Abstract
Background
Phthalates exposure might affect children’s intelligence development. This study aimed to determine (1) whether sex and age affect cognitive function and (2) whether sex differences in cognitive performance are wider with higher phthalate concentrations.
Methods
Data were collected from PubMed (1998–2022), PROQUEST (1997–2022), and SpringerLink (1995–2022). The study followed the PRISMA process. The included articles were followed by PECO framework. The GRADE applied to assess the certainty of evidence. Of 2422 articles obtained, nine were selected using inclusion criteria. The random-effects model was used to estimate the pooled effects.
Results
Our meta-regression indicated a significant difference between sex differences with age at phthalate concentration assessment (β = −0.25; 95% CI = −0.47, −0.03) and MEHP concentration (β = −0.20; 95% CI = −0.37, −0.03).
Conclusions
The limitation of the current article is it only provides information on intelligence level rather than other aspects of cognitive function. Thus, the sequelae of phthalate exposure on attention and executive function are still unclear. Our analysis shows significant difference between sex differences in cognitive function scores associated with age at phthalate concentration assessment. Girls might be more resilient in cognitive function at a younger age or during lower concentrations of phthalates metabolites.
Impact
-
This is the first meta-analysis to evaluate the pooled estimates of sex differences in objective cognitive functions among children with phthalate exposure.
-
The female might be a protective factor when exposed to toxic plasticizers while the concentration is low.
-
This study captures the possible role of sex in cognitive functioning and plasticizer exposure through a meta-analysis of children’s sex, cognitive scores, and plasticizer exposure.
Similar content being viewed by others
Introduction
Phthalates are common endocrine-disrupting chemicals in our daily lives, found in plastics ranging from bottles to flooring. After entering the body, most phthalates are excreted in urine and feces within 24 h.1 Nevertheless, repeated exposure to high levels of phthalates during pregnancy, breastfeeding, and infancy may impair neurodevelopment,2 including intelligence. Intelligence broadly refers to an individual’s ability to combine logical reasoning, comprehension, expression, language, and learning, profoundly affecting lifelong learning.3
As both prenatal and postnatal exposure to phthalates affects the development of cognitive function, an increasing number of epidemiological studies have assessed the potential association; however, the conclusions are varied because of several methodological discrepancies.4,5 The three main factors causing discrepancies include diverse types of phthalates, different indicators of cognition level (e.g., varying from using the Behavioral Assessment System for Children, Second Edition [BASC-2] to measure behavioral and emotional functioning to using the Bayley Scales of Infant and Toddler Development Version II [BSID-II] to measure mental and psychomotor developmental functions4), and potential biological differences in subjects (e.g., age and sex). Therefore, until a recent meta-analysis was published by Radke et al. in 2020,6 most review articles were unable to conduct a meta-analysis to quantify the effect of phthalate exposure on cognition.4,7,8 This meta-analysis used five to eight articles to assess the associations between five phthalates (i.e., DEHP, DBP, DIBP, BBP, and DEP) and two types of cognitive function in children (i.e., infant mental and psychomotor development).6 While most results revealed slight and non-significant inverse associations, there was a moderate and significant inverse association between BBP and psychomotor development in girls,6 indicating that sex may moderate the extent to which phthalates affect cognition.9
Before 3 years of age, girls usually have an earlier developmental timetable than boys, so sex differences in cognition level may exist.10 Jankowska reviewed the role of sex in neurodevelopment on plasticizer exposure and found that phthalates are essential factors determining children’s cognitive, psychomotor, behavioral, and emotional development.8 However, their findings had many confounding variables; the outcomes they included were merged with subjective and objective measures, and the cognitive function results were contaminated. In addition, they had no conclusions about cognitive functions after plasticizer exposure. It is still unclear whether sex differences in cognition levels persist when children reach a certain age.
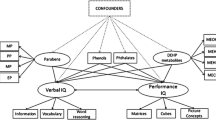
Standard intelligence tests used to assess research and clinical measures could be differentiated by age, below and above the age of three. The most common measurement of intelligence for children below 3 years of age is the BSID versions I, II, and III.11 The most common measurement for children above 3 years of age is the Wechsler Intelligence Scale (WIS),12,13 but some studies used the Intelligence and Development Scales (IDS) as an alternative.14 Each measure compares the norms for each age and sex group, and we should consider whether these differences vary with phthalate exposure. Therefore, this meta-analysis investigated (1) whether sex and age affect cognitive performance and (2) whether sex differences in cognitive performance are wider with higher phthalate concentrations.
Methods
We collected data from three databases: PubMed (1998–2022), PROQUEST (1997–2022), and SpringerLink (1995–2022). We used EndNoteX9 for importation and articles management. Restrictions were not put on the publication date or language of the publications, and the last search was conducted on December 23, 2022. The search keywords were categorized into three categories, each of which contained all three components: (1) phthalates; (2) child, preschool, infant, or teenager; and (3) cognitive function or intelligence.
This meta-analysis was conducted based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) process,15 reported in Supplementary Table 1. We followed the population, exposure, comparator, and outcome (PECO) framework to review and included articles in the meta-analysis. First, the population was defined as children aged 0–16 years. Second, the exposure and comparator were defined as male children (hereafter referred to as boys) vs. female children (hereafter referred to as girls) in different age groups and high vs. low phthalate exposure. Third, the outcome was lower cognitive performance, measured using a standardized intelligence score. The intelligence score currently measured using relevant ability scales is based on country and age norms. The standard deviation score was 15. We excluded articles in the first screening stage that were (1) non-human clinical trials, (2) non-child articles, (3) non-English language articles, (4) without cognitive assessment, and (5) not including the plasticizer metabolites data. In the final screening stages, we excluded the articles that (1) missing data, (2) overlapping population comprised duplicate data (i.e., different articles using the same data source; if this was the case, the article was selected for analysis with complete data), (3) cognitive assessment raw data was not provided. Two independent reviewers screened the relevant articles. Each article was evaluated for suitability for inclusion in the PRISMA process, as well as a keyword search of the article. If a discrepancy occurred, a third reviewer participated in the discussion until a consensus was reached.
We used Microsoft Excel for data curation and Comprehensive Meta-Analysis 3.0 and Stata version 16.1 (StataCorp, TX) for statistical analysis. Data records and descriptive statistics, such as participant distribution, number of participants, data sources, and measurement of cognitive function tools, were recorded using Microsoft Excel. A comprehensive meta-analysis was used to compare sex and cognitive function differences across the study and draw forest plots. Meta-regression was used to evaluate sex differences in cognitive function as a factor affecting phthalates metabolite content. Mean differences between the sexes were weighted by the standard deviation of the mean and the number of participants. Funnel plots and Egger’s tests were used to evaluate potential publication bias.16
We used the National Toxicology Program Office of Health Assessment and Translation (NTP/OHAT) approach for evidence integration to assess the risk of bias and GRADE to grade the certainty of evidence.17 We used I-squared (I2) and tau-squared (τ2) to investigate heterogeneity across selected studies between-study variance in the random-effects meta-analysis,16 respectively. I2 ranged from 0–25%, 25–50%, 50–75%, and >75%, representing no heterogeneity, low heterogeneity, moderate heterogeneity, and high heterogeneity, respectively.18
Results
Study selection and characteristics
Figure 1 shows the selected articles following the PRISMA process. The number of included articles in the initial stage was 2422. After removing 183 duplicate articles and eight book references, the number became 2231. In the next stage, 146 articles were included after applying the inclusion and exclusion criteria mentioned above. In the final screening stage, 31 articles lacked complete data, two had the same data, 50 did not provide complete cognitive assessment data, and five overlapping populations were excluded. Nine articles met the inclusion criteria. Eight of the 97 articles included in the review showed inconsistency in results between two independent reviewers, which were resolved through arbitration by a third reviewer. The inter-rater reliability of the selected articles was 0.92.
These nine articles included twelve study groups: one each from Poland, Sweden, Italy, Denmark, and Slovakia; two each from South Korea, and the United States; and three each from Taiwan. Participants were recruited from hospitals, environmental health centers, obstetrics and gynecology clinics, and child psychiatric clinics. Concerning cognitive instruments, three articles used Bayley, and nine used WIS. Regarding the timing of phthalate concentration assessment, five articles (five study groups) measured prenatal samples (mother’s urine), and four articles (seven study groups) measured postrenal samples (children’s urine). The characteristics of the nine articles included in the final analysis are summarized in Table 1.
Meta-analysis: sex comparison for cognitive function
The significant difference in cognitive scores between the sexes was not detected (weighted mean difference = 0.87; 95% CI = −0.24, 1.98; p = 0.20; I2 = 35.66%) (Fig. 2). Additionally, we performed subgroup analyses by study period, region, the timing of phthalate concentration assessment, and intelligence assessment tools (Table 2; Fig. 3a–d).
a By study period, b by region, c by the timing of phthalate concentration assessment, and d by intelligence assessment tools. BSID-II Bayley Scales of Infant and Toddler Development Version II, H2 h-squared, IDS Intelligence and Development Scales, I2 I-squared, p p-value, KEDI-WISC Korean Educational Development Institute-Wechsler Intelligence Scale for Children, τ2 tau-squared, WIS Wechsler Intelligence Scale.
Meta-regression and sex role in cognitive functions
Figure 4 reveals bubble plots of sex difference in cognitive function scores associated with age at phthalate concentration assessment (Fig. 4a; \(\beta \) = −0.25; 95% CI = −0.47, −0.03; p = 0.03), and MEHP concentration (Fig. 4b; \(\beta\) = −0.20; 95% CI = −0.37, −0.03; p = 0.02). Supplementary Fig. 1A–I shows insignificant results on MEOHP (\(\beta\) = −0.01; 95% CI = −0.22, 0.21; p = 0.96), MEHHP (\(\beta\) = −0.00; 95% CI = −0.11, 0.13; p = 0.97), ∑DEHP (\(\beta\) = 0.02; 95% CI = −0.03, 0.07; p = 0.37), MBzP (\(\beta \) = 0.00; 95% CI = −0.06, 0.06; p = 0.96), MiBP (\(\beta\) = −0.01; 95% CI = −0.05, 0.03; p = 0.50), MnBP (\(\beta \) = 0.01; 95% CI = −0.07, 0.10; p = 0.74), MBP (\(\beta\) = 0.03; 95% CI = −0.14, 0.21; p = 0.71), MEP (\(\beta\) = −0.00; 95% CI = −0.03, 0.02; p = 0.85), and age at intelligence assessment (\(\beta\) = −0.14; 95% CI = −0.48, 0.19; p = 0.40).
Further correlation analysis revealed that MEHP concentration was also associated with age at phthalate concentration assessment (both prenatal and postnatal; r = 0.71; p < 0.05; see Supplementary Fig. 2A) and age at intelligence assessment (r = 0.40; p < 0.05; see Supplementary Fig. 2B). However, the correlation coefficient between “MEHP concentration” and “age at phthalate concentration assessment (both prenatal and postnatal)” was higher. This finding suggests that the difference was more significant at younger ages, probably due to the lower concentration of age at the phthalate concentration assessment. In addition, both correlations showed that the higher the age, the higher the plasticizer concentration measured. Our findings suggest that the protective effect of age may only exist at young ages or low concentrations of plasticizers.
Publication bias
A symmetric inverted funnel shape indicated a low risk of publication bias among these nine articles with twelve study groups, as shown in Fig. 5 (\(\beta\) = −0.47; 95% CI = 0.59, 1.49; p = 0.66).
Risk of bias
The risk of bias assessments for studies investigating the association between plasticizer exposure and intelligence function are summarized in Table 3, and the details of each assessment are provided in Supplementary Table 2. The NTP/OHAT Risk of Bias tool focuses on confounding and detection biases, including exposure and outcome assessment. We assessed the risk of bias among the nine articles. For confounding bias, confounders such as parents’ smoking habits and education, household income, and lower socioeconomic status were not adjusted throughout the four articles. Thus, three articles were rated as having a “probably high risk of bias.”19,20 For exposure detection bias, none of the articles was rated as “probably low risk of bias.” However, for outcome detection bias, two articles were rated as “probably high risk of bias,” which provided insufficient information to justify the assessment.21 For exclusion bias, five articles that did not specify exclusion bias were rated as “probably high risk of bias.”20,21,22,23 Four articles that failed to report conflicts of interest or funding support were rated as “probably high risk.”20,21,22,23 For selective reporting bias, article was rated as “probably low risk of bias” due to a lack of significant and consistent evidence of inverse associations between three groups of cohort study exposures and FSIQ. In summary, articles were categorized as Tier 1 (n = 5) or Tier 2 (n = 4) for risk of bias, indicating a “plausible bias that raises some doubt about the results.”17
Certainty of evidence
A summary of the findings regarding the certainty of the evidence is provided in Table 4. Regarding the risk of bias rating for studies investigating plasticizers, the overall risk of bias was “not serious.” The inconsistency of the certainty assessment indicated low heterogeneity in the meta-analyses of the studies (I2 = 35.66%). However, the explanation for this heterogeneity could reveal some inconsistencies, such as differences in study design, population age, prenatal or postnatal exposure, and statistical methods. The respective category of the inconsistency of certainty assessment was “serious.” The indirectness and imprecision certainty assessment indicated a category of “not serious” because the outcomes of our study demonstrated that the evidence answers directly to population cognitive functions living near plasticizers, and the CI of the pooled analysis was narrow (−0.24 to 1.98). Funnel plots (Fig. 5) exhibited symmetrical patterns, and weighted mean difference did not yield evidence of publication bias, indicating an undetectable publication bias. In summary, the inherent risk of bias made our rating for the certainty of evidence from low to very low for the association between sex differences in cognitive function and plasticizer exposure.
Discussion
To the best of our knowledge, this is the first meta-analysis to evaluate the pooled estimates of sex differences in objective cognitive functions among children with phthalate exposure. We analyzed the data of twelve study groups from Poland, Sweden, Italy, Denmark, Slovakia, South Korea, Taiwan, and the United States. There was no significant difference in cognitive scores between sexes. However, our meta-regression of the weighted mean difference shows a significant difference between sex differences in cognitive function scores and age at phthalate concentration assessment. In other word, the younger the age of the child, the greater cognitive and difference in phthalate concentration between sexes. Another meta-regression of the weighted mean difference shows significant between sex differences in cognitive function scores and MEHP concentration. Specifically, the timing of phthalate concentration assessment and MEHP concentration from 6 reports were significantly associated with sex differences by a factor of 0.2 (\(\beta\) = −0.20; 95% CI = −0.37, −0.03; p = 0.02). The estimated sex differences in intelligence scores were higher among children with prenatal phthalate exposure and lower MEHP concentrations. Suggesting that when the concentration of MEHP is lower, sex plays as a protecting factor, making the difference in average IQ between males and females larger. Also, the difference in intelligence decreased by 0.02–0.03 points if the timing of phthalate measurement started 1 year later, and MEHP concentration (geometric mean) increased 1 (µg/L).
We observed a higher score for cognitive function in girls than in boys when phthalate exposure was at a younger age than at an older age. This finding suggests that girls have greater physical resilience when exposed to phthalates at a young age.24 A previous systematic review has shown that being female would act as a protective factor that could modulate the effects of substance residues on a child’s cognitive function, including bisphenol A and plasticizers.9 The reason prenatal or early exposure to phthalates had more adverse effects on neurodevelopment in males than in females might be that plasticizers could disrupt the development of androgen-dependent structures by inhibiting fetal testicular testosterone biosynthesis.25 Past studies further showed that in animal or human research, substance exposure would affect male cognitive function and not females and even cause behavioral problems.22,26,27
However, does phthalate do more harm to neurodevelopment in males than in females? Research on cognitive and intellectual development has discussed sex differences in the early stages of life; for example, girls had better intelligence than boys when they were 2 to 7 years old, and it was reported that girls had better processing speed from ages 4 to 7.28 Additionally, male toddlers were observed to be less tractable and manageable than girls, which could cause more injury or less parenting at early ages.29 However, as children grow up, these differences would dismiss.30,31 We found that female works as physical resilience at a younger early age.
Our correlation results showed a strong relationship between age (age at intelligence assessment and age at phthalate concentration assessment) and the concentration of phthalates metabolites; as the child grows up, more phthalates metabolites are detected. This phenomenon indicates that phthalates metabolites would affect the child strongly when exposed to more plastic products in their daily lives.
Limitations
This study has several strengths. First, cognitive function was measured by a professional psychologist following a standardization process to ensure objectivity. Moreover, the cognitive scores of the included articles proved that both boys and girls had normative cognitive scores (the mean cognitive score of norms was 100). The study objectively answers the review published by ref. 8 The included articles encompass different continents, such as Asia, North America, and Europe, to increase holistically.
However, our study had some limitations. First, the included articles measured intelligence levels. There was a lack of assessing other aspects of cognitive function, such as executive function and attention. Although recent intelligence measurements claimed that they could measure executive function or attention,32 these neuropsychological functions did not fully report in plasticizer exposure articles. These cognitive functions involve different concepts and do not always have similar outcomes.33 Thus, the sequelae of phthalate exposure on attention and executive function are still unclear. Future studies should include a comparison group (control group) and different objective cognitive function measurements. Additionally, phthalates were measured at different times (e.g., environmental measurements only and maternal or child subjects). Jankowska et al. reported different results in a prenatal and postnatal sampling study.20 It shows that prenatal (third trimester) and postnatal (at 2 years of age) measurements indicated that the prenatal phthalate metabolites were lower than those measured postnatally. Further studies have shown that different sequelae range between prenatal and postnatal exposures.20,34 Last and all, only observational studies were included in the analysis; we could only use the norm’s mean and standard deviation to check if the intelligence score of studies showed would be within or without the norm’s range. Nor were the studies comparing the intelligence score with their country/area demographic similar norms. In addition, a comparison of the exposure sample with the control sample is lacking. We suggest future studies should include a control group to examine the sequelae of exposure, even with artificial intelligence techniques, to find the critical neurodevelopmental disorder after exposure to a plasticizer.35
Conclusion
We suggest that girls had resilient cognitive function in cognitive scores and lower concentrations of phthalate metabolites compared with the boys at the same exposure level. Therefore, the female might be a protective factor when exposed to toxic plasticizers while the concentration is low. Moreover, when the child grows up, more phthalate metabolites are detected, suggesting that phthalate metabolites might affect the child strongly as they are exposed to more plastic products in their daily lives.
Data availability
Data from the primary studies included in this meta-analysis are available upon request from the corresponding author. The data consist of were collected from PubMed, PROQUEST, and SpringerLink between 1995–2022. Interested researchers must obtain permission from the original study authors and adhere to any applicable ethical guidelines. Researchers must also agree to cite the original studies and seek permission before using the data for any purpose other than that of the current meta-analysis.
References
Frederiksen, H., Skakkebaek, N. E. & Andersson, A. M. Metabolism of phthalates in humans. Mol. Nutr. Food Res. 51, 899–911 (2007).
Braun, J. M. Early-life exposure to EDCs: role in childhood obesity and neurodevelopment. Nat. Rev. Endocrinol. 13, 161–173 (2017).
Hunt, J. M. Intelligence and Experience (Ronald), (1961).
Ejaredar, M., Nyanza, E. C., Ten Eycke, K. & Dewey, D. Phthalate exposure and childrens neurodevelopment: a systematic review. Environ. Res. 142, 51–60 (2015).
Martínez-Martínez, M. I., Alegre-Martínez, A. & Cauli, O. Prenatal exposure to phthalates and its effects upon cognitive and motor functions: a systematic review. Toxicology 463, 152980 (2021).
Radke, E. G., Braun, J. M., Nachman, R. M. & Cooper, G. S. Phthalate exposure and neurodevelopment: a systematic review and meta-analysis of human epidemiological evidence. Environ. Int. 137, 105408 (2020).
Zhang, Q., Chen, X. Z., Huang, X., Wang, M. & Wu, J. The association between prenatal exposure to phthalates and cognition and neurobehavior of children-evidence from birth cohorts. Neurotoxicology 73, 199–212 (2019).
Jankowska, A., Nazareth, L., Kaleta, D. & Polanska, K. Review of the existing evidence for sex-specific relationships between prenatal phthalate exposure and children’s neurodevelopment. Int. J. Environ. Res. Public Health 18, 13013 (2021).
Palanza, P. et al. Sex-biased impact of endocrine disrupting chemicals on behavioral development and vulnerability to disease: of mice and children. Neurosci. Biobehav. Rev. 121, 29–46 (2021).
Halpern, D. F. Sex Differences in Cognitive Abilities (Psychology press, 2000).
Lowe, J. R., Erickson, S. J., Schrader, R. & Duncan, A. F. Comparison of the Bayley Ii Mental Developmental Index and the Bayley Iii cognitive scale: are we measuring the same thing? Acta Paediatr. 101, e55–e58 (2012).
Wechsler, D. Wechsler Intelligence Scale for Children 4th edn (Wisc-Iv, 2003).
Wechsler, D. Wechsler Preschool and Primary Scale of Intelligence—Fourth Edition. The Psychological Corporation San Antonio, TX (2012).
Grob, A., Meyer, C. & Hagmann-von Arx, P. Intelligence and Development Scales (Ids) (Hans Huber, 2009).
Page, M. J. et al. The prisma 2020 statement: an updated guideline for reporting systematic reviews. BMJ 372, n71 (2021).
Higgins, J P. T. (eds). Cochrane Handbook for Systematic Reviews of Interventions version 6.3 (updated February 2022) (Cochrane). Retrieved on July 14, 2022 from https://training.cochrane.org/handbook (2022).
Office of Health Assessment and Translation(OHAT) Division of the National Toxicology Program. Handbook for Conducting a Literature-Based Health Assessment Using Ohat Approach for Systematic Review and Evidence Integration (National Institute of Environmental Health Sciences). Retrieved on July 14, 2022 from https://ntp.niehs.nih.gov/ntp/ohat/pubs/handbookmarch2019_508.pdf (2019).
Higgins, J. P., Thompson, S. G., Deeks, J. J. & Altman, D. G. Measuring inconsistency in meta-analyses. BMJ 327, 557–560 (2003).
Tsai, C. S. et al. Phthalates, para-hydroxybenzoic acids, bisphenol-A, and gonadal hormones’ effects on susceptibility to attention-deficit/hyperactivity disorder. Toxics 8, 57 (2020).
Jankowska, A. et al. Prenatal and early postnatal phthalate exposure and child neurodevelopment at age of 7 years - polish mother and child cohort. Environ. Res. 177, 108626 (2019).
Cho, S. C. et al. Relationship between environmental phthalate exposure and the intelligence of school-age children. Environ. Health Perspect. 118, 1027–1032 (2010).
Kim, Y. et al. Prenatal exposure to phthalates and infant development at 6 months: prospective mothers and children’s environmental health (Moceh) study. Environ. Health Perspect. 119, 1495–1500 (2011).
Whyatt, R. M. et al. Maternal prenatal urinary phthalate metabolite concentrations and child mental, psychomotor, and behavioral development at 3 years of age. Environ. Health Perspect. 120, 290–295 (2012).
Bruneau, M. & Reinhorn, A. Proc. 8th US National Conference on Earthquake Engineering. 18–22.
Yen, T. H., Lin-Tan, D. T. & Lin, J. L. Food safety involving ingestion of foods and beverages prepared with phthalate-plasticizer-containing clouding agents. J. Formos. Med. Assoc. 110, 671–684 (2011).
Dai, Y., Yang, Y., Xu, X. & Hu, Y. Effects of uterine and lactational exposure to Di-(2-Ethylhexyl) phthalate on spatial memory and NMDA receptor of hippocampus in mice. Horm. Behav. 71, 41–48 (2015).
Lai, T.-J. et al. A cohort study of behavioral problems and intelligence in children with high prenatal polychlorinated biphenyl exposure. Arch. Gen. Psychiatry 59, 1061–1066 (2002).
Palejwala, M. H. & Fine, J. G. Gender differences in latent cognitive abilities in children aged 2 to 7. Intelligence 48, 96–108 (2015).
Matheny, A. P. Jr Injuries among toddlers: contributions from child, mother, and family1. J. Pediatr. Psychol. 11, 163–176 (1986).
Reynolds, M. R., Hajovsky, D. B. & Caemmerer, J. M. The sexes do not differ in general intelligence, but they do in some specifics. Intelligence 92, 101651 (2022).
Saggino, A. et al. Null sex differences in general intelligence among elderly. Pers. Individ Differ. 63, 53–57 (2014).
Montoya-Arenas, D. A., Aguirre-Acevedo, D. C., Díaz Soto, C. M. & Pineda Salazar, D. A. Executive functions and high intellectual capacity in school-age: completely overlap?. Int J. Psychol. Res. 11, 19–32 (2018).
Ardila, A., Pineda, D. & Rosselli, M. Correlation between intelligence test scores and executive function measures. Arch. Clin. Neuropsychol. 15, 31–36 (2000).
Polańska, K., Ligocka, D., Sobala, W. & Hanke, W. Effect of environmental phthalate exposure on pregnancy duration and birth outcomes. Int J. Occup. Med. Environ. Health 29, 683–697 (2016).
Allahyari, E. & Roustaei, N. Applying artificial neural-network model to predict psychiatric symptoms. BioMedicine 12, 1 (2022).
Funding
This work was supported by China Medical University, Taiwan (grant numbers: CMU111-ASIA-10 and CMU110-ASIA-13) and the National Science and Technology Council, Taiwan (grant number: 111-2314-B-039-020-MY2). The funding source had no role in the study design, data collection, data analysis, data interpretation, writing of the manuscript, or decision to submit the paper for publication. We thank the English-language editors at Editage for their careful copyediting.
Author information
Authors and Affiliations
Contributions
Y.L. conceptualized and designed the study, coordinated and supervised data collection, investigated the study, drafted the initial manuscript, and reviewed and revised the manuscript; Y.X. investigated the study, curated data, drafted the initial manuscript, and reviewed and revised the manuscript; J.C. investigated the study, curated data, drafted the initial manuscript; H.B. carried out the initial analyses and formal analysis, curated data, reviewed and revised the manuscript, and visualized the data; B.S. and Y.L. critically reviewed the manuscript; R.L. conceptualized and designed the study, coordinated and supervised data collection, carried out the initial analyses, drafted the initial manuscript, reviewed and revised the manuscript, project administration, and funding acquisition. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Liao, YC., Xu, YJ., Chen, JK. et al. Sex differences in children’s cognitive functions and phthalates exposure: a meta-analysis. Pediatr Res 94, 1609–1618 (2023). https://doi.org/10.1038/s41390-023-02672-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-023-02672-5
- Springer Nature America, Inc.
This article is cited by
-
Chemical toxicity by sex differences: a literature review of 2019–2023 studies
Toxicology and Environmental Health Sciences (2024)