R, Camarero J, Sancho-Lopez A, Salmonson T, et al. The European medicines agency review of pomalidomide in combination with low-dose dexamethasone for the treatment of adult patients with multiple myeloma: summary of the scientific assessment of the committee for medicinal products for human use. Oncologist. 2015;20:329–34.
Dimopoulos MA, Chen C, Spencer A, Niesvizky R, Attal M, Stadtmauer EA, et al. Long-term follow-up on overall survival from the MM-009 and MM-010 phase III trials of lenalidomide plus dexamethasone in patients with relapsed or refractory multiple myeloma. Leukemia. 2009;23:2147–52.
Celgene Corporation. POMALYST® (pomalidomide) capsules, for oral use. United States prescribing information. 2017.
Millennium Pharmaceuticals Inc. NINLARO® (ixazomib) capsules, for oral use. United States Prescribing Information. 2016.
Takeda Pharma A/S. NINLARO: European Product Assessment Report (EPAR) - Product Information. Annex I - Summary of Product Characteristics. 2017.
Kumar SK, LaPlant B, Roy V, Reeder CB, Lacy MQ, Gertz MA, et al. Phase 2 trial of ixazomib in patients with relapsed multiple myeloma not refractory to bortezomib. Blood Cancer J. 2015;5:e338.
Kumar SK, LaPlant BR, Reeder CB, Roy V, Halvorson AE, Buadi F, et al. Randomized phase 2 trial of ixazomib and dexamethasone in relapsed multiple myeloma not refractory to bortezomib. Blood. 2016;128:2415–22.
Rajkumar SV, Dimopoulos MA, Palumbo A, Blade J, Merlini G, Mateos MV, et al. International Myeloma Working Group updated criteria for the diagnosis of multiple myeloma. Lancet Oncol. 2014;15:e538–548.
Rajkumar SV, Harousseau JL, Durie B, Anderson KC, Dimopoulos M, Kyle R, et al. Consensus recommendations for the uniform reporting of clinical trials: report of the International Myeloma Workshop Consensus Panel 1. Blood. 2011;117:4691–5.
Miguel JS, Weisel K, Moreau P, Lacy M, Song K, Delforge M, et al. Pomalidomide plus low-dose dexamethasone versus high-dose dexamethasone alone for patients with relapsed and refractory multiple myeloma (MM-003): a randomised, open-label, phase 3 trial. Lancet Oncol. 2013;14:1055–66.
Dimopoulos MA, Palumbo A, Corradini P, Cavo M, Delforge M, Di Raimondo F, et al. Safety and efficacy of pomalidomide plus low-dose dexamethasone in STRATUS (MM-010): a phase 3b study in refractory multiple myeloma. Blood. 2016;128:497–503.
Kumar SK, Dimopoulos MA, Kastritis E, Terpos E, Nahi H, Goldschmidt H, et al. Natural history of relapsed myeloma, refractory to immunomodulatory drugs and proteasome inhibitors: a multicenter IMWG study. Leukemia. 2017;31:2443–8.
Kumar S, Moreau P, Hari P, Mateos MV, Ludwig H, Shustik C, et al. Management of adverse events associated with ixazomib plus lenalidomide/dexamethasone in relapsed/refractory multiple myeloma. Br J Haematol. 2017;178:571–82.
Richardson PG, Delforge M, Beksac M, Wen P, Jongen JL, Sezer O, et al. Management of treatment-emergent peripheral neuropathy in multiple myeloma. Leukemia. 2012;26:595–608.
Dimopoulos MA, Roussou M, Gavriatopoulou M, Psimenou E, Ziogas D, Eleutherakis-Papaiakovou E, et al. Cardiac and renal complications of carfilzomib in patients with multiple myeloma. Blood Adv. 2017;1:449–54.
Acknowledgements
This study was supported by Millennium Pharmaceuticals, Inc., a wholly owned subsidiary of Takeda Pharmaceutical Company Limited. We thank the patients and their families, as well as the physicians, nurses, study coordinators, and research staff for participation in the trial; Renda Ferrari, PhD, and Thomas G. Hedberg, PhD, of Takeda Development Center Americas, Inc. (TDCA) for editorial assistance. Medical writing support for the development of this manuscript, under the direction of the authors, was provided by Philippa Lloyd, BSc, and Steve Hill, PhD, of Ashfield MedComms, an Ashfield Health company, and funded by Takeda Development Center Americas, Inc. (TDCA), in compliance with Good Publication Practice 3 ethical guidelines (Battisti et al, Ann Intern Med 2015;163:461–4).
Funding
This study was sponsored by Millennium Pharmaceuticals, Inc., a wholly owned subsidiary of Takeda Pharmaceutical Company Limited.
Author information
Authors and Affiliations
Contributions
Study design: MAD, SK, RL. Study investigator: MAD, FS, XL, KR, OV, CL, RF, YGM, SK, HQ, VD, AP, HS. Enrolled patients: MAD, FS, XL, KR, M-DL, CL, OV, RF, YGM, SK, UHM, HQ, VD, AP, HS, HD. Data analysis: MAD, XL, JD, MD, OV, SK, RL, AV. Data interpretation: MAD, FS, XL, KR, M-DL, JD, MD, CL, RF, SK, RL, HQ, AV, HS. Manuscript preparation: All authors. Manuscript review and revisions: All authors. Final approval of manuscript: All author.
Corresponding author
Ethics declarations
Competing interests
MAD reports receiving honoraria from Amgen, Takeda, BMS, Janssen, and Beigene. FS reports receiving honoraria from Amgen, Takeda, and Sanofi; consulting/advisory roles for Celgene, Janssen, Oncopeptides, and Sanofi; and research funding from Celgene, Sanofi, Janssen, Oncopeptides, and GSK. VD reports receiving honoraria from Takeda, Sanofi, and Janssen; consulting/advisory roles for Celgene, Janssen, and Sanofi; and research funding from Celgene, Sanofi, Janssen, Agios Pharmaceuticals Inc, and Pfizer. OV reports receiving honoraria from Takeda, Sanofi, Janssen, and BMS; consulting/advisory roles for Celgene, Janssen, Sanofi, and BMS; and research funding from Celgene, Janssen, Sanofi, and BMS. HQ reports consulting/advisory roles for and receiving honoraria from GSK, Sanofi, Janssen Cilag, BMS, Celgene, CSL, Amgen, Takeda, Karyopharm, and Antengene; and research funding from GSK, Amgen, Celgene, Sanofi, and Karyopharm. XL reports receiving honoraria from Janssen, Takeda, Sanofi, Bas, Novartis, Amgen, Oncopeptide, Karyopharm, Incyte, and GSK. YGM reports no conflicts of interest. KR reports consulting/advisory roles for Celgene/BMS, Takeda, Janssen, Amgen, AbbVie, Sanofi, Oncopeptides, Karyopharm, GSK, Adaptive Biotech, and Pfizer; has served on speakers bureaus for Celgene/BMS, and Takeda; has received research funding from Celgene/BMS, Takeda, Janssen, and Amgen; and travel, accommodation and expenses from Celgene/BMS, Takeda, and Janssen. AP has received honoraria and travel, accommodation and expenses from Janssen, Celgene, Takeda, and Amgen. M-DL reports consulting/advisory roles for and receipt of travel, accommodation, and expenses from Takeda, AbbVie, Janssen, Roche, and Amgen. CL reports a consulting/advisory role for Janssen and research funding from BMS. UHM reports receiving honoraria from BMS, Janssen, Sandoz, Takeda, Amgen, and Sanofi; and consulting/advisory roles for Amgen and Oncopeptides. RF reports consulting/advisory roles for and has received honoraria and travel, accommodation, and expenses from BMS/Celgene, GSK, Amgen, and Janssen. HD reports a consulting/advisory role for Novartis, and has received honoraria from Amgen and Novartis, and travel expenses from Janssen. HS has received honoraria from Takeda, Novartis, and Janssen; research funding from Celgene; and travel, accommodation and expenses from Celgene and Takeda. AV, RL, JD and MD are employees of Takeda. SK reports consulting/advisory roles (with no personal payment) for AbbVie, Amgen, BMS, Janssen, Roche-Genentech, Takeda, KITE, and Astra-Zeneca; consulting/advisory roles (with personal payment) for Oncopeptides, Beigene, and Antagene; and has received institutional research funding from AbbVie, Amgen, BMS, Carsgen, Janssen, KITE, Merck, Astra-Zeneca, Novartis, Roche-Genentech, Takeda, and Tenebio.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Dimopoulos, M.A., Schjesvold, F., Doronin, V. et al. Oral ixazomib-dexamethasone vs oral pomalidomide-dexamethasone for lenalidomide-refractory, proteasome inhibitor-exposed multiple myeloma: a randomized Phase 2 trial. Blood Cancer J. 12, 9 (2022). https://doi.org/10.1038/s41408-021-00593-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41408-021-00593-2
- Springer Nature Limited