Abstract
Chromosomal rearrangements that result in oncogenic kinase activation are present in many solid and hematological malignancies, but none have been reported in multiple myeloma (MM). Here we analyzed 1421 samples from 958 myeloma patients using a targeted assay and detected fusion genes in 1.5% of patients. These fusion genes were in-frame and the majority of them contained kinase domains from either receptor tyrosine kinases (ALK, ROS1, NTRK3, and FGFR1) or cytoplasmic kinases (BRAF, MAP3K14, and MAPK14), which would result in the activation of MEK/ERK, NF-κB, or inflammatory signaling pathways. Fusion genes were present in smoldering MM, newly diagnosed MM, and relapse patient samples indicating they are not solely late events. Most fusion genes were subclonal in nature, but one EML4-ALK fusion was clonal indicating it is a driver of disease pathogenesis. Samples with fusions of receptor tyrosine kinases were not found in conjunction with clonal Ras/Raf mutations indicating a parallel mechanism of MEK/ERK pathway activation. Fusion genes involving MAP3K14 (NIK), which regulates the NF-κB pathway, were detected as were t(14;17) rearrangements involving NIK in 2% of MM samples. Activation of kinases in myeloma through rearrangements presents an opportunity to use treatments existing in other cancers.
Similar content being viewed by others
Introduction
Fusion genes are the product of genomic rearrangements where two genes are rearranged to create a new fusion gene with either increased or inappropriate expression and functionality that was not previously evident. One of the initial rearrangements defining fusion genes was the t(9;22), typical of chronic myeloid leukemia (CML) cases [1, 2]. This translocation generates a BCR-ABL1 fusion gene, which results in the activation of the tyrosine kinase domain of the ABL protein. The increased signaling, which occurs as a result of this rearrangement, can be therapeutically targeted by specific tyrosine kinase inhibitors (TKIs), resulting in clinically relevant responses [3, 4].
In solid tumors, fusion genes are also seen and similarly can result in the activation of kinase domains with key deregulated genes including the receptor tyrosine kinases (RTKs) ALK, ROS1, RET, FGFR1/2/3, and NTRK1/2/3, as well as cytoplasmic kinases such as BRAF [5, 6]. These rearrangements are not as frequent as the t(9;22) with rates of up to 12% in patients with thyroid carcinoma, but on the whole, across all types of cancer, the rate is in the order of 1–2% [7]. The most common signaling pathway deregulated by these fusion genes is the MEK/ERK pathway. Analogous to CML, these kinase fusion genes can also be therapeutically targeted, a key example of which is the EML4-ALK fusion gene seen in 3–7% of patients with adenocarcinoma of the lung [8].
Multiple myeloma (MM) is characterized by primary translocations into the immunoglobulin (Ig) loci, which occur in ~40% of patients [9]. These structural rearrangements place the Ig super-enhancer next to an oncogene, resulting in its overexpression [10]. The common gene fusions seen in MM involve the Ig loci, including cases with a t(4;14) where the breakpoint within MMSET results in an Ig-MMSET transcript, but an alteration in functionality as a consequence is not seen [11, 12]. A key signaling abnormality in MM is increased MEK/ERK pathway activation due to activating point mutations in KRAS, NRAS, and BRAF seen in up to 50% of patients [13]. However, despite the frequent deregulation of this pathway, functional gene fusions involving RTKs have not been described. Here we analyzed 1421 patient samples using a targeted assay able to detect gene fusions and describe their prevalence in MM.
Methods
Patient samples and nucleic acid extraction
We report on 1421 samples from 958 individuals diagnosed with monoclonal gammopathy of undetermined significance (MGUS), smoldering multiple myeloma (SMM), or MM who underwent targeted sequencing with the FoundationOne heme (F1H) assay [14, 15] between September 2013 and August 2016. All patients signed a written informed consent in keeping with institutional, federal, and Helsinki Declaration guidelines. Tumor samples were obtained from bone marrow aspirates, enriched by CD138+ selection using magnetic beads (AutoMACs, Miltenyi Biotech, Cologne, Germany or RoboSep, StemCell Technologies, Vancouver, Canada). RNA and DNA were extracted using the AllPrep DNA/RNA mini kit (Qiagen, Hilden, Germany), RNeasy RNA extraction kit (Qiagen), or Puregene DNA extraction kit (Qiagen).
Foundation One heme reporting
DNA ≥50 ng and/or RNA >300 ng was interrogated using the F1H Panel (Foundation Medicine, MA). The panel analyzes the complete coding DNA sequence of 405 genes, as well as selected introns of 31 genes involved in chromosomal rearrangements. It also interrogates the RNA sequence of 265 commonly rearranged genes resulting in gene fusions. Genes included in this assay encode known or likely targets of therapy, either FDA-approved or in clinical trials, or are otherwise known drivers of oncogenesis. Due to the capture strategies, most fusions are detected by either DNA or RNA sequencing and not both. Of the 1421 samples, 565 were processed on the DNA panel only and 856 were processed on the DNA and RNA panels. Data have been submitted to the European Genome-Phenome Archive under accession EGAS00001002874.
Sequencing was carried out to an average depth of 468x and was performed using a HiSeq 2500 (Illumina). Sequences were analyzed for base substitutions, indels, copy-number alterations (focal amplifications with ≥8 copies and homozygous deletions), and selected gene rearrangements. Variant processing is described elsewhere [14, 16], but importantly involved removal of germ line variants from the 1000 Genomes Project (dbSNP135), as a matched patient non-tumor sample is not used to identify truly somatic variants. All inactivating events (i.e., truncations and deletions) in known tumor suppressor genes were also called as significant. To maximize mutation-detection accuracy (sensitivity and specificity) in clinical specimens, the test has been optimized and validated to detect base substitutions at a ≥5% variant allele frequency (VAF) and indels with a ≥10% VAF to ≥99% accuracy. However, mutations are reported down to 1% VAF where the variant is a known hotspot and there is sufficient purity and sequencing depth. Reports were generated by Foundation Medicine and data files containing additional information (VAF, variant type, depth at variant location, genomic coordinates) were received.
RT-PCR confirmation of fusion genes
To confirm the fusion gene breakpoints, reverse transcription-polymerase chain reaction (RT-PCR) was performed on seven samples. Complementary DNA (cDNA) was synthesized from 200 ng RNA using iScript (Bio-Rad). Two microliters of a 1/10 dilution of the cDNA was used in subsequent PCR reactions. PCR bands were excised from agarose gels, purified, and sequenced by the Sanger method. Sanger sequence reads were compared against the genome using BLAST.
Determining clonality of fusion genes
Custom probe-based qPCR assays (Integrated DNA Technologies) were designed across the DNA breakpoints in order to determine if the fusion genes are clonal. Assays were also designed against the unarranged germ line sequence for comparison, as well as for any Ig translocations that had also been detected in the same samples. Only samples with an identified DNA breakpoint were analyzed as the sequence is required to design the assays. Rearranged and germ line sequence assays were labeled with FAM or HEX, respectively. DNA from the samples was used in a digital droplet PCR reaction to determine the proportion of sample with a rearrangement. If a primary IGH translocation breakpoint was assayed, it was assumed to be clonal and the percent positive droplets in the IGH and fusion gene assay adjusted to account for CD138 cell purity.
Gene expression profiling
Gene expression profiling (GEP) using Affymetrix U133 Plus 2.0 arrays was performed. The GEP-based 70 risk score (GEP70) and molecular subgroups were determined as previously described [17, 18]. Dataset GSE4581 was used to determine overexpression of MAP3K14. Overexpression was determined as values with >2 standard deviations of the mean for probeset 205192_at.
CoMMpass dataset validation
Data are available at dbGap under accession number phs000748.v5.p4. RNA-seq data were processed by STAR and Salmon. Gene level quantification of RNA expression was performed using Star (2.5.1b) generating read counts per gene while aligning the reads to the reference genome (hg38). Salmon (0.6.0) was run to calculate transcript level quantification. Additionally, the Salmon transcript values were summed to get quantification at the gene level.
Previously, aligned BAM files from whole-genome sequencing (WGS) were converted to FASTQ using Picard tools v2.1.1 to extract read sequences and base quality scores. All reads were realigned to the human genome assembly hg19 using BWA-mem. Base recalibration of alignments was performed using GATK v3.6. Translocations in whole-genome data were detected using Manta (version 0.29.6) with default settings.
Using the CoMMpass dataset, comprising 564 RNA-seq samples that have matching WGS, fusion genes were identified using MapSplice 2 (v2.2.1). The results were parsed to identify relevant kinase fusion genes. The identified fusion genes were confirmed using the WGS data where the translocation breakpoints at the DNA level were identified using Manta (v0.29.6).
Results
Identification of fusion genes in multiple myeloma
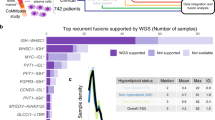
Using a capture panel for both RNA and DNA, 1421 samples from 958 patients were sequenced for clinical purposes. From these data, 39 potential fusion genes were identified and annotated from either their DNA breakpoints (n = 11) or RNA sequences matching multiple genes (n = 31) or both (n = 3). Of the 39 potential fusion genes, 21 were in-frame, potentially resulting in functional fusion proteins (Table 1). The patients with in-frame fusions included those with SMM (n = 1), newly diagnosed MM (n = 5), those who had been previously treated (n = 2), and those who had relapsed (n = 11).
Classes of fusion genes
The panel detects known common kinase and transcription factor fusions, and of the 21 in-frame fusion genes, 12 involved kinases, 6 involved transcription factors, and 3 were neither. Of the 3 that were neither kinases or transcription factors, 2 involved MMSET/WHSC1 and were both in t(4;14) patient samples. Of the 6 involving transcription factors, 3 involved MYC with common partners of MYC rearrangements in MM (TXNDC5 and FOXO3) and were seen in the hyperdiploid samples.
Where a kinase domain was involved, it was located at the 3′ end of the fusion gene, which is common for this type of fusion. The most common partner identified was BRAF (n = 4), followed by NTRK3 (n = 2). All other kinase fusion genes were unique and included ALK, FGFR1, MAP3K14, MAPK14, and ROS1. As expected, all fusions retained the kinase domain. Seven samples had RNA available and the fusion genes were verified by RT-PCR (Figs. 1 and 2). Sanger sequencing identified the breakpoints in the messenger RNA and schematics of the fusion genes are shown.
EML4-ALK rearrangement in myeloma results in increased expression of the tyrosine kinase domain. EML4 and ALK are in opposite transcriptional orientation on chromosome 2. An inversion results in the active promoter from EML4 being placed upstream of the 5′ end of ALK, resulting in expression of the EML4-ALK fusion protein containing a tyrosine kinase domain. The fusion gene was confirmed by RT-PCR and Sanger sequencing. ALK is not expressed in adult tissue but EML4 is, however, in this sample, expression of the EML4-ALK fusion transcript was detected using 3′ expression array analysis
The kinases involved in the fusion genes have been noted in other cancers, including BRAF (papillary thyroid cancer, cutaneous melanoma, and adenocarcinoma of the rectum), NTRK3 (thyroid, melanoma, colon adenocarcinoma, and invasive breast cancer), FGFR1 (adenocarcinoma of the lung and breast cancer), MAP3K14 (head and neck squamous cell carcinoma and ovarian cancer), ROS1 and ALK (adenocarcinoma of the lung).
ALK fusion genes in lung adenocarcinoma involve an inversion on chromosome 2, placing the promoter of EML4 in front of ALK. ALK is not normally expressed in adult tissue, whereas EML4 is expressed. The inversion of the promoters results in the EML4-ALK fusion gene and overexpression of the kinase domain within ALK. As we found an EML4-ALK fusion in our dataset, we used GEP data to determine if ALK was overexpressed. As expected, ALK was not expressed in any sample except for the one with the inversion (Fig. 2).
The kinase fusions fall into two categories: RTKs and cytoplasmic kinases. ROS1, ALK, FGFR1, and NTRK3 are all RTKs that are known to result in downstream MEK/ERK or PIK3CA signaling pathway activation leading to cell proliferation or prevent apoptosis. The cytoplasmic kinases are more diverse, with BRAF also being a member of the MEK/ERK activation pathway, whereas MAPK14 (p38) and MAP3K14, also known as NF-κB-inducing kinase (NIK), are involved in the inflammatory and NF-κB signaling pathways, respectively.
We analyzed the CoMMpass dataset comprising 487 patients and identified a similar spectrum of kinase fusion genes with evidence both in RNA and DNA sequencing including SND1-BRAF, TPR-NTRK1, FCHSD2-MAP3K14, TPM3-NTRK1, CREB1-ALK, IKZF3-MAP3K14, ARHGEF2-NTRK1, and BRAF-AGK (Table 2). These were present in a total of 2.5% of patients. Expression of the kinase gene was not always increased as a result of the fusion, e.g., in BRAF and MAP3K14 fusions (Fig. 3a, b), but was in others (NTRK1 (0.5% of samples) and ALK; Figs. 2 and 3c). The most frequent kinase fusion domain in this dataset involved NIK, where the N-terminus of NIK is replaced by different head genes. This may result in stabilization of NIK through loss of the N-terminal BIRC2 (cIAP-1)-binding domain that regulates degradation of NIK [19].
Expression of MAP3K14 and NTRK1 in newly diagnosed myeloma patient samples. a Spiked expression of probeset 205192_at on U133 Plus 2.0 arrays was used as a surrogate for the t(14;17). b Spiked expression of MAP3K14 in the CoMMpass RNA-seq dataset. c Spiked expression of NTRK1 in the CoMMpass RNA-seq dataset. Red and green circles indicate presence of an Ig or non-Ig translocation in the WGS data, respectively. Yellow circles are samples with a MAP3K14 or NTRK1 fusion gene in RNA-seq data. The solid gray line indicates the median expression of the probeset across all samples. The dotted gray line indicates the median plus 2 standard deviations and was used as the cutoff to determine spiked expression
Association of kinase fusion genes with other MEK/ERK pathway abnormalities
In myeloma, recurrent mutations affect Ras signaling leading to activation of the MEK/ERK pathway. KRAS, NRAS, and BRAF are most frequently mutated and collectively are present in up to 50% of patients. It is known that mutations in these three genes are mutually exclusive, although subclones with different mutations within a patient do exist. Using the data from the F1H assay, we determined if samples also had concomitant kinase fusions and MEK/ERK pathway mutations (Table 3). Five samples had both a kinase fusion and mutation of Ras/Raf genes, however, all but one were at low VAFs indicating they were subclonal.
We went on to determine if the kinase fusion genes were also subclonal by performing droplet digital PCR using breakpoint-specific sequences. DNA level breakpoints were available for four samples with kinase fusions and were tested for both the rearranged and wild-type alleles (Fig. 4). Taking into account the copy number of the involved chromosomes, the EML4-ALK fusion was fully clonal, being present in 55% of the droplets. This patient did not have any other identified MEK/ERK abnormalities but did have a t(14;16) suggesting that the EML4-ALK fusion was not an initiating event and had been selected for during progression. This patient was GEP70 high risk, but did not have gain of 1q or deletion of TP53, however there was a subclonal mutation in TP53 (p.S240R, 2% of reads). The three other patients all had subclonal kinase fusion genes (range 20–38% of cells) and had subclonal mutations in the Ras/Raf pathway genes, indicating parallel evolution of MEK/ERK pathway activation in these patients. One patient had three samples sequenced over a 5-month period and showed consistent expression of the TSPAN3-ROS1 fusion gene in all samples.
Kinase gene fusions can be both clonal or subclonal. Clonality of fusion gene breakpoints was determined using droplet digital PCR (ddPCR) with probes specific for fusion gene breakpoints, unarranged alleles, or IGH translocation breakpoints. The EML4-ALK fusion was clonal being present in ~50% of the DNA molecules, equivalent to one allele having the rearrangement. ARHGAP27-MAP3K14 fusion was subclonal (10% of DNA, 20% of cells), as were KANK1-BRAF (15%/30%; 16%/32% after adjusting for purity) and GTF2I-BRAF (13%/26%; 19%/38% after adjusting for purity)
Overexpression of NIK through an IGH translocation
Fusion genes involving NIK were found in this dataset and in the CoMMpass dataset. We also noticed translocations involving NIK in 2/98 (2%) samples from another targeted panel dataset, indicating multiple mechanisms of kinase activation. The translocations were t(14;17)(q32.33;q21.31) involving the IGH locus and resulted in overexpression of NIK. Both samples with the t(14;17) were hyperdiploid and did not have an additional IGH translocation. Based on a larger dataset of 414 samples, we saw spiked expression of NIK in nine samples, indicating that the frequency of increased kinase expression through the t(14;17) is 2.2% (Fig. 3a). Of these nine samples, there were four hyperdiploid samples, two t(4;14), and one of each t(11;14), t(14;16), and t(14;20). A similar spike in NIK expression was seen in the CoMMpass RNA-sequencing data (n = 734 NDMM) in 3.2% of patients, which coincided with both Ig and non-Ig translocations in those samples (Fig. 3b). The prevalence of primary IGH translocations alongside the t(14;17) indicates that the NIK translocations are secondary events (Table 2).
Discussion
We show for the first time that, analogous to other cancers, kinase domain fusion genes are seen in MM occurring at a similar frequency, ~1.5% of cases. Many of the fusion genes described here have been shown in other cancers to affect the MEK/ERK, PIK3CA, and NF-κB pathways, and are therefore likely to be functional in MM too.
Each of the pathways described is of relevance in MM. Here, we found fusion genes involving BRAF, NTRK3, ALK, FGFR1, and ROS1, which result in activation of the MEK/ERK pathway in other cancers [20]. Even though the kinase domain-containing genes are diverse, they all have similar domain structures and feed into the same downstream pathway that makes them targetable. This is analogous to adenocarcinoma of the lung where fusion genes involving MET, ROS1, and ALK are all treated with TKIs that are effective against each of the kinase domains.
The MEK/ERK pathway is commonly disrupted in MM, with activating point mutations seen in KRAS, NRAS, and BRAF in ~50% of patients [13, 21]. In samples with kinase fusion genes, we only identified subclonal mutations in KRAS, NRAS, and BRAF, consistent with the hypothesis that these mechanisms are mutually exclusive and serve the same functional purpose, constituting a parallel mechanism for pathway activation.
It is highly likely that the fusion genes described here are secondary events, due to their subclonal nature or association with primary translocations. However, these fusions were detected in SMM, as well as NDMM and relapse patients, indicating that they are not necessarily late progression events. A ROS1 fusion gene was detected in a SMM patient. ROS1 is a RTK that activates the MEK/ERK pathway through phosphorylation of Ras. ROS1 fusions are detected in ~2% of non-small cell lung carcinoma (NSCLC) patients [20]. Another key RTK in NSCLC is ALK, which was also identified in this dataset as a fusion gene. These RTK fusion genes are considered as driver events in NSCLC, as they are prevalent, activating, and often clonal in nature [22]. The EML4-ALK fusion gene was clonal in our MM sample, indicating that it is also a driver event in MM and may be a relevant clinical target in a small subset of patients.
NTRK1 gene expression was increased by gene fusion events, and we also saw that in total 0.5% of patient samples in the CoMMpass dataset had increased expression of NTRK1. It is unclear if this increase in expression is solely due to gene fusions with NTRK1 or if there are other genetic or epigenetic events contributing to this. However, this small subset of patients with this abnormality may benefit from TKI treatment options.
Both ROS1, NTRK1, and ALK fusion genes are treated using TKIs, such as crizotinib, in NSCLC with varying degrees of success, due to genetic heterogeneity. Many patients with ALK fusions in NSCLC respond to TKI treatment, but later relapse [23], due to succession of a clone with a mutation in the ALK tyrosine kinase domain or a clone with an alternative MEK/ERK signaling abnormality [24]. Genetic heterogeneity in MM is well described [25,26,27] and likely poses a similar problem in the treatment of patients with these secondary, subclonal events.
There was also evidence of NF-κB activation through fusion genes and Ig translocations involving NIK, which is a central component of the non-canonical NF-κB pathway [28]. NIK is normally targeted for degradation by the proteasome through the TRAF-cIAP destruction complex, and accumulation of NIK is associated with lymphoid malignancies [29]. It has been shown that negative regulators of the non-canonical pathway are frequently deleted or mutated in MM, including members of the TRAF-cIAP complex: TRAF2, TRAF3, BIRC2 (cIAP1), and BIRC3 (cIAP2) [13, 30, 31]. Activation of NIK in myeloma cell lines and patients has been seen through inactivation of this complex, but NIK translocations have only been noted in cell lines [30, 32]. We see here two means of NIK degregulation: firstly through overexpression with the t(14;17) Ig translocation, and secondly with fusion genes that result in loss of the BIRC2 binding domain that regulates NIK degradation. Taken together, these data indicate that NIK is a key player in regulating the non-canonical NF-κB pathway. Both the NIK fusion gene and the translocations are secondary events as they are either subclonal or the sample has a primary Ig translocation, indicating that this could be a key progression event.
The fusion genes described here were detected using a panel targeted against therapeutically tractable fusions that have been previously identified in other cancers. The probability of generating an in-frame fusion gene is 33%, therefore of the 39 fusion genes detected in this study, 13 may be in-frame by chance, and so out of the 21 in-frame fusions detected, 8 may be biologically relevant. However, as these were detected using a targeted panel using prior information from other cancer types, we have enriched for biologically relevant genes and we believe that all of the kinase fusions are biologically relevant. Indeed, fusion genes mostly involving Ig loci have recently been described and are associated with outcome, although no functional fusion genes were described [12]. These may be markers for genome instability rather than have any new biological function from the gene fusions.
References
Bartram CR, Kleihauer E, de Klein A, Grosveld G, Teyssier JR, Heisterkamp N, et al. C-abl and bcr are rearranged in a Ph1-negative CML patient. EMBO J. 1985;4:683–6.
Kurzrock R, Gutterman JU, Talpaz M. The molecular genetics of Philadelphia chromosome-positive leukemias. N Engl J Med. 1988;319:990–8.
Hehlmann R, Hochhaus A, Baccarani M, European L. Chronic myeloid leukaemia. Lancet. 2007;370:342–50.
Hochhaus A, O’Brien SG, Guilhot F, Druker BJ, Branford S, Foroni L, et al. Six-year follow-up of patients receiving imatinib for the first-line treatment of chronic myeloid leukemia. Leukemia. 2009;23:1054–61.
Ciampi R, Knauf JA, Kerler R, Gandhi M, Zhu Z, Nikiforova MN, et al. Oncogenic AKAP9-BRAF fusion is a novel mechanism of MAPK pathway activation in thyroid cancer. J Clin Invest. 2005;115:94–101.
Palanisamy N, Ateeq B, Kalyana-Sundaram S, Pflueger D, Ramnarayanan K, Shankar S, et al. Rearrangements of the RAF kinase pathway in prostate cancer, gastric cancer and melanoma. Nat Med. 2010;16:793–8.
Stransky N, Cerami E, Schalm S, Kim JL, Lengauer C. The landscape of kinase fusions in cancer. Nat Commun. 2014;5:4846.
Shaw AT, Hsu PP, Awad MM, Engelman JA. Tyrosine kinase gene rearrangements in epithelial malignancies. Nat Rev Cancer. 2013;13:772–87.
Morgan GJ, Walker BA, Davies FE. The genetic architecture of multiple myeloma. Nat Rev Cancer. 2012;12:335–48.
Bergsagel PL, Chesi M, Nardini E, Brents LA, Kirby SL, Kuehl WM. Promiscuous translocations into immunoglobulin heavy chain switch regions in multiple myeloma. Proc Natl Acad Sci USA. 1996;93:13931–6.
Keats JJ, Maxwell CA, Taylor BJ, Hendzel MJ, Chesi M, Bergsagel PL, et al. Overexpression of transcripts originating from the MMSET locus characterizes all t(4;14)(p16; q32)-positive multiple myeloma patients. Blood. 2005;105:4060–9.
Cleynen A, Szalat R, Kemal Samur M, Robiou du Pont S, Buisson L, Boyle E, et al. Expressed fusion gene landscape and its impact in multiple myeloma. Nat Commun. 2017;8:1893.
Walker BA, Boyle EM, Wardell CP, Murison A, Begum DB, Dahir NM, et al. Mutational spectrum, copy number changes, and outcome: results of a sequencing study of patients with newly diagnosed myeloma. J Clin Oncol. 2015;33:3911–20.
Frampton GM, Fichtenholtz A, Otto GA, Wang K, Downing SR, He J, et al. Development and validation of a clinical cancer genomic profiling test based on massively parallel DNA sequencing. Nat Biotechnol. 2013;31:1023–31.
He J, Abdel-Wahab O, Nahas MK, Wang K, Rampal RK, Intlekofer AM, et al. Integrated genomic DNA/RNA profiling of hematologic malignancies in the clinical setting. Blood. 2016;127:3004–14.
Chavan SS, He J, Tytarenko R, Deshpande S, Patel P, Bailey M, et al. Bi-allelic inactivation is more prevalent at relapse in multiple myeloma, identifying RB1 as an independent prognostic marker. Blood Cancer J. 2017;7:e535.
Zhan F, Huang Y, Colla S, Stewart JP, Hanamura I, Gupta S, et al. The molecular classification of multiple myeloma. Blood. 2006;108:2020–8.
Shaughnessy JD Jr., Zhan F, Burington BE, Huang Y, Colla S, Hanamura I, et al. A validated gene expression model of high-risk multiple myeloma is defined by deregulated expression of genes mapping to chromosome 1. Blood. 2007;109:2276–84.
Lee S, Challa-Malladi M, Bratton SB, Wright CW. Nuclear factor-κB-inducing kinase (NIK) contains an amino-terminal inhibitor of apoptosis (IAP)-binding motif (IBM) that potentiates NIK degradation by cellular IAP1 (c-IAP1). J Biol Chem. 2014;289:30680–9.
Takeuchi K, Soda M, Togashi Y, Suzuki R, Sakata S, Hatano S, et al. RET, ROS1 and ALK fusions in lung cancer. Nat Med. 2012;18:378–81.
Chapman MA, Lawrence MS, Keats JJ, Cibulskis K, Sougnez C, Schinzel AC, et al. Initial genome sequencing and analysis of multiple myeloma. Nature. 2011;471:467–72.
Mano H. The EML4-ALK oncogene: targeting an essential growth driver in human cancer. Proc Jpn Acad Ser B Phys Biol Sci. 2015;91:193–201.
Gridelli C, Peters S, Sgambato A, Casaluce F, Adjei AA, Ciardiello F. ALK inhibitors in the treatment of advanced NSCLC. Cancer Treat Rev. 2014;40:300–6.
Doebele RC, Pilling AB, Aisner DL, Kutateladze TG, Le AT, Weickhardt AJ, et al. Mechanisms of resistance to crizotinib in patients with ALK gene rearranged non-small cell lung cancer. Clin Cancer Res. 2012;18:1472–82.
Walker BA, Wardell CP, Melchor L, Brioli A, Johnson DC, Kaiser MF, et al. Intraclonal heterogeneity is a critical early event in the development of myeloma and precedes the development of clinical symptoms. Leukemia. 2014;28:384–90.
Walker BA, Wardell CP, Melchor L, Hulkki S, Potter NE, Johnson DC, et al. Intraclonal heterogeneity and distinct molecular mechanisms characterize the development of t(4;14) and t(11;14) myeloma. Blood. 2012;120:1077–86.
Rasche L, Chavan SS, Stephens OW, Patel PH, Tytarenko R, Ashby C, et al. Spatial genomic heterogeneity in multiple myeloma revealed by multi-region sequencing. Nat Commun. 2017;8:268.
Razani B, Reichardt AD, Cheng G. Non-canonical NF-κB signaling activation and regulation: principles and perspectives. Immunol Rev. 2011;244:44–54.
Sun SC. Non-canonical NF-κB signaling pathway. Cell Res. 2011;21:71–85.
Keats JJ, Fonseca R, Chesi M, Schop R, Baker A, Chng WJ, et al. Promiscuous mutations activate the noncanonical NF-κB pathway in multiple myeloma. Cancer Cell. 2007;12:131–44.
Walker BA, Leone PE, Chiecchio L, Dickens NJ, Jenner MW, Boyd KD, et al. A compendium of myeloma-associated chromosomal copy number abnormalities and their prognostic value. Blood. 2010;116:e56–65.
Annunziata CM, Davis RE, Demchenko Y, Bellamy W, Gabrea A, Zhan F, et al. Frequent engagement of the classical and alternative NF-κB pathways by diverse genetic abnormalities in multiple myeloma. Cancer Cell. 2007;12:115–30.
Acknowledgements
Funding support for the CoMMpass dataset was provided by the Myeloma Genome Project. The CoMMpass dataset was generated by the Multiple Myeloma Research Foundation in collaboration with the Multiple Myeloma Research Consortium.
Author contributions
GJM and BAW analyzed data and wrote the paper. JH, SZ, MB, SA, JR, VAM, PS, and TM analyzed data. OWS, RT, PP, and SD prepared samples and performed additional experiments. NW, CS, LR, ST, MZ, FvR, and FED provided input and patient material.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
JH, SZ, MB, SA, JR, VAM, PS, and TM are employees of Foundation Medicine and hold stocks and shares. The remaining authors declare that they have no conflict of interest..
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Morgan, G.J., He, J., Tytarenko, R. et al. Kinase domain activation through gene rearrangement in multiple myeloma. Leukemia 32, 2435–2444 (2018). https://doi.org/10.1038/s41375-018-0108-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41375-018-0108-y
- Springer Nature Limited
This article is cited by
-
Plasma cell myeloma: role of histopathology, immunophenotyping, and genetic testing
Skeletal Radiology (2022)
-
RNA-sequencing of myxoinflammatory fibroblastic sarcomas reveals a novel SND1::BRAF fusion and 3 different molecular aberrations with the potential to upregulate the TEAD1 gene including SEC23IP::VGLL3 and TEAD1::MRTFB gene fusions
Virchows Archiv (2022)
-
The genetic heterogeneity and drug resistance mechanisms of relapsed refractory multiple myeloma
Nature Communications (2022)
-
Integrative multi-omics identifies high risk multiple myeloma subgroup associated with significant DNA loss and dysregulated DNA repair and cell cycle pathways
BMC Medical Genomics (2021)
-
Non-secretory multiple myeloma with unusual TFG-ALK fusion showed dramatic response to ALK inhibition
npj Genomic Medicine (2021)