Abstract
Mood and anxiety disorders typically begin in adolescence and have overlapping clinical features but marked inter-individual variation in clinical presentation. The use of multimodal neuroimaging data may offer novel insights into the underlying brain mechanisms. We applied Heterogeneity Through Discriminative Analysis (HYDRA) to measures of regional brain morphometry, neurite density, and intracortical myelination to identify subtypes of youth, aged 9–10 years, with mood and anxiety disorders (N = 1931) compared to typically developing youth (N = 2823). We identified three subtypes that were robust to permutation testing and sample composition. Subtype 1 evidenced a pattern of imbalanced cortical-subcortical maturation compared to the typically developing group, with subcortical regions lagging behind prefrontal cortical thinning and myelination and greater cortical surface expansion globally. Subtype 2 displayed a pattern of delayed cortical maturation indicated by higher cortical thickness and lower cortical surface area expansion and myelination compared to the typically developing group. Subtype 3 showed evidence of atypical brain maturation involving globally lower cortical thickness and surface coupled with higher myelination and neural density. Subtype 1 had superior cognitive function in contrast to the other two subtypes that underperformed compared to the typically developing group. Higher levels of parental psychopathology, family conflict, and social adversity were common to all subtypes, with subtype 3 having the highest burden of adverse exposures. These analyses comprehensively characterize pre-adolescent mood and anxiety disorders, the biopsychosocial context in which they arise, and lay the foundation for the examination of the longitudinal evolution of the subtypes identified as the study sample transitions through adolescence.
Similar content being viewed by others
Introduction
Mood and anxiety disorders collectively account for over 65% of the disease burden attributable to mental disorders in individuals aged 10–24 years [1]. Most of the disability is accounted for by the early onset of these disorders [2] and their persistence into adulthood [3, 4]. There is international consensus on the need to prioritize youth mental health initiatives aiming at prevention and early intervention [5,6,7] which importantly acknowledges the major challenge posed by the incomplete understanding of the underlying neurobiology [8].
The diagnostic boundaries of mood (i.e., major depression, bipolar disorders) and anxiety disorders, as currently defined [9, 10] are highly permeable due to overlapping psychopathology; negative affective states in particular are both shared and central clinical features of mood and anxiety disorders [11] including bipolar disorder where depressive symptoms are the dominant psychopathology [12]. The clinical overlap between mood and anxiety disorders is even more pronounced in youth as depressive and anxiety symptoms typically predate the onset of syndromal diagnoses [13, 14]. Similarly, research into the neurobiological correlates of mood and anxiety disorders has failed to produce disorder-specific biomarkers but has identified instead transdiagnostic neuroimaging and genetic features [15,16,17].
In response, research efforts have shifted towards new approaches to classification that use data-driven methods to identify subgroups of individuals with mood and anxiety disorders based on their shared biological properties. This approach has yielded promising findings in adults [17,18,19] and has recently begun to be applied in youth. Kaczkurkin and colleagues (2020) [20] identified two neuroanatomical subtypes in youth (aged 8–23 years) with internalising disorders broadly corresponding to an “impaired” and a “spared” group. A similar spared-impaired pattern was reported by Fan and colleagues (2021) [21] who used data-driven methods to distinguish 9–10-year-olds with internalizing disorders into two subgroups with either high or low impulsivity and then contrasted their neuroanatomical profiles.
The present study leverages the resources from the baseline assessment wave of the Adolescent Brain Cognitive Development (ABCD) Study (https://abcdstudy.org/) which acquires comprehensive phenotypic information and high-quality multimodal neuroimaging data from the largest currently available sample of 9–10-year-olds at study entry [22]. Our working hypothesis is that participants with mood and anxiety disorders in this cross-sectional sample can be partitioned into data-driven biologically informative subtypes based on their neuroimaging features. The study design enriches prior literature in three distinct ways. First, we expanded the phenotypic spectrum to encompass the entire range of mood and anxiety disorders, including bipolar disorder. Bipolar disorder is typically excluded from the construct of “internalizing” disorders despite compelling evidence for the dominance of internalizing symptoms, which is even more pronounced in young people [14]. Second, we expanded the neuroimaging phenotypic space to include measures of neurite density [23] and myelination [24, 25] that could yield insights into the brain maturational processes that may differentiate potential subtypes. Third, both psychopathology and brain organization are influenced by the environment in which individuals live. Prior research has emphasized the importance of perinatal history, parental socioeconomic status and psychopathology, family and neighborhood environment [26,27,28]; measures pertaining to these domains were tested to identify potentially distinct associations with the neuroimaging-based subtypes. These analyses comprehensively characterize pre-adolescent mood and anxiety disorders, the biopsychosocial context in which they arise, and lay the foundation for the investigation of the longitudinal evolution of the maturational and clinical trajectories of these subtypes in follow-up studies of this sample.
Methods
Sample
De-identified data were accessed from the 2.0 and 3.0 data releases (https://data-archive.nimh.nih.gov/abcd) of the ABCD study. Participants at baseline (N = 11878) were recruited using multi-stage probability sampling to ensure that the sample is nationally representative in terms of sex, race/ethnicity, socio-economic status, and urbanicity [29] (Supplementary Material S1). The coordinating center is the University of California, San Diego, which has oversight of the ethical approval of the ABCD protocols. At each site, parents provided written informed consent, and youth provided assent.
The current study sample comprises ABCD participants selected based on the completeness of diagnostic data from the Kiddie Schedule for Affective Disorders and Schizophrenia for School-Age Children (KSADS-5) [30, 31] and high-quality neuroimaging data across all modalities based on ABCD quality control criteria [32] (Supplementary Material S2, Supplementary Table S1). The flowchart of the selection process is presented in Supplementary Fig. S1.
Based on conventional psychiatric labels the study sample included the following five groups: (i) 2823 typically developing participants that were free of any lifetime psychopathology [age: 119.13 (7.50) months, 52.60% female]; (ii) 178 participants diagnosed only with a depressive disorder [age: 119.08 (7.08) months, 50.00% female]; (iii) 307 participants diagnosed only with a bipolar disorder [age: 118.65 (7.13) months, 51.79% female]; (iv) 1197 participants diagnosed only with an anxiety disorder [age: 118.97 (7.60) months, 55.89% female], and (v) 249 participants with comorbid mood and anxiety disorders [age: 119.00 (7.55) months, 54.62% female] (additional details in Supplementary Tables S2). Depressive disorders comprised major depressive disorder, dysthymia, or unspecified depressive disorder. Bipolar disorders comprised bipolar disorder I, bipolar disorder II, or unspecified bipolar disorder. Anxiety disorders comprised separation anxiety disorder, social anxiety disorder, generalized anxiety disorder, specific phobia, or other unspecified anxiety disorders. Further details of the diagnostic composition of the sample are provided in Supplementary Table S3.
Non-imaging measures
Participants’ internalizing and externalizing behaviors were assessed with the Child Behavior Checklist (CBCL) [33] and their crystallized and fluid intelligence were evaluated using the NIH Toolbox [34]. The PhenX ToolKit [35] and study-specific questionnaires [36,37,38] were used to record information about participants’ prenatal and obstetric history, parental characteristics (i.e., parental socioeconomic status and psychopathology), exposure to adverse life events, and quality of family, school and neighborhood environment (i.e., family function, quality of parental engagement, school performance and engagement and neighborhood deprivation). Definitions of these measures are provided in Supplementary Material S4 and Supplementary Table S4).
Neuroimaging
The protocols for data acquisition, processing, and neuroimaging feature extraction [32, 39] are outlined in Supplementary Material S5. Neuroimaging measures were extracted from whole-brain T1-weighted, and diffusion-weighted magnetic resonance imaging scans mapped Desikan-Killiany cortical parcellations [40] and subcortical regions mapped to the Fischl probabilistic atlas [41] and were accessed through the ABCD repository. These comprised (a) FreeSurfer derived measures of cortical thickness and surface area and regional subcortical volume; (b) Restriction Spectrum Imaging (RSI) [23] derived measures of neural density (ND); and (c) regional cortical myelination obtained through the gray/white matter contrast (GWC), which is the ratio of the signal intensity on either side of the surface formed at the gray-white matter cortical boundary [24, 25]. All neuroimaging measures were site harmonized using the ComBat algorithm (https://github.com/Jfortin1/ComBatHarmonization) [42] before further analyses.
Statistical analysis
Missing Data
Only participants with complete demographic, clinical, and neuroimaging data were included. Missingness for all other non-imaging measures was low (range 0.04%–8.50%) and missing values were imputed using multivariate imputation by chained equations (implemented in R 4.0.5 using the “mice” function) [43].
Semi-supervised machine learning for subtype identification
We employed Heterogeneity Through Discriminative Analysis (HYDRA), a nonlinear semi-supervised machine learning algorithm [44] to identify homogeneous clusters (i.e., subtypes) amongst pre-adolescents with mood and anxiety disorders. HYDRA, unlike other clustering methods, does not rely on similarity indices that are susceptible to the effects of non-specific factors such as age and sex but employs indices of deviation between a group of interest (here participants with mood and anxiety disorders) and a reference group (here typically developing participants) based on a combination of multiple hyperplanes (using a convex polytope) thus accommodating non-linearity (Supplementary Material S6; Supplementary Fig. S2).
HYDRA (https://github.com/evarol/HYDRA) was applied to the regional morphometric, GWC, and ND measures. The diagnostic labels of the clinical group were not included in the input features of HYDRA while age, sex, and race/ethnicity were included as covariates. We assessed solutions with 2–7 clusters and chose the optimal solution following a 5-fold cross-validation based on the Adjusted Rand Index (ARI) [45]. Statistical significance was determined through permutation at P < 0.05. First, we assessed whether the solution obtained in the main analyses differed from that generated following random grouping of participants over 100 permutations. Second, we assessed whether the solution obtained in the main analyses differed from that generated if the sample did not include a clinical group; this analysis also involved 100 permutations (details in Supplementary Material S6). The distinctive neuroimaging features of each cluster, compared to the typical youth, were identified in a series of post-hoc analyses with false discovery rate (FDR) [46] correction for multiple testing at PFDR < 0.005.
Comparison of personal, family, and social characteristics of the identified subtypes
After identifying clusters through HYDRA, we conducted post-hoc analyses comparing each cluster’s personal, family and social characteristics to those of the typically developing group using a series of mixed linear models implemented with the “lme4” package in R. Comparisons involved neuroimaging, personal, family, and social characteristics. The effects of age, sex, and race/ethnicity were regressed from all models and total intracranial volume (TIV) from models for cortical surface area, subcortical volume, and neurite density (Supplementary Figs. S3, S4). The threshold for statistical significance for these post-hoc analyses was set at PFDR < 0.005.
Sensitivity Analyses
We conducted two sensitivity analyses to test the robustness of the clustering solution to reproducibility and relatedness (details in Supplementary Material S7). We also tested the sensitivity of the results to alternate definitions of intracortical myelin by using the T1w/T2w [47] instead of GWC (details in Supplementary Material S7).
Results
HYDRA-Subtypes
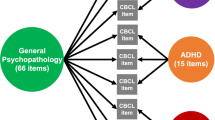
The 3-cluster HYDRA solution had the highest ARI and was robust to permutation testing (Supplementary Material S8; Supplementary Fig. S5) and sample composition (Supplementary Material S9; Supplementary Fig. S7). Each of these clusters (henceforth referred to as subtypes) comprised 631, 649, and 651 ABCD participants. The diagnostic composition of each subtype was generally proportional to the diagnostic distribution in the entire clinical sample (Fig. 1); subtype 3 however had relatively fewer participants with anxiety disorders and relatively more with bipolar disorders compared to subtype 1 (χ2 = 15.80, P = 0.001) and subtype 2 (χ2 = 10.12, P = 0.02).
The figure illustrates the composition of each cluster in terms of traditional clinical diagnoses. Chi-squared tests revealed that subtype 1 (N = 631) and subtype 2 (N = 649) had equal proportions of participants with mood and anxiety disorders (χ2 = 2.45, P = 0.48), and subtype 3 (N = 651) had more participants with bipolar disorders, and fewer participants with anxiety disorders relative to subtype 1 (χ2 = 15.80, P = 0.001) and subtype 2 (χ2 = 10.12, P = 0.02).
Clinical, Family, and Social Characteristics of the HYDRA-subtypes
Differences between each HYDRA subtype and the typically developing group that remained significant at PFDR < 0.005 are summarized in Table 1 and information about all comparisons (including effect size) are presented in Supplementary Tables S5, S6.
Obstetric and Perinatal events
Maternal use of alcohol, substances, and prescribed medications was higher in all subtypes while subtype 1 had higher birth weight.
Psychopathology and Cognition
All subtypes had higher CBCL scores for externalizing and internalizing psychopathology; subtype 1 had higher crystallized intelligence while subtype 3 had lower crystallized and fluid intelligence.
Academic Performance and School Environment
Participants in all subtypes reported less positive school involvement and had a less positive view of their school environment as a place of opportunity and support but only participants in subtype 3 reported higher alienation from academic goals and were more likely to drop in grades
Parental characteristics
The prevalence of parental psychiatric diagnosis and help-seeking for mental health problems were higher in all subtypes while the parents of participants in subtype 3 were less likely to live together.
Family and Peer Environment
Greater family conflict and higher exposure to adverse life events and bullying were common to all subtypes. Additionally, participants in subtypes 2 and 3 reported less parental engagement and those in subtype 3 were also less likely to have a regular group of friends.
Socioeconomic environment
ABCD participants in subtype 1 came from families with higher family income and higher education level, but participants in all subtypes experienced periods of financial adversity. Only ABCD participants in subtype 3 lived in neighborhoods with a higher area deprivation index.
Neuroimaging Profiles of the HYDRA-subtypes
Figure 2 presents the differences between each subtype and the typically developing group for each neuroimaging phenotype at PFDR < 0.005. Subtype 1 was comparable to the typically developing group in terms of neurite density but had larger amygdala, striatal, and thalamic volumes and globally greater surface area expansion; they also had lower cortical thickness and higher intra-cortical myelin in dorsal prefrontal regions. Subtype 2 evidenced globally higher cortical thickness and globally lower cortical surface expansion and cortical myelin, as well as lower neurite density in ventromedial prefrontal, parietal, and occipital cortices. Subtype 3 was characterized by lower cortical thickness primarily in posterior brain regions and globally lower surface area, lower subcortical volume coupled with higher cortical myelination and neural density.
Regional differences (represented by the corresponding t-values) between each subtype and the typically developing (TD) group in cortical thickness (CT), cortical surface area (CSA), subcortical volumes (SV), neurite density (ND) and gray/white matter contrast (GWC). Only significant differences at PFDR < 0.005 are shown. Warm and cool colors respectively indicate higher and lower values in each subtype compared to the TD group.
Discussion
We leveraged the power of machine learning with the unique resources of the ABCD study to test for the presence of neuroimaging-defined subtypes in a cross-sectional sample of youth with juvenile mood and anxiety disorders. We identified three transdiagnostic subtypes that evidenced greater exposure to parental psychopathology, family conflict, and adverse experiences including bullying, compared to the typically developing group. Beyond these similarities, the three subtypes had distinct neuroimaging profiles potentially implicating different developmental mechanisms.
The HYDRA subtypes transcend clinical diagnoses
The diagnostic labels of the ABCD participants were not used in defining the HYDRA-derived clusters. However, it is worth noting that the diagnostic composition of the ABCD sample in terms of mood and anxiety disorders is similar to that reported in other studies. For instance, the US National Survey of Children’s Health (2016–2020), which included 174,551 children, found a prevalence of anxiety disorders around 9% and depression around 4% while the corresponding rates here are 10% for anxiety disorders, and 3.5% for depression [48]. The prevalence of all pediatric bipolar disorder subtypes in the ABCD sample (around 3.7%) is also comparable to the 3.9% rate of pediatric bipolar spectrum disorders reported in a 2019 meta-analysis [49].
The HYDRA subtypes identified using neuroimaging-based clustering included a mixture of ABCD participants with mood and anxiety disorders that may reflect diagnostic equivocality with some youth showing diagnostic continuity while others may change diagnostic status or may recover [50,51,52]. However, neuroimaging studies in adults with established and chronic diagnoses of mood and anxiety disorders have also failed to identify diagnosis-specific changes in neuroimaging phenotypes suggesting that the mechanisms underlying symptom expression transcend diagnostic categories regardless of age [15, 17].
The HYDRA subtypes had similar exposures to adversity
Risk factors for mood and anxiety disorders have been widely researched. The literature provides robust evidence that risk factors are not diagnosis-specific but are shared across multiple disorders. These include familial predisposition [53,54,55], perinatal exposure to substances and medications [56, 57], multiple indicators of lower socioeconomic status (lower parental education, low family income, and neighborhood deprivation) [58, 59], family dysfunction, and poor parental practices [60, 61], unfavorable view of the school environment [62] and exposure to a range of adverse experiences, including difficulties in forming peer-relationships and bullying [63]. The findings of the current study largely reinforce this pattern with the exception of subtype 1, in which several socioeconomic indicators relating to family income and education and positive parental engagement were better than those in the typically developing group. Subtype 3 evidence a greater burden of adversity with greater negative exposures across most domains.
The HYDRA subtypes had distinct neuroimaging profiles
Prior neuroimaging studies in mood and anxiety disorders in youth have focused exclusively on brain morphometry using diagnosis-specific case-control comparisons. Excessive cortical thinning and lower surface area have been reported in youth depression [64, 65] and mania/hypomania [66]. Some studies have associated juvenile anxiety disorders with both higher and lower cortical thickness in different prefrontal areas, and lower subcortical volumes [67,68,69] but others found no case-control differences [70, 71]. A key supposition of these neuroimaging studies was that each diagnostic group would be associated with a distinct pattern of brain structural alterations underpinning the specific disorder-related symptoms. The present findings do not support such formulation as the three subtypes identified were transdiagnostic.
Instead, our results suggest that different brain maturational profiles can give rise to a range of clinical symptoms. Typical brain maturational events that dominate late childhood and adolescence are cortical myelination along a posterior-to-anterior gradient [72] and adaptive synaptic/dendritic density reduction [73, 74]. Associated brain morphometric changes involve cortical thinning [25, 75, 76], cortical surface area expansion [77], and a gradual reduction in subcortical volumes from childhood onwards [78, 79]. During typical development, cortical thinning, surface area expansion, and cortical myelination have been associated with higher cognitive abilities [80,81,82,83], Conversely, excessive cortical thinning and diminished cortical surface area expansion have been associated with lower cognitive abilities [84, 85].
In this context, the neuroimaging profile of subtype 1 suggests a pattern of imbalanced cortical-subcortical maturation compared to the typically developing group, with subcortical regions lagging behind prefrontal cortical thinning, myelination and cortical surface expansion. This interpretation is aligned with cortical maturation being associated with better cognitive abilities [80,81,82,83] which were also characteristic of subtype 1 (Fig. 2, Table 1). The neuroimaging profile of subtype 2 is suggestive of a pattern of generally delayed cortical and subcortical maturation evidenced by higher cortical thickness and subcortical volume and lower cortical surface area expansion and myelination compared to the typically developing group (Fig. 2). This pattern was also associated with cognitive disadvantage (Table 1). Subtype 3 showed evidence of atypical brain maturation involving globally lower cortical thickness and surface coupled with higher myelination and neural density and significant disadvantage in cognition, academic performance, and peer relationships (Fig. 2; Table 1). These findings can be interpreted as potential evidence of abnormalities in cell size and packing density which resonates with abnormalities in brain maturation seen in neurodevelopmental disorders [86, 87].
The ABCD study includes planned follow-up assessments using the same protocols that cover a 10-year period of development and will thus enable us to assess the value of neuroimaging-based classification of mood and anxiety disorders identified here. Specifically, the availability of longitudinal data will enable the comparison of this approach to traditional diagnosis-based models for the prediction of functional outcomes of ABCD participants, with regards to cognition, psychopathology and social functioning.
Methodological considerations
Compared to previous studies, the large sample size, narrow age range, and the epidemiological approach to recruitment are major strengths as they optimize the capacity to examine brain variability in mood and anxiety disorders while minimizing challenges arising from selection bias and the inclusion of participants at different developmental stages. The use of HYDRA is a methodological advance over other clustering techniques as it enables the identification of clinical subgroups that differ from the normative reference group along multiple dimensions. Analyses of data from subsequent follow-up waves of the ABCD study will enable the investigation of the longitudinal evolution of the clusters identified and their prognostic values for mental health.
Taken together, the findings of this study uncovered brain structural heterogeneity in youth with mood and anxiety disorders indicating marked differences in maturational profiles but lack diagnostic specificity. Future follow-up analyses will enhance understanding of the functional consequences for cognition and psychopathology of further brain development as ABCD participants transition through adolescence.
Data availability
The ABCD dataset can be accessed through the National Data Archive (https://data-archive.nimh.nih.gov/abcd).
Code availability
The code for computing HYDRA clusters is freely available at https://github.com/evarol/HYDRA.
References
GBD 2019 Diseases and Injuries Collaborators. Global burden of 369 diseases and injuries in 204 countries and territories, 1990-2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet. 2020;396:12041222.
Solmi M, Radua J, Olivola M, Croce E, Soardo L, Salazar de Pablo G, et al. Age at onset of mental disorders worldwide: large-scale meta-analysis of 192 epidemiological studies. 2022;27:281–95.
Kim-Cohen J, Caspi A, Moffitt TE, Harrington H, Milne BJ, Poulton R. Prior juvenile diagnoses in adults with mental disorder: developmental follow-back of a prospective-longitudinal cohort. Arch Gen Psychiatry. 2003;60:709–17.
Roza SJ, Hofstra MB, van der Ende J, Verhulst FC. Stable prediction of mood and anxiety disorders based on behavioral and emotional problems in childhood: a 14-year follow-up during childhood, adolescence, and young adulthood. Am J Psychiatry. 2003;160:2116–21.
Malla A, Shah J, Iyer S, Boksa P, Joober R, Andersson N, et al. Youth mental health should be a top priority for health care in Canada. Can J Psychiatry. 2018;63:216–22.
Javed A. WPA action plan 2020-2023: a way forward. World Psychiatry. 2020;19:411–2.
World Health Organization. Global strategy for women’s, children’s and adolescents’ health (2016–2030). New York: every woman every child; 2015. Retrieved: http://www.everywomaneverychild.org/wp-content/uploads/2016/12/EWEC_Global_Strategy_EN_inside_LogoOK_web.pdf
Mei C, Fitzsimons J, Allen N, Alvarez-Jimenez M, Amminger GP, Browne V, et al. Global research priorities for youth mental health. Early Inter Psychiatry. 2020;14:3–13.
World Health Organization. International classification of diseases for mortality and morbidity statistics (11th Revision). (2018). Retrieved: https://icd.who.int/browse11/l-m/en
American Psychiatric Association. Diagnostic and statistical manual of mental disorders (DSM-5). Washington, DC: American Psychiatric Association; 2013.
Beard C, Millner AJ, Forgeard MJ, Fried EI, Hsu KJ, Treadway MT, et al. Network analysis of depression and anxiety symptom relationships in a psychiatric sample. Psychol Med. 2016;46:3359–69.
Judd LL, Schettler PJ, Akiskal HS, Maser J, Coryell W, Solomon D, et al. Long-term symptomatic status of bipolar I vs. bipolar II disorders. Int J Neuropsychopharmacol. 2003;6(Jun):127–37.
Hafeman DM, Merranko J, Axelson D, Goldstein BI, Goldstein T, Monk K, et al. Toward the definition of a bipolar prodrome: dimensional predictors of bipolar spectrum disorders in at-risk youths. Am J Psychiatry. 2016;173:695–704.
Duffy A, Goodday S, Keown-Stoneman C, Grof P. The emergent course of bipolar disorder: observations over two decades from the Canadian high-risk offspring cohort. Am J Psychiatry. 2019;176:720–9.
Janiri D, Moser DA, Doucet GE, Luber MJ, Rasgon A, Lee WH, et al. Shared neural phenotypes for mood and anxiety disorders: a meta-analysis of 226 task-related functional imaging studies. JAMA Psychiatry. 2020;77:172–9.
Morneau-Vaillancourt G, Coleman JRI, Purves KL, Cheesman R, Rayner C, Breen G, et al. The genetic and environmental hierarchical structure of anxiety and depression in the UK Biobank. Depress Anxiety. 2020;37:512–20.
Pelin H, Ising M, Stein F, Meinert S, Meller T, Brosch K, et al. Identification of transdiagnostic psychiatric disorder subtypes using unsupervised learning. Neuropsychopharmacology 2021;46:1895–905.
Yang T, Frangou S, Lam RW, Huang J, Su Y, Zhao G, et al. Probing the clinical and brain structural boundaries of bipolar and major depressive disorder. Transl Psychiatry. 2021;11:48. 14
Tokuda T, Yoshimoto J, Shimizu Y, Okada G, Takamura M, Okamoto Y, et al. Identification of depression subtypes and relevant brain regions using a data-driven approach. Sci Rep. 2018;8:14082.
Kaczkurkin AN, Sotiras A, Baller EB, Barzilay R, Calkins ME, Chand GB, et al. Neurostructural heterogeneity in youths with internalizing symptoms. Biol Psychiatry. 2020;87:473–82.
Fan H, Kuang N, Wu X, Yu G, Jia T, Sahakian BJ, et al. Anxiety-impulsivity subtypes in adolescent internalizing disorder are characterized by distinguishable neurodevelopmental, neurocognitive and clinical trajectory signatures. MedRxiv preprint 2021: https://doi.org/10.1101/2021.10.30.21265692.
Karcher NR, Barch DM. The ABCD study: understanding the development of risk for mental and physical health outcomes. Neuropsychopharmacology 2021;46:131–42.
White NS, Leergaard TB, D’Arceuil H, Bjaalie JG, Dale AM. Probing tissue microstructure with restriction spectrum imaging: histological and theoretical validation. Hum Brain Mapp. 2013;34:327–46.
Eickhoff S, Walters NB, Schleicher A, Kril J, Egan GF, Zilles K, et al. High-resolution MRI reflects myeloarchitecture and cytoarchitecture of human cerebral cortex. Hum Brain Mapp. 2005;24:206–15.
Westlye LT, Walhovd KB, Dale AM, Bjørnerud A, Due-Tønnessen P, Engvig A, et al. Differentiating maturational and aging-related changes of the cerebral cortex by use of thickness and signal intensity. Neuroimage 2010;52:172–85.
Arango C, Dragioti E, Solmi M, Cortese S, Domschke K, Murray R, et al. Risk and protective factors for mental disorders beyond genetics: an evidence-based atlas. World Psychiatry. 2021;20:417–36.
Modabbernia A, Janiri D, Doucet GE, Reichenberg A, Frangou S. Multivariate patterns of brain-behavior-environment associations in the adolescent brain and cognitive development study. Biol Psychiatry. 2021;89:510–20.
Modabbernia A, Reichenberg A, Ing A, Moser DA, Doucet GE, Artiges E, et al. Linked patterns of biological and environmental covariation with brain structure in adolescence: a population-based longitudinal study. Mol Psychiatry. 2021;26:4905–18.
Garavan H, Bartsch H, Conway K, Decastro A, Goldstein RZ, Heeringa S, et al. Recruiting the ABCD sample: design considerations and procedures. Dev Cogn Neurosci. 2018;32:16–22.
Kaufman J, Birmaher B, Brent D, Rao U, Flynn C, Moreci P, et al. Schedule for affective disorders and schizophrenia for school-age children-present and lifetime version (K-SADS-PL): initial reliability and validity data. J Am Acad Child Adolesc Psychiatry. 1997;36:980–8.
Kobak KA, Taylor LH, Dottl SL, Greist JH, Jefferson JW, Burroughs D, et al. Computerized screening for psychiatric disorders in an outpatient community mental health clinic. Psychiatr Serv. 1997;48:1048–57.
Hagler DJ, Hatton S, Cornejo MD, Makowski C, Fair DA, Dick AS, et al. Image processing and analysis methods for the Adolescent Brain Cognitive Development Study. Neuroimage. 2019:116091.
Achenbach TM, Rescorla LA. Manual for the ASEBA school-age forms & profiles. Research center for children, youth, & families. Burlington, VT; 2001.
Weintraub S, Dikmen SS, Heaton RK, Tulsky DS, Zelazo PD, Bauer PJ, et al. Cognition assessment using the NIH Toolbox. Neurology. 2013;80:S54–64.
Hamilton CM, Strader LC, Pratt JG, Maiese D, Hendershot T, Kwok RK, et al. The PhenX Toolkit: get the most from your measures. Am J Epidemiol. 2011;174:253–60.
Barch DM, Albaugh MD, Avenevoli S, Chang L, Clark DB, Glantz MD, et al. Demographic, physical and mental health assessments in the adolescent brain and cognitive development study: rationale and description. Dev Cogn Neurosci. 2018;32:55–66.
Luciana M, Bjork JM, Nagel BJ, Barch DM, Gonzalez R, Nixon SJ, et al. Adolescent neurocognitive development and impacts of substance use: Overview of the adolescent brain cognitive development (ABCD) baseline neurocognition battery. Dev Cogn Neurosci. 2018;32:67–79.
Zucker RA, Gonzalez R, Feldstein Ewing SW, Paulus MP, Arroyo J, Fuligni A, et al. Assessment of culture and environment in the adolescent brain and cognitive development study: rationale, description of measures, and early data. Dev Cogn Neurosci. 2018;32:107–20.
Casey BJ, Cannonier T, Conley MI, Cohen AO, Barch DM, Heitzeg MM, et al. The Adolescent Brain Cognitive Development (ABCD) study: Imaging acquisition across 21 sites. Dev Cogn Neurosci. 2018;32:43–54.
Desikan RS, Ségonne F, Fischl B, Quinn BT, Dickerson BC, Blacker D, et al. An automated labeling system for subdividing the human cerebral cortex on MRI scans into gyral based regions of interest. Neuroimage. 2006;31:968–80.
Fischl B, Salat DH, Busa E, Albert M, Dieterich M, Haselgrove C, et al. Whole brain segmentation: automated labeling of neuroanatomical structures in the human brain. Neuron. 2002;33:341–55.
Fortin JP, Cullen N, Sheline YI, Taylor WD, Aselcioglu I, Cook PA, et al. Harmonization of cortical thickness measurements across scanners and sites. 2018;167:104–20.
van Buuren S, Groothuis-Oudshoorn K. Mice: multivariate imputation by chained equations in R. J Stat Softw. 2010;1–68.
Varol E, Sotiras A, Davatzikos C. Alzheimer’s Disease Neuroimaging Initiative. HYDRA: Revealing heterogeneity of imaging and genetic patterns through a multiple max-margin discriminative analysis framework. Neuroimage. 2017;145:346–64.
Hubert L, Arabie P. Comparing partitions. J Classif. 1985;2:193–218.
Benjamini Y, Hochberg Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc (Ser B). 1995;57:289–300.
Glasser MF, Van Essen DC. Mapping human cortical areas in vivo based on myelin content as revealed by T1- and T2-weighted MRI. J Neurosci. 2011;31:11597–616.
Lebrun-Harris LA, Ghandour RM, Kogan MD, Warren MD. Five-year trends in us children’s health and well-being, 2016-2020. JAMA Pediatr. 2022;176:e220056.
Van Meter A, Moreira ALR, Youngstrom E. Updated meta-analysis of epidemiologic studies of pediatric bipolar disorder. J Clin Psychiatry. 2019;80:18r12180.
Kwong ASF, Manley D, Timpson NJ, Pearson RM, Heron J, Sallis H, et al. Identifying critical points of trajectories of depressive symptoms from childhood to young adulthood. J Youth Adolesc. 2019;48:815–27.
Caspi A, Houts RM, Ambler A, Danese A, Elliott ML, Hariri A, et al. Longitudinal assessment of mental health disorders and comorbidities across 4 decades among participants in the dunedin birth cohort study. JAMA Netw Open. 2020;3:e203221.
Kessler RC, Wang PS. The descriptive epidemiology of commonly occurring mental disorders in the United States. Annu Rev Public Health. 2008;29:115–29.
Sullivan PF, Neale MC, Kendler KS. Genetic epidemiology of major depression: review and meta-analysis. Am J Psychiatry. 2000;157:1552–62.
Song J, Bergen SE, Kuja-Halkola R, Larsson H, Landén M, Lichtenstein P. Bipolar disorder and its relation to major psychiatric disorders: a family-based study in the Swedish population. Bipolar Disord. 2015;17:184–93.
Meier SM, Trontti K, Purves KL, Als TD, Grove J, Laine M, et al. Genetic variants associated with anxiety and stress-related disorders: a genome-wide association study and mouse-model study. JAMA Psychiatry. 201;76:924–32.
Creeley CE, Denton LK. Use of prescribed psychotropics during pregnancy: a systematic review of pregnancy, neonatal, and childhood outcomes. Brain Sci. 2019;9:235.
Sandtorv LB, Hysing M, Rognlid M, Nilsen SA, Elgen IB. Mental health in school-aged children prenatally exposed to alcohol and other substances. Subst Abus. 2017;11:1178221817718160.
Adkins DE, Wang V, Elder GH Jr. Structure and stress: trajectories of depressive symptoms across adolescence and young adulthood. Soc Forces. 2009;88:31.
Kinge JM, Øverland S, Flatø M, Dieleman J, Røgeberg O, Magnus MC, et al. Parental income and mental disorders in children and adolescents: prospective register-based study. Int J Epidemiol. 2021;50:1615–27.
Rask K, Astedt-Kurki P, Paavilainen E, Laippala P. Adolescent subjective well-being and family dynamics. Scand J Caring Sci. 200;17:129–38.
Eun JD, Paksarian D, He JP, Merikangas KR. Parenting style and mental disorders in a nationally representative sample of US adolescents. Soc Psychiatry Psychiatr Epidemiol. 2018;53:11–20.
Aldridge JM, McChesney K. The relationships between school climate and adolescent mental health and wellbeing: a systematic literature review. Int J Educ Res. 2018;88:121–45.
Chudal R, Tiiri E, Brunstein Klomek A, Ong SH, Fossum S, Kaneko H, et al. Eurasian child mental health study (EACMHS) Group. Victimization by traditional bullying and cyberbullying and the combination of these among adolescents in 13 European and Asian countries. Eur Child Adolesc Psychiatry. 2021;31:1391–404.
Schmaal L, Hibar DP, Sämann PG, Hall GB, Baune BT, Jahanshad N, et al. Cortical abnormalities in adults and adolescents with major depression based on brain scans from 20 cohorts worldwide in the ENIGMA Major Depressive Disorder Working Group. Mol Psychiatry. 2017;22:900–9.
Bos MGN, Peters S, van de Kamp FC, Crone EA, Tamnes CK. Emerging depression in adolescence coincides with accelerated frontal cortical thinning. J Child Psychol Psychiatry. 2018;59:994–1002.
Hibar DP, Westlye LT, Doan NT, Jahanshad N, Cheung JW, Ching CRK, et al. Cortical abnormalities in bipolar disorder: an MRI analysis of 6503 individuals from the ENIGMA Bipolar Disorder Working Group. Mol Psychiatry. 2018;23:932–42.
Gold AL, Steuber ER, White LK, Pacheco J, Sachs JF, Pagliaccio D, et al. Cortical thickness and subcortical gray matter volume in pediatric anxiety disorders. Neuropsychopharmacology 2017;42:2423–33.
Strawn JR, Hamm L, Fitzgerald DA, Fitzgerald KD, Monk CS, Phan KL. Neurostructural abnormalities in pediatric anxiety disorders. J Anxiety Disord. 2015;32:81–88.
Feurer C, Suor JH, Jimmy J, Klumpp H, Monk CS, Phan KL, et al. Differences in cortical thinning across development among individuals with and without anxiety disorders. Depress Anxiety. 2021;38:372–81.
Suffren S, Chauret M, Nassim M, Lepore F, Maheu FS. On a continuum to anxiety disorders: adolescents at parental risk for anxiety show smaller rostral anterior cingulate cortex and insula thickness. J Affect Disord. 2019;248:34–41.
Ducharme S, Albaugh MD, Hudziak JJ, Botteron KN, Nguyen TV, Truong C, et al. Anxious/depressed symptoms are linked to right ventromedial prefrontal cortical thickness maturation in healthy children and young adults. Cereb Cortex. 2014;24:2941–50.
Williamson JM, Lyons DA. Myelin dynamics throughout life: an ever-changing landscape? Front Cell Neurosci. 2018;12:424.
Huttenlocher PR. Synaptic density in human frontal cortex - developmental changes and effects of aging. Brain Res. 1979;163:195–205.
Petanjek Z, Judas M, Kostović I, Uylings HB. Lifespan alterations of basal dendritic trees of pyramidal neurons in the human prefrontal cortex: a layer-specific pattern. Cereb Cortex. 2008;18:915–29.
Frangou S, Modabbernia A, Williams SCR, Papachristou E, Doucet GE, Agartz I, et al. Cortical thickness across the lifespan: Data from 17,075 healthy individuals aged 3–90 years. Hum Brain Mapp. 2022;43:431–51.
Zhou D, Lebel C, Treit S, Evans A, Beaulieu C. Accelerated longitudinal cortical thinning in adolescence. Neuroimage. 2015;104:138–45.
Tamnes CK, Herting MM, Goddings AL, Meuwese R, Blakemore SJ, Dahl RE, et al. Development of the cerebral cortex across adolescence: a multisample study of inter-related longitudinal changes in cortical volume, surface area, and thickness. J Neurosci. 2017;37:3402–12.
Raznahan A, Shaw PW, Lerch JP, Clasen LS, Greenstein D, Berman R, et al. Longitudinal four-dimensional mapping of subcortical anatomy in human development. Proc Natl Acad Sci USA. 2014;111:1592–7.
Dima D, Modabbernia A, Papachristou E, Doucet GE, Agartz I, Aghajani M, et al. Subcortical volumes across the lifespan: data from 18,605 healthy individuals aged 3–90 years. Hum Brain Mapp. 2022;43:452–69.
Schnack HG, van Haren NE, Brouwer RM, Evans A, Durston S, Boomsma DI, et al. Changes in thickness and surface area of the human cortex and their relationship with intelligence. Cereb Cortex. 2015;25:1608–17.
Fjell AM, Westlye LT, Amlien I, Tamnes CK, Grydeland H, Engvig A, et al. High-expanding cortical regions in human development and evolution are related to higher intellectual abilities. Cereb Cortex. 2015;25:26–34.
Lewis JD, Evans AC, Tohka J, Brain Development Cooperative Group, Pediatric Imaging, Neurocognition and Genetics Study. T1 white/gray contrast as a predictor of chronological age, and an index of cognitive performance. Neuroimage. 2018;173:341–50.
Norbom LB, Doan NT, Alnæs D, Kaufmann T, Moberget T, Rokicki J, et al. Probing Brain developmental patterns of myelination and associations with psychopathology in youths using gray/white matter contrast. Biol Psychiatry. 2019;85:389–98.
Burgaleta M, Johnson W, Waber DP, Colom R, Karama S. Cognitive ability changes and dynamics of cortical thickness development in healthy children and adolescents. Neuroimage. 2014;84:810–9.
Tadayon E, Pascual-Leone A, Santarnecchi E. Differential contribution of cortical thickness, surface area, and gyrification to fluid and crystallized intelligence. Cereb Cortex. 2020;30:215–25.
Caverzasi E, Mandelli ML, Hoeft F, Watson C, Meyer M, Allen IE, et al. Abnormal age-related cortical folding and neurite morphology in children with developmental dyslexia. Neuroimage Clin. 2018;18:814–21.
Fischi-Gomez E, Bonnier G, Ward N, Granziera C, Hadjikhani N. Ultrahigh field in vivo characterization of microstructural abnormalities in the orbitofrontal cortex and amygdala in autism. Eur J Neurosci. 2021;54:6229–36.
Author information
Authors and Affiliations
Contributions
All authors contributed to the conception of the study; RG and SF performed the data analysis; all authors wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
RG, RS and SF report no conflicts of interest. LNY is a consultant and/or has received speaker fees and/or sits on the advisory board and/or receives research funding from Abbvie, Alkermes, Allergan, Canadian Network for Mood and Anxiety Treatments (CANMAT), Canadian Institutes of Health Research (CIHR), Dainippon Sumitomo Pharma, Gedeon Richter, GSK, Intracellular Therapies, Lundbeck, Merck, Otsuka, Sanofi and Sunovion over the past 3 years.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ge, R., Sassi, R., Yatham, L.N. et al. Neuroimaging profiling identifies distinct brain maturational subtypes of youth with mood and anxiety disorders. Mol Psychiatry 28, 1072–1078 (2023). https://doi.org/10.1038/s41380-022-01925-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41380-022-01925-9
- Springer Nature Limited