Abstract
Background
The impact of long-term serum uric acid (SUA) exposure and time course of SUA accumulation on diabetes mellitus (DM) is unknown. This study aimed to evaluate the association of cumulative SUA (cumSUA) exposure and its accumulation time course with risk of DM.
Methods
This prospective study included 46,434 participants without DM and underwent three examinations at 2006, 2008, and 2010. CumSUA from 2006 to 2010 was calculated, multiplying mean values between consecutive examinations by time intervals between visits. Time course of SUA accumulation was categorized as the slope of SUA versus time from 2006 to 2010, or by splitting the overall accumulation into an early (cumSUA06-08) and late accumulation (cumSUA08-10).
Results
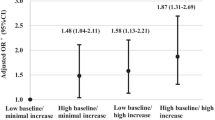
During 6.99 years of follow-up, we identified 2971 incident DM cases. In the fully adjusted model, a higher risk of DM was observed in participants with the highest quartile of cumSUA (hazard ratio [HR], 1.31; 95% confidence interval [CI], 1.17–1.46), cumulative burden >0 (HR, 1.23; 95% CI, 1.08–1.40), and with 6 year of hyperuricemia exposure duration (HR, 1.25; 95% CI, 1.01–1.55). When considering the time course of SUA accumulation, participants with a negative slope (HR, 1.05; 95% CI, 1.01–1.12), or combined with cumSUA ≥ median and a negative slope had elevated risk of DM (HR, 1.58; 95% CI, 1.18–2.11).
Conclusions
Incident DM risk depends on cumulative exposure of SUA and time course of SUA accumulation. Early SUA accumulation resulted in a greater risk increase compared with later accumulation, emphasizing the importance of optimal SUA control early in life.
Similar content being viewed by others
Introduction
Diabetes mellitus (DM) is a worldwide public health problem [1]. In China, the prevalence of total DM was 12.8% and the healthcare cost associated with diabetes is 110 billion international dollars (purchasing power parity) in 2017 [2]. DM is also a well-established risk factor for cardiovascular disease, renal dysfunction, dementia, and all-cause mortality [3,4,5]. Primary prevention strategies including early detection and control risk factors are necessary to reduce the impeding incidence of DM and its related complications.
Serum uric acid (SUA), the end product of purine metabolism by xanthine oxidoreductase, is correlated with many recognized cardiovascular risk factors. Animal experiments [6] and few intervention studies [7] in human have shown that reducing SUA might improve insulin resistance, which raises great interest in the relation between SUA and DM. In recent decades, a number of prospective observational studies reported a positive association between SUA levels and incident DM risk [8,9,10,11]. In contrast, some indicated a negative association [12] or supported bystander role of SUA in the development of DM [13,14,15]. The discrepancy may be attributable to that the majority of these published studies only analyzed the single measurement of SUA levels, failing to take the potential effect of long-term cumulative exposure of SUA into account, which might bias the true relation between SUA and DM risk. Considering the methodological limitations, several recent studies have investigate the effect of change in hyperuricemia status on DM risk [15, 16], however, changes in SUA in the aforementioned study were defined on the basis of 2 measurements of SUA levels and were unable to account for the longitudinal patterns of SUA over a period. On this consideration, the method of cumulative metrics has been proposed, which can capture both the duration and intensity of SUA over several years [17,18,19,20]. Furthermore, incorporation both the SUA levels and exposure duration into a single risk parameter for future DM is intuitively appealing, although a data-based demonstration of the utility of this metric is insufficient. Additionally, it is also unclear whether the time course of SUA accumulation plays an important role in modulating the risk conferred by a give overall exposure.
Therefore, we conducted the present study to (1) quantify the association of cumulative exposure to SUA (cumSUA) with DM risk; (2) evaluate the impact of cumulative burden of SUA; (3) hyperuricemia exposure duration; (4) and the modulated effect of time course of SUA accumulation on DM risk based on a large community-based prospective cohort study.
Subjects and methods
Study design and population
The Kailuan study is an ongoing prospective cohort study conducted in Tangshan City, China. The study was designed to investigate the risk factors for common noncommunicable diseases and the detailed study design and characteristics of the study population have been described previously [21, 22]. In brief, a total of 101,510 participants (aged 18–98 years, 81,110 men and 20,400 women) completed the first survey from June 2006 to October 2007 and underwent a comprehensive biennial health examination thereafter. In the current, we calculated cumSUA using SUA levels in 2006, 2008, and 2010 to predict incident DM after 2010, the time line of the study is presented in Fig. S1. We excluded 44,677 participants who did not have three times health examinations, 8693 participants who were diagnosed with DM (the criteria for DM was described below) to minimize the possible effect of reverse causality, and 1706 participants with missing data on SUA. Consequently, 46,434 participants were included in the present analysis (Fig. S2). A comparison of included and excluded participant characteristics owing to missing data or follow-up is presented in Table S1. The study was performed according to the guidelines of the Helsinki Declaration and was approved by the Ethics Committee of Kailuan General Hospital and Beijing Tiantan Hospital. All participants provided written informed consent.
Calculation of cumSUA, cumulative burden, duration of hyperuricemia exposure, and time course of SUA accumulation
Blood samples were collected following an 8–12 h overnight fast at baseline and was then measured biennially. SUA concentrations were measured at the central laboratory in the Kailuan General Hospital. The inter-assay coefficient of variation for each measurement conducted using an auto analyzer (Hitachi 7600, Tokyo, Japan) with an oxidase method was ≤6.0%.
CumSUA was defined as the sum of products of average SUA in 2 consecutive follow-up examinations multiplied by the follow-up interval in years [17,18,19], detailed in Fig. 1, and the formula calculated as follows:
where SUA2006, SUA2008, and SUA2010 indicate SUA at the first (baseline), second, and third examinations, and time2006–2008 and time2008–2010 indicate the participant-specific time intervals between consecutive examinations in years [23]. The means of time2006–2008 and time2008–2010 were 2.10 and 1.95 years, respectively.
Average SUA between consecutive examinations as A1 and A2. Cumulative SUA was calculated as (A1 × time06–08 + A2 × time08–10), showed by the dotted area, μmol/L × year. SUA slope was obtained using a linear regression, SUA values at follow-up visits were used to calculate the SUA. SUA serum uric acid.
Cumulative burden is defined as the weighted sum of the portion of 2 adjacent, average measurements that falls above the cutoff value that is then multiplied by time intervals between consecutive examinations in years:
Hyperuricemia was defined as a SUA level ≥420 μmol/L for men and ≥360 μmol/L for women [24], which was the cutoff in the formula. If the values of the cumulative burdens between 2 consecutive examinations were less than 0 this value would be considered as 0 [25].
Hyperuricemia exposure duration was defined as the times of visits with hyperuricemia at visit 2006, 2008, and 2010. We give 1 point to hyperuricemia, 0 point for normouricemia at each visit, therefore, hyperuricemia exposure duration was ranged from 0 years (never had hyperuricemia), 2 years (had hyperuricemia once), 4 years (had hyperuricemia twice), and 6 years (had hyperuricemia at all three study visits) [26].
Time course of SUA accumulation was categorized in 2 ways: a slope of SUA versus time from 2006 to 2010 using the linear regression and the least-squares principle, with a positive or negative slope indicating an increase or decrease in SUA over time (illustrated in Fig. 1); or alternatively, the cumSUA between 2006 to 2008 (cumSUA06-08), and 2008–2010 (cumSUA08-10) were calculated as early and late SUA exposure measures, respectively [27].
Follow-up and assessment of DM
For each participant, follow-up started from 2010 and ended at the occurrence of DM, death, or the end of the follow-up on 31 December 2017, which event came first. DM was defined as either FBG ≥ 7.0 mmol/L, self-reported of a physician diagnosis, or self-reported use of antidiabetic medication.
Assessment of covariates
All individuals completed a standardized questionnaire that collected information on their demographic characteristics, lifestyle habits, and medical history. Active physical activity was defined as exercising more than four times a week and at least 20 min each time. Body mass index (BMI) was calculated by dividing weight (kilogram) by squared height (meter2). Blood pressure were measured two times with the participants in the seated position with a mercury sphygmomanometer according to the JCN7 recommendation. The average of two readings was used for the analyses.
The fasting blood samples were tested using a Hitachi 747 auto-analyzer (Hitachi, Tokyo, Japan) at the central laboratory of the Kailuan Hospital, including fasting blood glucose (FBG), serum lipid profiles, serum creatinine, and high-sensitivity C-reactive protein (hs-CRP). Estimated glomerular filtration rate (eGFR) was calculated using the creatinine-based Chronic Kidney Disease Epidemiological Collaboration equation [28].
Hypertension was defined as any self-reported hypertension or use of antihypertensive drug, or blood pressure ≥140/90 mmHg. Dyslipidemia was defined as any self-reported history or use of lipid-lowering drugs, or serum total cholesterol ≥5.17 mmol/L or triglyceride ≥1.69 mmol/L or low-density lipoprotein cholesterol ≥3.62 mmol/L or high-density lipoprotein cholesterol ≤1.04 mmol/L.
Statistical analysis
Continuous variables were described as mean ± standard deviation and differences across baseline groups were compared using Student’s t-test or ANOVA. Categorical variables were described as frequency with percentage and were compared using Chi-square test. Person-years was determined from the date when the message was collected to either the date of incident DM, death, or the date of participating in the last examination in the analysis, whichever came first.
Cox proportional hazard regression was applied to calculate he hazard ratio (HR) and 95% confidence interval (CI) for the risk of DM. According to Schoenfeld residuals and log-log inspection, our models met the proportional assumption criteria. Four models were constructed systematically, model 1 was unadjusted; model 2 was adjusted for age and sex; model 3 was further adjusted for education, income, smoking status, drinking status, history of hypertension and dyslipidemia, BMI, systolic blood pressure, diastolic blood pressure, and FBG; model 4 was further adjusted for antihypertensive agents, diuretics, lipid-lowering agents, eGFR, and hs-CRP. For each model, a trend test was performed after the median value of each quartile was entered into the model and treated as a continuous variable. The restricted cubic spline models were with 4 knots (at the 5th, 35th, 65th, and 95th percentiles) were conducted to test whether there was a dose-response or nonlinear association of cumSUA as a continuous variable with the risk of DM, adjusting for all the covariates above-mentioned. When considering the combined effect of SUA accumulation and its time course, participants were divided into 4 categories according to the median of cumSUA (1118.7 μmol/L × year) and the direction of slope (negative or positive). The association between combined effect of SUA accumulation and its time course and DM was also assessed using multivariable Cox proportional hazard models.
To test the robustness of our findings, we further conducted several sensitivity analyses. First, competing risk model was applied considering non-DM deaths as competing risk events. Second, to explore the potential impact of reverse causality, we excluded the outcome events that occurred within the first 2 years of the follow-up period. Third, considering diuretics may have a potential effect on SUA concentration, we excluded participants who used diuretics during the whole study period.
All analyses were conducted using SAS version 9.4 (SAS Institute Inc., Cary, NC, USA). A two-sided P < 0.05 was considered statistically significant.
Results
Baseline characteristics
The mean age of the enrolled participants was 48.66 ± 11.97 years, 75.75% were men.
Baseline characteristics according to the combined effect of cumSUA accumulation and its time course are presented in Table 1. Compared with participants with cumSUA < median, slope ≥ 0, participants in other groups were more likely to be older, men, educated, had higher income, more current smokers, current alcohol users, active physical activity, a higher prevalence of hypertension, dyslipidemia, more likely to take antihypertensive agents, lipid-lowering agents, a higher level of BMI, FBG, SBP, DBP, hs-CRP, and a lower level of eGFR. The characteristics of participants according to quartiles of cumSUA are presented in Table S2. There was also a significant difference among the four categories in all the baseline characteristics.
Association of cumSUA, cumulative burden, and hyperuricemia exposure duration with the risk of DM
During a median follow-up of 6.99 years, 2971 (6.40%) incident DM were detected. The associations of cumulative SUA parameters with the risk of DM are presented in Table 2 and Fig. 2. The incidence rate per 1000 person-years of DM increased across the cumSUA quartiles, ranging from 7.41 (95% CI, 6.83–8.03) in the lowest quartile to 11.50 (95% CI, 10.70–12.30) in the highest quartile group. The association remained significant even after adjustment for potential variables, the fully adjusted HR (Model 4) was 1.31 (95% CI, 1.17–1.46; P for trend < 0.0001) in the lowest quartile versus the highest quartile of cumSUA. Multivariable-adjusted spline regression models yielded the same pattern of results when cumSUA was treated as a continuous variable (HR for per 1 SD was 1.08 [95% CI, 1.04–1.12]; Fig. 3).
cumSUA, cumulative serum uric acid, SUA serum uric acid. The four categories of combination of cumSUA and SUA slope were as follows: <M, ≥0: cumSUA < Median, and slope ≥ 0; <M, <0: cumSUA < Median, and slope < 0; ≥M, ≥0: cumSUA ≥ Median, and slope ≥ 0; ≥M, <0: cumSUA ≥ Median, and slope < 0. Adjusted for age, sex, education, income, smoking status, drinking status, history of hypertension and dyslipidemia, antihypertensive agents, hypoglycemic agents, diuretics, lipid-lowering agents, body mass index, fasting blood glucose, systolic blood pressure, diastolic blood pressure, estimated glomerular filtration rate, and high-sensitivity C-reactive protein.
Hazard ratio (solid line) and 95% confidence interval (dash lines) from Cox regression using restricted cubic spline regression with 4 knots with placed at the 5th, 35th, 65th, 95th percentiles. Multivariate adjustment was for age, sex, education, income, smoking status, drinking status, history of hypertension and dyslipidemia, antihypertensive agents, hypoglycemic agents, diuretics, lipid-lowering agents, body mass index, fasting blood glucose, systolic blood pressure, diastolic blood pressure, estimated glomerular filtration rate, and high-sensitivity C-reactive protein.
Similar results were observed for cumulative burden of SUA and hyperuricemia exposure duration. For those with cumulative burden >0 compared with cumulative burden ≤0, the risk of DM increased by 123% (HR, 1.23; 95% CI, 1.08–1.40, P = 0.0022). When comparing the participants with the longest to the shortest hyperuricemia exposure duration, participants with a 6 year exposure duration of hyperuricemia had 1.25-fold higher risk of MI (HR, 1.25; 95% CI, 1.01–1.55, P for trend = 0.0014). The sensitivity analyses with competing risk model, excluding outcome within the first year of follow-up (n = 858), or excluding participants who used diuretics at baseline and during the follow-up (n = 1455) did not materially change the results (Table 3).
Time course of SUA accumulation and risk of DM
The associations of the time course of SUA accumulation evaluated by adding the slope of SUA time course over 2006–2010 to the analysis, or by splitting the overall accumulation into an early (cumSUA06-08) and late accumulation (cumSUA08-10) in the models are presented in Table 4. After adjusted for potential covariates, participants with negative slope of SUA time course (decreasing SUA trend) had elevated DM risk (HR, 1.05; 95% CI, 1.01–1.12; P = 0.0303), compared with participants with positive slope of SUA time course (increasing SUA trend). Consistently, the later accumulation was not associated with DM after adjusted for early accumulation in the multivariable model (P = 0.2161).
When considering the combined effect of cumSUA accumulation and its time course, in the fully adjusted model, participants with cumSUA ≥ median and a negative slope of SUA time course had the highest risk of DM (HR, 1.20; 95% CI, 1.07-1.34), compared with those with cumSUA< median and a positive slope of SUA time course (Table 2). Sensitivity analysis showed the similar results (Table 3).
Discussion
This study shows that the risk of future incident DM is associated with the total umulative exposure of SUA. Importantly, this risk is modulated by the time course of SUA accumulation. Notably, our data suggest that cumSUA accumulated early confers greater risk of DM than when the same cumSUA is accumulated later. These findings underscore the importance of optimal SUA early in life, because lower SUA later, even when low enough to result in the same accumulation at the same time point, does not fully reverse risk acquired earlier. However, it is important to recognize that these data do not in any way suggest that there is no primary prevention benefit to lowering SUA no matter when elevated SUA lowering is started. Our results indicate an apparent persistent increase in later DM risk conferred by high SUA levels experienced early in life.
The present analyses showed that participants with high cumSUA levels, or a higher cumulative burden of SUA over time had a higher risk of developing DM, relative to their counterparts with lower cumSUA over time. Most previous studies but not all, which were generally based on a single SUA assessment, generated consistent results regarding the association between SUA and DM risk. For example, data from the Framingham Heart Study original and offering cohorts showed that individuals with higher SUA are at a higher future risk of DM independent of other known risk factors [9]. The positive association was also supported by the Rotterdam Study [29], the National Health Interview Survey [10], the China Stroke Primary Prevention Trial [11], and some other cohort studies [30, 31]. Of note, a single measurement of SUA in these studies may not adequately reflect the longitudinal variation and cumulative burden associated with elevated SUA level. In contrast, the measurement of cumSUA could capture both the duration and intensity of SUA exposure over several decades. Our findings taken together with previous studies suggested that not only baseline SUA, but also a longitudinal cumSUA is important in predicting the risk of DM.
In addition, our study investigated the association between exposure duration of hyperuricemia and DM risk, the results showed that the risk of DM increased with hyperuricemia exposure duration. Hyperuricemia is a metabolic problem that has become increasing common worldwide and its association with DM was observed in meta-analysis, as well as in many epidemiological studies [32, 33]. However, the evidence on the effect of hyperuricemia duration on DM risk was scarce. Experimental studies revealed that hyperuricemia is associated with kidney damage via stimulating RAS activity and promoting endothelial damage along with oxidative stress [34], which are important contributors in the development of DM. Furthermore, hyperuricemia is a disease with stepwise progression [35], the damage in organisms is aggravating with the duration of hyperuricemia, thus leading to increasing DM risk with hyperuricemia duration. This finding highlights the potential benefit of early control and treatment of hyperuricemia for the prevention of DM.
Another interest implications of our study, particularly when viewed in the context of other studies, is that the time course of SUA accumulation was taken into account. The results showed individuals with the same cumulative exposure to SUA but with a greater fraction of that exposure occurring earlier in life had a greater risk of incident DM risk. That means the same cumSUA exposure accumulated earlier is associated with higher risk of DM compared with later in life. These data underscore the dependence of risk, not just on the present amount of cumSUA level, but also the SUA versus time history, and offer a model to quantify the modulation of risk by the time course of SUA. Future clinical trials of lowering SUA early should be conducted to investigate whether there is a major reduction in DM incidence compared to risk reduction started later.
There are several biological mechanisms underlying the relationship of cumSUA and its time course with DM. Several in vitro and in vivo studies showed that high SUA levels can induce oxidative stress, which has been established as a pathological pathway for the development of DM [36]. First, high cumSUA produces oxidative stress via pathways involving the activation of nicotinamide adenine dinucleotide phosphate oxidase and the generation of oxidized lipids and inflammatory mediators [6]. Second, high cumSUA can regulate enzymes associated with glucose and lipid metabolism primarily in the liver, adipose tissue, and skeletal muscle [37]. Through positive feedback, adipose tissue could produce and secrete additional SUA via xanthine oxidoreductase [38]. Third, elevated cumSUA would decrease insulin sensitivity and lead to insulin resistance through altering glucose metabolism [39]. High cumSUA can induce endothelial dysfunction and nitric oxide inhibition, which in turn contribute to insulin resistance and thus diabetes [40]. This is supported by findings that fructose-induced hyperuricemia in rats leads to insulin resistance along with other components of metabolic syndrome, and these conditions are improved by decreasing uric acid levels [6].
The strengths of our prospective study is that when assessing the association between cumSUA and DM, we considered the modulated effect of time course of cumSUA accumulation, which may provide additional information. However, our study still has several limitations. First, the use of urate-lowering agents was not available. Given the low rate of hyperuricemia therapy and the controversial benefit of pharmacological treatment for asymptomatic hyperuricemia, our conclusion would not meaningfully change. Second, due to the large sample, our cohort did not take oral glucose tolerance test and measure hemoglobin A1c to diagnose DM. Considering the same diagnosis criteria being used in all the participants which will lead to nondifferentiated misclassification, the association of cumSUA and its time course with DM would be underestimated. We therefore excluded participants diagnosed with diabetes less than 2 years to avoid misclassification at baseline and increase specificity of diabetes diagnosis. Finally, our population is from a region in North China; therefore, the finding may not be generalized.
Conclusions
In conclusion, incident DM risk depends on both long-term cumulative exposure to SUA, and importantly, on the time course of SUA accumulation. The same cumulative exposure acquired earlier in life, compared with later, leading to a greater risk increase. The findings stress the importance of optimal SUA control starting early in life for preventing or reducing incident DM.
Data availability
Data are available to researchers on request for purposes of reproducing the results or replicating the procedure by directly contacting the corresponding author.
Code availability
Code is available to researchers on request for purposes of reproducing the results or replicating the procedure by directly contacting the corresponding author.
References
Cho NH, Shaw JE, Karuranga S, Huang Y, da Rocha Fernandes JD, Ohlrogge AW, et al. IDF Diabetes Atlas: Global estimates of diabetes prevalence for 2017 and projections for 2045. Diabetes Res Clin Pract. 2018;138:271–81.
Li Y, Teng D, Shi X, Qin G, Qin Y, Quan H, et al. Prevalence of diabetes recorded in mainland China using 2018 diagnostic criteria from the American Diabetes Association: national cross sectional study. BMJ. 2020;369:m997.
Emerging Risk Factors C, Sarwar N, Gao P, Seshasai SR, Gobin R, Kaptoge S, et al. Diabetes mellitus, fasting blood glucose concentration, and risk of vascular disease: a collaborative meta-analysis of 102 prospective studies. Lancet. 2010;375:2215–22.
Rao Kondapally Seshasai S, Kaptoge S, Thompson A, Di Angelantonio E, Gao P, Sarwar N, et al. Diabetes mellitus, fasting glucose, and risk of cause-specific death. N Engl J Med. 2011;364:829–41.
Kielstein JT. Glucose levels and risk of dementia. N Engl J Med. 2013;369:1863.
Baldwin W, McRae S, Marek G, Wymer D, Pannu V, Baylis C, et al. Hyperuricemia as a mediator of the proinflammatory endocrine imbalance in the adipose tissue in a murine model of the metabolic syndrome. Diabetes. 2011;60:1258–69.
Takir M, Kostek O, Ozkok A, Elcioglu OC, Bakan A, Erek A, et al. Lowering uric acid with allopurinol improves insulin resistance and systemic inflammation in asymptomatic hyperuricemia. J Invest Med. 2015;63:924–9.
Dehghan A, van Hoek M, Sijbrands E, Hofman A, Witteman J. High serum uric acid as a novel risk factor for type 2 diabetes. Diabetes Care. 2008;31:361–2.
Bhole V, Choi J, Kim S, de Vera M, Choi H. Serum uric acid levels and the risk of type 2 diabetes: a prospective study. Am J Med. 2010;123:957–61.
Wu W, et al. Serum uric acid level as a harbinger of type 2 diabetes: a prospective observation in Taiwan. Int J Environ Res Public Health. 2020;17:2277
Zhou C, Liu M, Zhang Z, Zhang Y, Nie J, Liang M, et al. Positive association of serum uric acid with new-onset diabetes in Chinese women with hypertension in a retrospective analysis of the China Stroke Primary Prevention Trial. Diabetes Obes Metab. 2020;22:1598–606.
Taniguchi Y, Hayashi T, Tsumura K, Endo G, Fujii S, Okada K. Serum uric acid and the risk for hypertension and Type 2 diabetes in Japanese men: The Osaka Health Survey. J Hypertens. 2001;19:1209–15.
Tsouli S, Liberopoulos E, Mikhailidis D, Athyros V, Elisaf M. Elevated serum uric acid levels in metabolic syndrome: an active component or an innocent bystander? Metab Clin Exp. 2006;55:1293–301.
Keerman M, et al. Mendelian randomization study of serum uric acid levels and diabetes risk: evidence from the Dongfeng-Tongji cohort. BMJ Open Diabetes Res Care. 2020;8:e000834
Volpe A, Ye C, Hanley AJ, Connelly PW, Zinman B, Retnakaran R. Changes over time in uric acid in relation to changes in insulin sensitivity, beta-cell function, and glycemia. J Clin Endocrinol Metab. 2020;105:e651–9.
Lou Y, Qin P, Wang C, Ma J, Peng X, Xu S, et al. Sex-specific association of serum uric acid level and change in hyperuricemia status with risk of type 2 diabetes mellitus: a large cohort study in China. J Diabetes Res. 2020;2020:9637365.
Zemaitis P, Liu K, Jacobs DR Jr, Cushman M, Durazo-Arvizu R, Shoham D, et al. Cumulative systolic BP and changes in urine albumin-to-creatinine ratios in nondiabetic participants of the multi-ethnic study of atherosclerosis. Clin J Am Soc Nephrol. 2014;9:1922–9.
Jenkins LM, Garner CR, Kurian S, Higgins JP, Parrish TB, Sedaghat S, et al. Cumulative blood pressure exposure, basal ganglia, and thalamic morphology in midlife. Hypertension. 2020;75:1289–95.
Yano Y, Ning H, Allen N, Reis JP, Launer LJ, Liu K, et al. Long-term blood pressure variability throughout young adulthood and cognitive function in midlife: the Coronary Artery Risk Development in Young Adults (CARDIA) study. Hypertension. 2014;64:983–8.
Mo J, Chen Z, Xu J, Wang A, Meng X, Zhao X, et al. The impact of the cumulative burden of LDL-c and hs-CRP on cardiovascular risk: a prospective, population-based study. Aging. 2020;12:11990–2001.
Jin C, Chen S, Vaidya A, Wu Y, Wu Z, Hu FB, et al. Longitudinal change in fasting blood glucose and myocardial infarction risk in a population without diabetes. Diabetes Care. 2017;40:1565–72.
Wang C, Yuan Y, Zheng M, Pan A, Wang M, Zhao M, et al. Association of age of onset of hypertension with cardiovascular diseases and mortality. J Am Coll Cardiol. 2020;75:2921–30.
Tian X, Wang A, Wu S, Zuo Y, Chen S, Zhang L, et al. Cumulative serum uric acid and its time course are associated with risk of myocardial infarction and all-cause mortality. J Am Heart Assoc. 2021;10:e020180.
Iseki K, Ikemiya Y, Inoue T, Iseki C, Kinjo K, Takishita S. Significance of hyperuricemia as a risk factor for developing ESRD in a screened cohort. Am J Kidney Dis. 2004;44:642–50.
Kang K, Wang Y, Wu J, Wang A, Zhang J, Xu J, et al. Association between cumulative exposure to increased low-density lipoprotein cholesterol and the prevalence of asymptomatic intracranial atherosclerotic stenosis. Front Neurol. 2020;11:555274.
Zhao Q, Li H, Wang A, Guo J, Yu J, Luo Y, et al. Cumulative resting heart rate exposure and risk of all-cause mortality: results from the Kailuan Cohort Study. Sci Rep. 2017;7:40212.
Domanski MJ, Tian X, Wu CO, Reis JP, Dey AK, Gu Y, et al. Time course of LDL cholesterol exposure and cardiovascular disease event risk. J Am Coll Cardiol. 2020;76:1507–16.
Pottel H, Delanaye P, Schaeffner E, Dubourg L, Eriksen BO, Melsom T, et al. Estimating glomerular filtration rate for the full age spectrum from serum creatinine and cystatin C. Nephrol Dial Transpl. 2017;32:497–507.
van der Schaft N, Brahimaj A, Wen KX, Franco OH, Dehghan A. The association between serum uric acid and the incidence of prediabetes and type 2 diabetes mellitus: The Rotterdam Study. PLoS ONE. 2017;12:e0179482.
Kim SC, Liu J, Solomon DH. Risk of incident diabetes in patients with gout: a cohort study. Arthritis Rheumatol. 2015;67:273–80.
Liu Y, Jin C, Xing A, Liu X, Chen S, Li D, et al. Serum uric acid levels and the risk of impaired fasting glucose: a prospective study in adults of north China. PLoS ONE. 2013;8:e84712.
Lv Q, Meng XF, He FF, Chen S, Su H, Xiong J, et al. High serum uric acid and increased risk of type 2 diabetes: a systemic review and meta-analysis of prospective cohort studies. PLoS ONE. 2013;8:e56864.
Mortada I. Hyperuricemia, type 2 diabetes mellitus, and hypertension: an emerging association. Curr Hypertens Rep. 2017;19:69.
Jitraknatee J, Ruengorn C, Nochaiwong S. Prevalence and risk factors of chronic kidney disease among type 2 diabetes patients: a cross-sectional study in primary care practice. Sci Rep. 2020;10:6205.
Zhou Y, Zhao M, Pu Z, Xu G, Li X. Relationship between oxidative stress and inflammation in hyperuricemia: Analysis based on asymptomatic young patients with primary hyperuricemia. Medicine (Baltimore). 2018;97:e13108.
Zhang Y, Yamamoto T, Hisatome I, Li Y, Cheng W, Sun N, et al. Uric acid induces oxidative stress and growth inhibition by activating adenosine monophosphate-activated protein kinase and extracellular signal-regulated kinase signal pathways in pancreatic β cells. Mol Cell Endocrinol. 2013;375:89–96.
Lima WG, Martins-Santos ME, Chaves VE. Uric acid as a modulator of glucose and lipid metabolism. Biochimie. 2015;116:17–23.
Tsushima Y, Nishizawa H, Tochino Y, Nakatsuji H, Sekimoto R, Nagao H, et al. Uric acid secretion from adipose tissue and its increase in obesity. J Biol Chem. 2013;288:27138–49.
Fabbrini E, Serafini M, Colic Baric I, Hazen SL, Klein S. Effect of plasma uric acid on antioxidant capacity, oxidative stress, and insulin sensitivity in obese subjects. Diabetes. 2014;63:976–81.
Krishnan E, Pandya BJ, Chung L, Hariri A, Dabbous O. Hyperuricemia in young adults and risk of insulin resistance, prediabetes, and diabetes: a 15-year follow-up study. Am J Epidemiol. 2012;176:108–16.
Acknowledgements
We thank all study participants, their relatives, the members of the survey teams at the 11 regional hospitals of the Kailuan Medical Group; and the project development and management teams at the Beijing Tiantan Hospital and the Kailuan Group.
Funding
This work was supported by National Key Research and Development Program of China (2018YFC1312400 and 2018YFC1312402), Beijing Municipal Administration of Hospitals Incubating Program (PX2020021), Beijing Excellent Talents Training Program (2018000021469G234), Young Elite Scientists Sponsorship Program by CAST (2018QNRC001), and National Natural Science Foundation of China (81773512).
Author information
Authors and Affiliations
Contributions
XT and AW wrote the manuscript. XT, AW, and YZ researched data. SC, LZ, YZ, and LL researched data and contributed to discussion. SW, YL, and JG contributed to the discussion and reviewed/edited the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
The study was performed according to the guidelines of the Helsinki Declaration and was approved by the Ethics Committee of Kailuan General Hospital (approval number: 2006-05) and Beijing Tiantan Hospital (approval number: 2010-014-01). All participants were agreed to take part in the study and provided informed written consent.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tian, X., Wang, A., Zuo, Y. et al. Time course of serum uric acid accumulation and the risk of diabetes mellitus. Nutr. Diabetes 12, 1 (2022). https://doi.org/10.1038/s41387-021-00179-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41387-021-00179-8
- Springer Nature Limited
This article is cited by
-
Association between cumulative atherogenic index of plasma exposure and risk of myocardial infarction in the general population
Cardiovascular Diabetology (2023)
-
Association of the time course of Chinese visceral adiposity index accumulation with cardiovascular events in patients with hypertension
Lipids in Health and Disease (2023)
-
Time course of the triglyceride glucose index accumulation with the risk of cardiovascular disease and all-cause mortality
Cardiovascular Diabetology (2022)