Abstract
Hepatic glucose production by glucagon is crucial for glucose homeostasis during fasting, yet the underlying mechanisms remain incompletely delineated. Although CD38 has been detected in the nucleus, its function in this compartment is unknown. Here, we demonstrate that nuclear CD38 (nCD38) controls glucagon-induced gluconeogenesis in primary hepatocytes and liver in a manner distinct from CD38 occurring in the cytoplasm and lysosomal compartments. We found that the localization of CD38 in the nucleus is required for glucose production by glucagon and that nCD38 activation requires NAD+ supplied by PKCδ-phosphorylated connexin 43. In fasting and diabetes, nCD38 promotes sustained Ca2+ signals via transient receptor potential melastatin 2 (TRPM2) activation by ADP-ribose, which enhances the transcription of glucose-6 phosphatase and phosphoenolpyruvate carboxykinase 1. These findings shed light on the role of nCD38 in glucagon-induced gluconeogenesis and provide insight into nuclear Ca2+ signals that mediate the transcription of key genes in gluconeogenesis under physiological conditions.
Similar content being viewed by others
Introduction
The liver is the primary organ in the maintenance of glucose homeostasis under fasting conditions1. During the early stages of fasting, glucagon secreted by pancreatic α-cells acts on hepatocytes to convert stored glycogen to glucose via glycogenolysis2,3. As fasting progresses, glucagon stimulates glucose production from noncarbohydrate precursors, such as amino acids or lactate, via gluconeogenesis, which then becomes the main contributor to hepatic glucose production1,4. In hepatic gluconeogenesis, glucose-6-phosphatase (G6Pase) and phosphoenolpyruvate carboxykinase 1 (Pck1) represent the key regulatory enzymes; their expression is closely regulated by Ca2+ signaling5,6. Glucagon increases cytosolic Ca2+ through the protein kinase-A (PKA)-mediated phosphorylation of the inositol 1,4,5-trisphosphate receptor (IP3R), leading to increased calcineurin activity and the subsequent dephosphorylation of the cAMP response element-binding protein (CREB) coactivator CRTC25. We have previously demonstrated that glucagon induces CD38-mediated cyclic ADP-ribose (cADPR) production and sustained increases in cytosolic Ca2+, which in turn induce nuclear translocation of CRTC and gluconeogenic gene expression6.
CD38 is a mammalian ADP-ribosyl cyclase that catalyzes the synthesis of multiple Ca2+-mobilizing messengers: cADPR, nicotinic acid adenine dinucleotide phosphate (NAADP), and ADP-ribose (ADPR)7. Although CD38 was initially identified as a type II transmembrane ectoenzyme in the plasma membrane, it was later revealed that CD38 exists in various intracellular organelles, including endosomes, lysosomes, and the nucleus8,9,10. The nuclear localization of CD38 has been demonstrated in a variety of cell types8,11,12. CD38 is assimilated in the inner nuclear membrane with its catalytic site within the nucleoplasm, suggesting that its product, cADPR, activates ryanodine receptors (RyR) on the inner nuclear membrane to cause an increase in nucleoplasmic Ca2+8. However, the specific characterization of this nuclear form of CD38 (nCD38) remains controversial, and the mechanism of its activation also remains unknown.
The nucleus has an autonomous Ca2+ signaling system that can generate its own Ca2+ transients that modulate processes such as gene transcription13. However, much remains unknown regarding how such spatially-distinct Ca2+ signals achieve specificity for gene transcription and how these nuclear Ca2+ signals are regulated. Numerous studies have demonstrated that the nucleus possesses the biochemical machinery required to produce Ca2+-mobilizing messengers13,14,15. These Ca2+-mobilizing messengers, IP3 and cADPR, stimulate the release of Ca2+ from the nuclear envelope (NE) into the nucleoplasm via IP3R and RyR located on the inner membrane of the nucleus16,17,18. Moreover, Gerasimenko et al.16 described ATP-dependent accumulation of Ca2+ in the NE, indicating that the NE serves as a “storage facility” for Ca2+ influx into the nucleoplasm. Together, these findings indicate that nuclear Ca2+ signaling operates independently of cytosolic signaling, although how these processes interact is not yet fully understood.
In this study, we investigated the role of nCD38 in glucagon-mediated Ca2+ signaling as well as hepatic gluconeogenic gene expression. Our findings support the view that nCD38 in hepatocytes produces a novel Ca2+ signaling messenger, ADPR, in response to glucagon, and thereby plays a critical role in glucagon-induced gluconeogenic gene expression. We provide evidence that ADPR production is regulated by the transfer of the substrate NAD+ into the perinuclear space from the nucleoplasm via connexin43 (Cx43). Cx43 is in turn regulated by protein kinase C (PKC) δ-mediated phosphorylation. ADPR targets the transient receptor potential melastatin 2 (TRPM2) channel to gate Ca2+ from the NE into the nucleoplasm. Finally, we present evidence that ADPR production is upregulated in diabetic animals and that treating diabetic animals with an ADPR antagonist mitigates the abnormally increased glucose levels. Our findings emphasize that ADPR-mediated nuclear Ca2+ signaling is essential for hepatic gluconeogenesis under both normal physiological conditions and in diabetes.
Materials and methods
Animals
CD38 knockout mice (Cd38−/−; B6.129P2-Cd38tm/Lud) and B6. BKS(D)-Leprdb/J (db/db) mice were purchased from Jackson Laboratory (Bar Harbor, ME). TRPM2 KO mice were kindly provided by Yasuo Mori (Kyoto University, Japan). All experimental animals were used under a protocol approved by the institutional animal care and use committee at Jeonbuk National University Medical School (CBU 2014-00031).
Reagents
Xestospongin C was obtained from Santa Cruz Biotechnology. Trans-Ned19, GF10923X, Go6976, rottlerin, and SKF96365 were obtained from Tocris Bioscience. 8-Br-ADPR and ara-2’-F-NAD were obtained from Biolog Life Science Institute. All other reagents were obtained from Sigma-Aldrich.
Adenoviral constructs
Generation of NLS-R GECO Adenovirus: NLS R GECO adenovirus was constructed using NLS R GECO plasmid (Addgene, 32462)19. Generation of NLS-CD38 and NLS-Flag Cx43 Adenovirus: CD38 or Cx43 was amplified via PCR from pCMV-CD38 (Sino Biological Inc., MG50191-UT) or pCMV3- Flag Cx43 (Sino Biological Inc., G52427-NF) by using primers to add NLS, which was inserted into the pCMV/myc/nuc vector (Thermo Fisher Scientific, V82120). NLS-CD38 E230D and NLS-Flag Cx43 S368A were constructed by the addition of NLS via site-directed mutagenesis. The resulting construct was subcloned into the pENTR D-TOPO vector (Thermo Fisher Scientific, K240020), after which a recombinant vector was generated using the pAd/CMV/V5-DEST adenoviral expression vector system (Life Technologies, 43-0200). Recombinant adenovirus was amplified in HEK293A cells (Thermo Fisher Scientific, R70507) and purified by cesium chloride gradient ultracentrifugation.
Primary hepatocyte culture and glucose production
Primary hepatocytes were isolated from 8- to 12-week-old male C57BL/6J mice or db/db mice as previously described20. Hepatocytes were transduced with adenoviral constructs 6 h after plating at a multiplicity of infection of 50 plaque-forming units per cell for 16 h, and experiments were performed 36 h after transduction. Glucose production assays were carried out as described21.
In vivo imaging
Seven-week-old male C57BL/6 J mice were intravenously injected with 1 × 109 p.f.u of CRE luciferase adenovirus (db/db male mice, 5 × 1010 p.f.u) from Vector Biolabs. After 3 days, 6 h-fasted mice were injected intraperitoneally with 100 μg/kg glucagon 1 h before imaging. Then, the mice were injected intraperitoneally with 150 mg/kg firefly d‐luciferin (LUCK-100, GoldBio, St Louis, MO, USA). After 10 min, mice were anesthetized and imaged at 15 min using the IVIS Luminar XR Imaging System (Caliper Life Sciences, Hopkinton, MA, USA).
Pyruvate tolerance test and glycerol tolerance test
Mice were fasted for 19 h and injected intraperitoneally with sodium pyruvate or glycerol (2 g/kg body weight). Tail vein blood was sampled for glucose measurement at the indicated time points after injection. Blood glucose values were determined using a LifeScan automatic glucometer.
Biochemical analysis
Plasma levels of insulin (ALPCO) and glucagon (MyBioSource), as well as glycogen content (BioAssay Systems), were assessed as per the manufacturer’s instructions.
Isolation of nuclei
Intact nuclei from hepatocytes were isolated as previously described17. To prepare the outer membrane of the nucleus and nucleoplast (outer membrane removed nuclei), 2% (w/v) sodium citrate was added to the nuclear suspension, incubated for 1 h on ice while stirring and centrifuged for 15 min at 1000 g. The supernatant contained the outer membrane of the nucleus, and the pellet contained the nucleoplast.
NAD glycohydrolase activity
NAD glycohydrolase activity was determined fluorometrically by using 1,N6-etheno-NAD as a substrate22.
siRNA transfection
Hepatocytes were transfected with 100 nM siRNA specific for TRPM2 (Genolution), PKCδ, (Ambion, s71696), PLCδ1 (Ambion, s71805), PLCδ3 (Ambion, s91013), or control siRNA (Ambion, 4390843) using Lipofectamine RNAi MAX reagent according to the manufacturer’s instructions. Transfected hepatocytes were cultured for 36 h before experiments. The siRNA sequences were as follows: TRPM2 siRNA-1, sense, 5’-GCACUCUGCAUACAAUCUAtt-3’, antisense, 5’-UAGAUUGUAUGCAGAGUGCgg-3’; TRPM2 siRNA-2, sense, 5’-GCGUCUUCACUGAGUGCUAtt-3’, antisense, 5’-UAGCACUCAGUGAAGACGCgg-3’; PKCδ siRNA, sense, 5’-GAUUCAAGGUUUAUAACUAtt-3’, antisense, 5’-UAGUUAUAAACCUUGAAUCgg-3’; PLCδ1 siRNA, sense, 5’-GCUACACUUUUACCUCUAAtt -3’, antisense, 5’-UUAGAGGUAAAAGUGUAGCca-3’; and PLCδ3 siRNA, sense, 5’-GAGUGAGGAUGGUCGAAUUtt-3’, antisense, 5’-AAUUCGACCAUCCUCACUCcg-3’.
Quantitative real-time PCR
Total cellular RNA was extracted from primary hepatocytes using the RNeasy kit (Qiagen, Valencia, CA). cDNA was synthesized by reverse transcription from 50 ng of total RNA using a cDNA synthesis kit (TaKaRa, RR037A). PCR was carried out on a 384-well plate using the ABI Prism 7900HT Sequence Detection System (Applied Biosystems) and SYBR Green Master Mix (Applied Biosystems, 4367659). Real-time PCR primers for mouse G6pc, Pck1, and GAPDH were as follows: G6pc (for, 5′-CGACTCGCTATCTCCAAGTGA-3′, and rev, 5′-GTTGAACCAGTCTCCGACCA-3′); Pck1 (for, 5′-AAGCATTCAACGCCAGGTTC-3′, and rev, 5′-GGGCGAGTCTGTCAGTTCAAT-3′); GAPDH (for, 5′-CGTCCCGTAGACAAAATGGT-3′, and rev, 5′-TTGATGGCAACAATCTCCAC-3′). GAPDH mRNA expression was quantified to normalize all data. Mouse β-Actin and PLC isoforms primers were as follows: β-Actin (for, 5′- AAGGCCAACCGTGAAAAGATGACC-3′, and rev, 5′-ACCGCTCGTTGCCAATAGTGA TGA-3′); PLCβ1 (for, 5′-AGACCTGGTGAACATTTCCCA-3′, and rev, 5′-ACAAGCCTCTAGTGCAGTTTC-3′); PLCβ2 (for, 5′-CTCAACCCTGTTCTATTGCCC-3′, and rev, 5′-TCGGATACTCGTGACATCCAG-3′); PLCβ3 (for, 5′-TGCCCAAGGACCCTAAGATTC-3′, and rev, 5′-GCTTCGTGTATGCTTTCCGC-3′); PLCβ4 (for, 5′-AGTGCTAGAATGTTCCCTCATCA-3′, and rev, 5′- GAAGCCGATATTCACCAGATCC-3′); PLCδ1 (for, 5′-CAAGGACCAGCGCAATACC-3′, and rev, 5′-CTTCCTGGCGTAGCTGTCATC-3′); PLCδ3 (for, 5′- GGCTACGGGCACTGAAGAAG-3′, and rev, 5′-GCTGCACGAAGAATATGTGCTT-3′); PLCδ4 (for, 5′-ATTCAAGACCTACTAGCCACTGA-3′, and rev, 5′- CTCCACCAGATAGCGCAACAA-3′); PLCγ1 (for, 5′-TCTCGGGACTTTGACCGCTA-3′, and rev, 5′-CTCTCGGTTACGATCCACTGA-3′); PLCγ2 (for, 5′-GTGGACACCCTTCCAGAATATG-3′, and rev, 5′-ACCTGCCGAGTCTCCATGAT-3′).
Nuclear and cytoplasmic protein extraction
Nuclear and cytoplasmic proteins from hepatocytes or liver tissue were extracted using the NE-PER Nuclear and Cytoplasmic Extraction Kit (Thermo Fisher Scientific, 78833) according to the manufacturer’s instructions.
Immunoblotting
Cells were lysed in RIPA buffer (50 mM Tris–HCl, pH 7.4, 150 mM NaCl, 1% sodium deoxycholate, 0.1% SDS, and 1% NP 40 supplemented with phosphatase and protease inhibitor cocktail, Roche). Lysates were boiled in Laemmli sample buffer for 10 min, separated by SDS-polyacrylamide gel electrophoresis, transferred to PVDF membranes, and probed with primary antibodies. We used antibodies for the following proteins: pCREB (Ser133) (Cell Signaling, 9198), pCaMKII (Cell Signaling, 3361), pCaMKIV (Santa Cruz, sc-28443-R), CREB (Cell Signaling, 9197), CaMKII (Cell Signaling, 3362), CaMKIV (Cell Signaling, 4032), PARP-1 (Santa Cruz, sc-53643), Actin (Merck Millipore, MAB1501), pCx43(Ser368) (Sigma-Aldrich, SAB4300504), Cx43 (Cell Signaling, 3512), Flag (Sigma-Aldrich, F7425), Lamin B1 (Cell Signaling, 12586), PKCδ (Cell Signaling, 9616), TRPM2 (Novus, NB110-81601), Na+K+-ATPase (Novus, NB300-146), Calregulin (Santa Cruz, sc-7431), Nesprin 3 (MyBioSource, MBS2535184), Myc (Invitrogen, 46-0603), CD38 (Santa Cruz, sc-7049), PLCβ1 (Santa Cruz, sc-5291), PLCβ3 (Santa Cruz, sc-133231), PLCβ4 (Santa Cruz, sc-404), PLCδ1 (Santa Cruz, sc-365811), PLCδ3 (Santa Cruz, sc-514912), PLCδ4 (Santa Cruz, sc-373875), PLCγ1 (Santa Cruz, sc-7290), PLCγ2 (Santa Cruz, sc-5283), HSP90 (Cell Signaling, 4874), GAPDH (Santa Cruz, sc-166574), and LAMP-1 (BD Biosciences, 553792). After incubation with secondary antibodies conjugated with horseradish peroxidase (Cell Signaling), chemiluminescence was detected by using the LAS4000 system (GE Healthcare). Western blot densitometric quantification was performed using ImageJ (NIH, Bethesda, MD).
Immunofluorescence
Hepatocytes were grown on collagen-coated confocal dishes and fixed with 3.8% paraformaldehyde for 20 min, after which they were washed three times with ice-cold PBS. To permeabilize, cells and nuclei were treated with 0.25% Triton X-100 in PBS for 10 min. After blocking with 3% BSA, 0.25% Triton X-100, and PBS at RT for 1 h, samples were incubated overnight at 4 °C with the indicated antibodies: TRPM2 (Novus, NB110-81601), CD38 (Thermo Fisher Scientific, 14-0381-85), PKCδ (Santa Cruz, sc-8402), Cx43 (Santa Cruz, sc-13558), PLCδ1 (Santa Cruz, sc-365811), PLCδ3 (Santa Cruz, sc-514912), or Lamin B1 (Santa Cruz, sc-6216). Alexa Fluor-conjugated secondary antibodies (546 donkey anti-rabbit antibody, Thermo Fisher Scientific, A10040; 555 donkey anti-goat antibody, Thermo Fisher Scientific, A-21432; 488 donkey anti-rat antibody, Thermo Fisher Scientific, A-21208; 488 donkey anti-mouse antibody, Thermo Fisher Scientific, A-21202; or 488 donkey anti-rabbit antibody, Thermo Fisher Scientific, A-21206) were incubated at 1:500 dilutions in the presence of 1% BSA at RT for 1 h. The nuclei were stained with DAPI (Thermo Fisher Scientific, 62248). Cells and nuclei were visualized with a Zeiss LSM510 Axiovert 200 M laser-scanning confocal microscope.
Measurement of [Ca2+]i
Hepatocytes infected with adenovirus regarding GCaMP6m (Vector Biolabs, 1909) and NLS-R-GECO for 24 h were subsequently serum-starved for 16 h in Medium 199, after which changes in [Ca2+]i were determined at 488 nm excitation/530 nm emission and 543 nm excitation/560 nm emission using a confocal microscope (Nikon, Japan). Isolated nuclei were loaded as reported23,24. The membrane-impermeant Ca2+ dye Calcium Green Dextran (Thermo Fisher Scientific, C3713) was loaded into the nucleoplasm, while the membrane permeant Ca2+ dye Fluo-4 AM (Thermo Fisher Scientific, F14201) was loaded into the nuclear envelope. Ca2+ imaging in single isolated nuclei was performed using Nikon confocal microscopy.
Measurement of NAD, [ADPR]i, and [cADPR]i
NAD and ADPR levels were measured using LC-MS/MS as described previously25, and cADPR was measured by the cycling method described previously26.
Statistical analysis
Statistical analyses were performed using GraphPad Prism 8. Data were analyzed by Student’s unpaired t test and one-way ANOVA followed by Tukey’s multiple comparison test. Data are represented as the mean ± standard error of the mean (SEM). The number of independent experiments and information about the statistical details and methods are shown in the relevant figure legends. A value of P < 0.05 was considered significant.
Results
Gluconeogenesis by glucagon is dependent on nuclear CD38
CD38 is ubiquitously expressed in various cellular organelles, including the nucleus8,27. Although nCD38 has been found to regulate nuclear Ca2+ homeostasis8,27, the role of nCD38 in physiological functions remains unknown. First, we verified the existence of CD38 in the nucleus of primary hepatocytes. Confocal microscopy using anti-CD38 antisera revealed perinuclear immunofluorescence with ring-like labeling around the nucleus, which co-localized with Lamin B1 (Fig. 1a). Furthermore, CD38 was primarily located in the inner membrane of the nucleus but not the outer nuclear membrane (Supplementary Fig. 1a, b). In addition, nCD38 was detected in primary hepatocytes isolated from CD38 wild-type (Cd38+/+) but not CD38 knockout (Cd38−/−) mice (Fig. 1b). To further investigate the role of nCD38 in glucagon-induced hepatic gluconeogenesis, we overexpressed nCD38 in primary hepatocytes using adenovirus encoding a nuclear localizing signal (NLS)-fused CD38 (Ad-wtCd38-NLS). We also constructed an adenovirus encoding an NLS-fused catalytically inactive mutant CD38E230D (Ad-Cd38E230D-NLS) as a negative control (Fig. 1c). Western blot analyses revealed the expression of our constructs only in the nuclear fractions (Fig. 1d). CD38 overexpression from Ad-wtCd38-NLS resulted in robust NADase activity, whereas Ad-Cd38E230D-NLS-transfected cells did not exhibit any significant change in NADase activity compared to control Ad-NLS overexpressing cells (Fig. 1e). nCD38 overexpression led to further increases in ADPR production in response to glucagon relative to control Ad-NLS infected cells, which were not evident in cells overexpressing mutant nCD38 (Fig. 1f). However, nCD38 overexpression did not further increase cADPR production in response to glucagon (Supplementary Fig. 1d). In addition, to test the glucagon-induced ADPR increase in the nucleus, we measured the ADPR level in the nucleoplast, a nucleus stripped of its outer membrane, after treating nCD38-overexpressed primary hepatocytes with glucagon. ADPR production was increased in the nucleoplast of nCD38-overexpressed hepatocytes in response to glucagon when compared to the control Ad-NLS infected cells (Supplementary Fig. 1e). Next, to determine the function of nCD38 in hepatic glucose metabolism, the expression of the G6pc and Pck1 genes encoding G6Pase and phosphoenolpyruvate carboxykinase 1, which are the major rate-limiting enzymes for the glucose-generating pathway28, was analyzed after glucagon treatment of hepatocytes. The increase in nCD38 by transfection with Ad-wtCd38-NLS enhanced the levels of G6pc and Pck1 mRNA in the presence of glucagon relative to control Ad-NLS expressing cells, while Ad-Cd38E230D-NLS expression did not elicit this response (Fig. 1g, h). Consistently, the increased expression of nCD38, but not mutated nCD38, significantly promoted glucose production relative to control Ad-NLS expressing cells (Fig. 1i). Furthermore, we confirmed that the overexpression of nCD38 in Cd38−/− hepatocytes via Ad-wtCd38-NLS significantly increased G6pc and Pck1 mRNA levels in response to glucagon (Supplementary Fig. 1f, g). Therefore, these findings suggest that nCD38 may be involved in the regulation of gluconeogenesis-related gene expression and glucose metabolism.
a After isolating nuclei with sucrose ultracentrifugation, the outer membrane of the nuclei was separated by treatment with sodium citrate. Nucleoplasts were fixed with 3.8% paraformaldehyde for 20 min, permeabilized, and then stained with anti-mouse CD38 antibody (green). Lamin B1 was used as a marker for the inner membrane of the nucleus. The nucleus was stained with DAPI (blue). Scale bar, 5 μm. b After nuclei were isolated from Cd38+/+ or Cd38−/− mouse primary hepatocytes, the outer membrane of the nucleus was removed by sodium citrate treatment. The nucleoplasts (outer membrane-deprived nuclei) were stained with an anti-mouse CD38 antibody (C90). Scale bar, 5 μm. c Adenoviral vector constructs of Control (Ad-NLS), catalytic active-nCD38 (wtCd38), and catalytic inactive-nCD38 (Cd38E230D). d Hepatocytes were transduced with adenoviral vectors expressing Ad-NLS, wtCd38-NLS, or Cd38E230D-NLS at an MOI of 50 for 24 h, and then cytoplasmic and nuclear extracts were isolated. Immunoblotting was performed to detect Myc, LAMP-1 (a marker of lysosomes), GAPDH (a cytoplasmic marker), and Lamin B1 (a nuclear marker). e NAD glycohydrolase activity. f ADPR levels in hepatocytes treated with glucagon (100 nM) for 30 sec. g, h qRT-PCR of G6pc (g) and Pck1 (h) after incubation with glucagon (100 nM) for 4 h. i Glucose production after incubation with glucagon (100 nM) for 5 h. e–i n = 6 independent experiments, mean values ± SEMs are shown, and P values were calculated by one-way ANOVA followed by Tukey’s multiple comparison test. **P < 0.01, ***P < 0.001, and not significant (NS). For a, b, d, representative images of three independent experiments are shown.
CD38 activation in the nucleus requires Cx43 phosphorylation by PKCδ
Given that nCD38 is constitutively active and exists in a type II orientation on the inner membrane of the nucleus (Fig. 1b), we assumed that the regulation of nCD38 activity in the perinuclear space occurred through substrate availability. To determine whether Cx43, a known NAD+ transporter29, was involved in glucagon-induced nuclear Ca2+ signaling, we examined the effect of the Cx43 inhibitor oleamide on glucagon-induced ADPR production. Oleamide abolished glucagon-induced ADPR production as well as G6pc and Pck1 mRNA expression levels (Fig. 2a, b).
a Glucagon-induced ADPR production after preincubation with oleamide (50 μM) for 30 min. n = 6 independent experiments. ***P < 0.001 and not significant (NS). b Effect of oleamide on glucagon-induced mRNA expression of G6pc and Pck1. n = 6 independent experiments. **P < 0.01, ***P < 0.001. c After isolating nuclei, nucleoplasts were stained with anti-Cx43 antibody (green). Lamin B1 was used as a marker for the inner membrane of the nucleus. The nucleus was stained with DAPI (blue). Scale bar, 5 μm. d The bar graph represents the localization of Cx43. n = 6 independent experiments. ***P < 0.001. e Hepatocytes were transduced with adenoviral vectors expressing Ad-Cx43-NLS or Ad-Cx43S368A-NLS at an MOI of 50 for 24 h and then assayed for immunoblot analysis for Ad-NLS, Ad-Cx43-NLS, or Ad-Cx43S368A-NLS and qRT-PCR of G6pc and Pck1 in hepatocytes infected with Cx43 WT or S368A before and after treatment with glucagon. n = 8 independent experiments. **P < 0.01, ***P < 0.001. f, g Blood glucose levels during fasting (f) and after pyruvate challenge (g) in mice after treatment with Ad-NLS, Ad-Cx43-NLS, or Ad-Cx43S368A-NLS. n = 10 mice per group. *P < 0.05, **P < 0.01, ***P < 0.001, #P < 0.05; Ad-NLS vs. Ad-Cx43-NLS, ##P < 0.001; Ad-Cx43-NLS vs. Ad-Cx43S368A-NLS. h Nucleoplasts were stained with anti-PKCδ antibody (green). Lamin B1 was used as a marker for the inner membrane of the nucleus. The nucleus was stained with DAPI (blue). Scale bar, 5 μm. i Glucagon-induced G6pc and Pck1 mRNA levels. GF109203X (1 μM), Go6976 (1 μM), or rottlerin (3 μM) was preincubated for 30 min. n = 6 independent experiments. **P < 0.01, ***P < 0.001. j Effects of PKCδ KD on glucagon-induced G6pc and Pck1 gene expression. Immunoblotting of PKCδ and β-actin (upper). n = 6 independent experiments. **P < 0.01, ***P < 0.001. k Nucleoplasts were incubated with OAG for the indicated times. Immunoblotting for pCx43 (S368), Cx43, and Lamin B1. l NAD and ADPR levels after nucleoplasts were incubated with OAG (100 μM) for 30 sec. Ara-2’-F-NAD (200 nM) was preincubated for 30 min. n = 6 independent experiments. ***P < 0.001. m NAD and ADPR levels in PKCδ-knockdown nucleoplasts. OAG (100 μM) was incubated for 30 sec. Immunoblotting of PKCδ and Lamin B1 (upper). n = 6 independent experiments. **P < 0.01, ***P < 0.001. Data are represented as the mean ± SEM. Statistics were determined by one-way ANOVA followed by Tukey’s multiple comparison test except d where unpaired t test was used. For confocal images and immunoblotting, representative images of three independent experiments are shown.
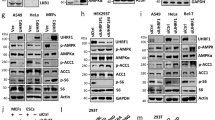
Cx43 is regulated through Ser368 phosphorylation via PKC30. We explored whether Ser368 of Cx43 is phosphorylated during glucagon signaling in hepatocytes. Glucagon-induced Ser368 phosphorylation occurred as early as 1 min after stimulation (Supplementary Fig. 2a), which was also observed in isolated nuclei (Supplementary Fig. 2b). In addition, using confocal analysis, we confirmed the existence of Cx43 in the inner membrane of the nucleus (Fig. 2c, d and Supplementary Fig. 1c). To confirm the importance of Ser368 phosphorylation in glucagon signaling, we generated an adenovirus encoding an NLS-fused Cx43S368A mutant (Supplementary Fig. 2c). Western blot analyses revealed the expression of our constructs only in the nuclear fractions (Fig. 2e). Overexpression of nuclear-targeting Cx43 S368A in hepatocytes decreased glucagon-induced gluconeogenic gene expression levels compared to overexpression of wild-type nuclear-targeting Cx43 (Fig. 2e). Furthermore, the overexpression of nuclear-targeting Cx43S368A in mice via adenovirus resulted in decreased blood glucose levels in both the fasting state and in response to a pyruvate or glycerol challenge test when compared to levels from mice expressing wild-type Cx43 (Fig. 2f, g and Supplementary Fig. 2d). Western blot analyses of liver tissues revealed the expression of our constructs only in the nuclear fractions (Supplementary Fig. 2e). As described above, body weight, food intake, plasma insulin, and plasma glucagon showed minimal differences between the control and experimental groups (Supplementary Fig. 2f). In line with blood glucose data, hepatic G6pc and Pck1 mRNA expression levels were lower in mice injected with the nuclear-targeting Cx43 S368A adenovirus (Supplementary Fig. 2g).
Because nuclear Cx43 inhibition resulted in a reduced response to glucagon in hepatic G6pc and Pck1 mRNA expression, we compared liver glycogen content and found that the overexpression of nuclear-targeting Cx43S368A in mice resulted in significantly higher liver glycogen content, as opposed to mice expressing nuclear-targeting wild-type Cx43 (Supplementary Fig. 2h). These data further indicate that phosphorylation of Serine 368 in nuclear Cx43 affects plasma glucose levels, the conversion of pyruvate into glucose, and the gene expression of gluconeogenic enzymes.
As PKCδ is responsible for Ser368 phosphorylation31, we tested whether PKCδ was involved in glucagon-induced signaling. First, we confirmed that PKCδ is located in the inner membrane of the nucleus (Fig. 2h). Subsequently, we found that glucagon-induced Pck1 mRNA expression was inhibited by both a broad-range PKC inhibitor, GF109203X, and the PKCδ inhibitor rottlerin but not by the PKCα/PKCβ inhibitor, Go6976 (Fig. 2i). PKCδ involvement in glucagon-induced G6pc and Pck1 mRNA expression levels was confirmed by PKCδ knockdown (KD) experiments (Fig. 2j). Furthermore, we measured NAD+ and ADPR levels in the nucleoplast preparation before and after treatment with diacylglycerol (DAG) to activate Cx43. Treating the nucleoplast with 1-oleoyl-2-acetyl-sn-glycerol (OAG), a synthetic diacylglycerol analog that induces Cx43 phosphorylation, resulted in reduced NAD+ levels while increasing ADPR levels (Fig. 2k, l). Pretreatment with ara-F-NAD, a membrane-impermeable CD38 inhibitor, significantly inhibited OAG-induced ADPR production (Fig. 2l). PKCδ-KD nucleoplasts showed a significant decrease in OAG-induced ADPR production and an increase in NAD levels compared with control nucleoplasts (Fig. 2m). Based on these results, we concluded that Cx43 plays a role in regulating NAD+ transport to CD38, the catalytic site of which faces the perinuclear space.
Nuclear CD38-mediated ADPR is required for the increase in nuclear Ca2+
Previously we revealed that CD38 is involved in glucagon-induced cytosolic Ca2+ signaling in hepatocytes and produces a Ca2+ second messenger, cADPR6. In addition, oleamide inhibited glucagon-induced cADPR production (Supplementary Fig. 2i), which suggests its regulation by Cx43. As nCD38 can also produce Ca2+ second messengers through NAD+ glycohydrolase activity, we investigated whether nuclear Ca2+ signals are increased by nCD38 in glucagon-treated primary hepatocytes. To simultaneously measure the Ca2+ changes in hepatocyte cytosol and nucleus after glucagon treatment, we infected hepatocytes with adenovirus carrying the genetically-encoded fluorescent Ca2+ indicators, GCaMP6m and NLS-R-GECO. Increases in nuclear Ca2+ concentrations ([Ca2+]nu) lagged a few seconds behind glucagon treatment when compared to cytosolic Ca2+ levels, which increased immediately following exposure to glucagon, indicating that nuclear Ca2+ signals follow Ca2+ fluctuations in the cytosol (Fig. 3a, b). When cytosolic Ca2+ was chelated by pretreatment with BAPTA-AM, the glucagon-induced increase in [Ca2+]nu was completely ablated (Fig. 3c), reinforcing the idea that nuclear Ca2+ signals are dependent on their cytosolic counterparts. As CD38-mediated cytosolic Ca2+ signals are involved in glucagon-induced gluconeogenesis in hepatocytes6, we also examined the possibility that nuclear Ca2+ signals may be dependent on CD38. Unlike primary hepatocytes of Cd38+/+ mice, glucagon-induced sustained Ca2+ signals in the nucleus as well as in the cytosol were absent in hepatocytes from Cd38−/− mice (Fig. 3b). Pretreating Cd38−/−primary hepatocytes with xestospongin C (XeC; an inhibitor of IP3R) completely abolished glucagon-induced Ca2+ signals, including the initial signals, in the cytosol and the nucleus, but XeC only inhibited the sustained Ca2+ signal and not the initial Ca2+ signal in both the cytosol and nucleus in Cd38+/+ primary hepatocytes (Supplementary Fig. 3a). These findings indicate that glucagon-induced Ca2+ signals in both compartments are dependent on IP3 and Ca2+ second messenger(s) generated by CD38. We then sought to determine which Ca2+ second messengers are involved in glucagon-induced Ca2+ changes by using various antagonistic analogs. Glucagon-induced sustained Ca2+ signals in the cytosol and the nucleus were blocked by treatment with either XeC or 8-Br-cADPR (an antagonistic analog of cADPR) (Supplementary Fig. 3a, d), suggesting that IP3 and cADPR act as the first messengers in the initial Ca2+ signaling in both the cytosol and the nucleus of hepatocytes in response to glucagon treatment. These findings also indicate that both IP3- and cADPR-mediated Ca2+ signals are prerequisites for these glucagon-induced sustained Ca2+ signals. Intriguingly, 8-Br-ADPR (an antagonistic ADPR analog) only inhibited sustained Ca2+ signals in the nucleus (Fig. 3e). Ned19 (an NAADP antagonist) had no effect on glucagon-induced Ca2+ signaling in either the cytosol or the nucleus (Fig. 3f). These findings indicate that initial Ca2+ signals in the cytosol and the nucleus are dependent on IP3 and cADPR and that ADPR plays a role in the later phase of Ca2+ signaling in the nucleus.
a, b Hepatocytes were infected with GCaMP6m and NLS-R-GECO adenoviruses, and Ca2+ signals were recorded. The time point where 100 nM glucagon (Gcg) is added is indicated by the arrow. Scale bar, 10 μm. c Cytosolic and nuclear Ca2+ responses to glucagon after preincubation with BAPTA-am (10 μM). d–f Cytosolic and nuclear Ca2+ responses to glucagon after incubation with various inhibitors of Ca2+ second messengers [8-Br-cADPR (100 μM, d), 8-Br-ADPR (100 μM, e), or Ned19 (10 μM, f)]. g, h Cytosolic and nuclear Ca2+ responses to glucagon after overexpression of nCD38 using wtCd38-NLS adenovirus in Cd38+/+ hepatocytes (g) or Cd38−/− hepatocytes (h). n = 20 cells for each condition in b–h.
Given that sustained Ca2+ signals in the cytosol and the nucleus were abolished by an IP3R blocker and cADPR antagonist (Supplementary Fig. 3a, d), we inferred that sustained Ca2+ signals require extracellular Ca2+ influx, which may be mediated through store-operated Ca2+ entry (SOCE) following Ca2+ store depletion by both IP3 and cADPR. As expected, glucagon-induced, sustained nuclear, and cytosolic Ca2+ signals were completely abolished under Ca2+-free conditions and by the SOCE inhibitor SKF 96365 (Supplementary Fig. 3b, c). These results suggest that both IP3 and cADPR-mediated cytosolic Ca2+ signals induce SOCE, leading to ADPR-mediated nuclear sustained Ca2+ signals.
To further examine the roles of nCD38 in glucagon-induced Ca2+ signaling, we measured Ca2+ changes in nCD38-overexpressing Cd38+/+ hepatocytes or Cd38−/− hepatocytes. Overexpression of nCD38 increased glucagon-mediated nuclear sustained Ca2+ signals but not cytosolic Ca2+ signals in Cd38+/+ hepatocytes or Cd38−/− hepatocytes (Fig. 3g, h). These results demonstrate that nCD38-produced ADPR is essential for glucagon-induced nuclear sustained Ca2+ signals.
Nuclear Ca2+ influx requires nuclear TRPM2 activated by ADPR
ADPR activates TRPM2, a Ca2+-permeable nonselective cation channel, to elicit Ca2+ influx or Ca2+ release from intracellular stores32,33,34. We examined whether TRPM2 was located in the nucleus of primary hepatocytes. Confocal microscopy revealed perinuclear immunofluorescence with ring-like labeling around the nucleus (Fig. 4a). TRPM2 was also detected in both nuclear and cytoplasmic fractions (Fig. 4b). Specifically, the TRPM2 channel is likely localized to the inner membrane, but not the outer membrane, of the nucleus (Fig. 4c and Supplementary Fig. 1c). We further examined whether glucagon-induced nuclear Ca2+ signals were involved with the TRPM2 channel and perinuclear Ca2+ stores. To this end, we loaded hepatocyte nuclei with either the membrane-permeable Fluo-4 AM Ca2+ probe to measure Ca2+ in the perinuclear region or membrane-impermeable Calcium Green Dextran to measure Ca2+ in the nucleoplasm18,23. Intriguingly, confocal microscopy revealed that ADPR-induced reciprocal Ca2+ exchanges between the perinuclear area and the nucleoplasm, showing a decrease in perinuclear Ca2+ concentration and an increase in nucleoplasm Ca2+ concentration (Fig. 4d, e). The addition of ADPR led to decreased perinuclear Ca2+ levels, which correlated with an increase in nucleoplasm Ca2+. This is consistent with the previous finding that the perinuclear envelope serves as a Ca2+ store for the nucleoplasm23. 8-Br-ADPR completely blocked ADPR-induced Ca2+ exchange between the two spaces (Fig. 4d, e). Moreover, ADPR failed to induce Ca2+ efflux in hepatocytes from TRPM2 knockout (KO) mice (Fig. 4d, e). These findings suggest that ADPR activates the TRPM2 channel to induce Ca2+ efflux from perinuclear Ca2+ stores into the nucleoplasm to be utilized for nuclear Ca2+ signals in primary hepatocytes. These findings were corroborated by TRPM2 KD experiments using TRPM2-targeting siRNAs, showing that TRPM2 KD blocks ADPR-induced Ca2+ flux, as well as gluconeogenic gene transcription and glucose production (Fig. 4f–j). Furthermore, we measured both cytosol and nuclear Ca2+ changes in TRPM2 KO hepatocytes in response to glucagon. TRPM2 KO hepatocytes showed inhibition of nuclear Ca2+ signaling but not cytosolic Ca2+ signaling in response to glucagon compare to TRPM2 WT hepatocytes (Fig. 4k). These results indicate that TRPM2 is specifically involved in glucagon-induced nuclear Ca2+ signaling for hepatocyte gluconeogenesis, although TRPM2 is located in the lysosome.
a Nucleoplasts were stained with anti-TRPM2 antibody (green). Lamin B1 was used as a marker for the inner membrane of the nucleus. The nuclei were stained with DAPI (blue). Scale bar, 5 μm. b Hepatocytes were separated into the cytoplasm and nucleus. TRPM2 localization was analyzed by immunoblot using antibodies against each marker: plasma membrane (Na+K+-ATPase), nucleus (Lamin B1), and endoplasmic reticulum (Calregulin). c Immunoblot for TRPM2, Nesprin3 (a marker for the outer membrane of the nucleus), and Lamin B1 (a marker for the inner membrane of the nucleus). d, e Changes in perinuclear Ca2+ levels were measured with Fluo-4 AM (d), and changes in the nucleoplasm Ca2+ levels were measured with Calcium Green dextran, a membrane-impermeant Ca2+ dye (e) after preincubation with 8-Br-ADPR (100 μM) for 30 min before treatment with ADPR (100 μM) in intact nuclei isolated from WT or TRPM2 KO hepatocytes. n = 5 nuclei for each condition. Scale bar, 5 μm. f Immunoblotting of TRPM2 expression after TRPM2 knockdown by siRNA. g, h Isolated nuclei after TRPM2 KD were measured for changes in perinuclear Ca2+ levels (g) and in nucleoplasm Ca2+ levels (h). n = 5 nuclei for each condition. i, j Effects of TRPM2 KD on glucagon-induced G6pc and Pck1 gene expression (i) and glucose production (j). n = 6 independent experiments. **P < 0.01, ***P < 0.001. k Cytosolic and nuclear Ca2+ responses to glucagon in TRPM2 WT hepatocytes or TRPM2 KO hepatocytes. n = 20 cells for each condition in k. The time point where 100 nM glucagon (Gcg) was added is indicated by the arrow. Data are represented as the mean ± SEM. Statistics were determined by one-way ANOVA followed by Tukey’s multiple comparison test. For a–c, and f representative images of three independent experiments are shown.
Nuclear Ca2+ increased by CD38/ADPR promotes gluconeogenesis via the CaMKII/CaMKIV/CREB pathway
In accordance with the data demonstrating ADPR as a potential messenger in nuclear Ca2+ signals, ADPR levels were increased in hepatocytes from Cd38+/+ mice at 20 sec in response to glucagon treatment (Fig. 5a), which correlates with the time course of glucagon-induced nuclear Ca2+ increase (Fig. 3a, b). As expected, hepatocytes from Cd38−/− mice failed to produce ADPR in response to glucagon. Moreover, basal ADPR levels in hepatocytes from Cd38−/− mice were also significantly lower than those in hepatocytes from Cd38+/+ mice (Fig. 5a). These findings indicate that CD38 is responsible for the glucagon-induced production of ADPR in hepatocytes. Pretreatment with 8-Br-cADPR completely blocked glucagon-induced ADPR production, while XeC only partially blocked ADPR production (Supplementary Fig. 4a, b). Thus, we tested whether 8-Br-cADPR inhibits glucagon-induced Cx43 phosphorylation, and as expected, it abolished glucagon-induced Cx43 phosphorylation (Supplementary Fig. 4c). Given that SOCE following IP3 and cADPR-mediated Ca2+ signals is critical for sustained Ca2+ signals and that 8-Br-ADPR blocks sustained nuclear Ca2+ signals (Fig. 3f), we considered the possibility that glucagon-induced ADPR production may require extracellular Ca2+ influx. This was confirmed by the observation that glucagon-induced ADPR production was blocked by SKF 96365 and extracellular Ca2+ deprived conditions (Fig. 5b). In contrast, glucagon-induced cADPR production was unaffected by SKF 96365 and extracellular Ca2+ deprivation (Supplementary Fig. 4d). In addition, pretreatment with SKF 96365 or extracellular Ca2+ free conditions inhibited the mRNA expression of G6pc and Pck1 in response to glucagon (Fig. 5c, d). These results indicate that ADPR production by nCD38 requires SOCE, through which sustained nuclear Ca2+ signals, essential to glucagon-induced hepatic gluconeogenic gene expression, are enabled. Based on these results, we tested the effects of 8-Br-ADPR on gluconeogenesis. Pretreatment with 8-Br-ADPR significantly inhibited glucagon-induced gluconeogenic gene expression, as well as glucose production (Fig. 5e, f). We then tested whether exogenous ADPR increases gluconeogenic gene expression. Treating intact hepatocytes with ADPR increased gluconeogenic gene expression in a dose-dependent manner (Fig. 5g). Exogenous ADPR also increased glucose production (Supplementary Fig. 4e). Moreover, exogenous ADPR-induced gluconeogenic gene expression was unaffected by SKF 96365 and extracellular Ca2+ deprivation (Supplementary Fig. 4f). This result suggests that ADPR-mediated signaling is downstream of SOCE in gluconeogenic gene expression. Since CaMKII/CREB phosphorylation is required for hepatic gluconeogenesis in response to glucagon and nuclear Ca2+ signals induce the phosphorylation of CaMKIV/CREB in various cells24,35,36, we investigated whether ADPR-mediated nuclear Ca2+ signals were involved in CaMKII/CaMKIV/CREB phosphorylation in glucagon-treated hepatocytes. CaMKII/CaMKIV/CREB phosphorylation was induced as early as 5 min following glucagon treatment and was sustained until 30 min, which was inhibited by pretreatment with 8-Br-ADPR (Fig. 5h). Moreover, 8-Br-ADPR significantly inhibited glucagon-induced CaMKII/CaMKIV/CREB phosphorylation in the nucleus but not in the cytosol (Fig. 5i). Furthermore, the administration of 8-Br-ADPR reduced CRE luciferase activity as well as G6pc and Pck1 mRNA expression levels in fasting mice, resulting in decreased blood glucose levels in both the fasting state and in response to a pyruvate challenge when compared with the control group (Fig. 5j–l and Supplementary Fig. 4g). However, the administration of 8-Br-ADPR to mice resulted in a decrease in body weight, white adipose tissue, plasma insulin, and hepatic glycogen content, with no change in plasma glucagon levels under fasting conditions (Supplementary Fig. 4h–k). Glucagon-induced cytosolic Ca2+ increase activates CaMKII to promote FoxO1 nuclear translocation through p38α, resulting in hepatic gluconeogenesis36. Because the inhibition of nuclear Ca2+ signaling by 8-Br-ADPR prevented the phosphorylation of CaMKII (Fig. 5h, i), we further examined whether glucagon-induced nuclear Ca2+ signaling by ADPR is involved in FoxO1 nuclear translocation. 8-Br-ADPR did not affect either glucagon-induced FoxO1 nuclear translocation or FoxO1 expression (Supplementary Fig. 5a, b). Because glucagon increased the expression of FoxO1, we further examined the effect of 8-Br-ADPR on glucagon-induced nuclear translocation of FoxO1 from the cytosol in the presence of cycloheximide, an inhibitor of protein synthesis. Although the inhibition of glucagon-induced nuclear Ca2+ signaling by 8-Br-ADPR prevented the phosphorylation of CaMKII (Fig. 5h, i), 8-Br-ADPR did not affect FoxO1 nuclear translocation (Supplementary Fig. 5c, d). Taken together, these data suggest that glucagon stimulates gluconeogenesis through nuclear Ca2+ signals increased by nuclear CD38/ADPR activity.
a Glucagon-induced ADPR production in Cd38+/+ or Cd38−/− primary hepatocytes. n = 6 independent experiments. **P < 0.01; Cd38+/+ vs. Cd38+/+. b ADPR levels in hepatocytes treated with glucagon (100 nM) for 30 sec after preincubation with vehicle or SKF 96365 (SKF, 50 μM), or under Ca2+-deprived extracellular conditions. n = 6 independent experiments. **P < 0.01. c, d Effects of SOCE on glucagon-induced mRNA levels of G6pc (c) and Pck1 (d). n = 8 independent experiments. ***P < 0.001. e, f Effects of 8-Br-ADPR on glucagon-induced mRNA levels of G6pc (e, left) and Pck1 (e, right) and glucose production (f). 8-Br-ADPR (100 μM) was preincubated for 30 min. n = 6 independent experiments. **P < 0.01, ***P < 0.001. g Effects of exogenous ADPR on G6pc and Pck1 gene expression. n = 6 independent experiments. ***P < 0.001. h Immunoblotting for pCREB, pCaMKII, pCaMKIV, and β-actin in total cell lysates. i Immunoblotting cytoplasmic and nuclear extracts after treating hepatocytes with glucagon for 30 min. Cells were preincubated with 8-Br-ADPR (100 μM) for 30 min. The PARP-1 antibody was used as a marker of nuclear extracts. j, k Effects of 8-Br-ADPR on CRE luciferase activity in fasting mice (j, n = 3 mice per group) and the pyruvate tolerance test (k, n = 5 mice per group). 8-Br-ADPR (32 mg/kg) was intravenously administered to mice. **P < 0.01 and ***P < 0.001; vehicle vs. 8-Br-ADPR. l Effects of 8-Br-ADPR on G6pc and Pck1 mRNA levels in fasting mice. n = 8 mice per group. ***P < 0.001. Data are represented as the mean ± SEM. Statistics were determined by unpaired t test (a, g, k, l) or one-way ANOVA followed by Tukey’s multiple comparison test (b–f). h, i Representative images of three independent experiments are shown.
PLCδ1/δ3 nuclear translocation by glucagon mediates nuclear Ca2+ increase and gluconeogenesis
Nuclei possess phosphoinositide signaling mechanisms that are similar to those that occur at the plasma membrane, which regulates the amounts of the membrane phospholipid phosphatidylinositol 4,5-bisphosphate [PtdIns(4,5)P2] and phosphoinositide-specific phospholipase C (PtdIns-PLC)37,38,39. Nuclear PtdIns-PLC acts as positive feedback for nuclear Ca2+ signaling40. Thus, we investigated whether the increase in the nuclear Ca2+ signal with glucagon is affected by nuclear PLC activation. First, we identified the expression of PLCβ1, β3, β4, δ1, δ3, δ4, γ1, and γ2 isoforms in mouse primary hepatocytes (Supplementary Fig. 6a). Although most PLC isoforms display cytoplasmic localization in the unstimulated state, some PLC isoforms translocate to the nucleus after agonist treatment40. Therefore, we examined which PLC isoforms translocate to the nucleus upon stimulation with glucagon. Glucagon treatment resulted in the nuclear translocation of PLCδ1 and PLCδ3 but not the PLCβ1, β3, β4, δ4, γ1, or γ2 isoforms (Fig. 6a and Supplementary Fig. 6b). PLCδ1 and PLCδ3 were distributed outside of the nucleus during the nonstimulated state, whereas after stimulation with glucagon, the PLCs accumulated in clumps in the nuclear membrane. This translocation took place only in the presence of Ca2+ (Fig. 6b). These findings are consistent with our data showing that glucagon-induced nuclear Ca2+ signals were abolished in the Ca2+-free state (Supplementary Fig. 3b). PLCδ1 and PLCδ3 involvement in glucagon-induced nuclear Ca2+ signals and gluconeogenic gene expression was confirmed by PLCδ1 and PLCδ3 KD experiments (Fig. 6c, d). In contrast, PLCδ1 and PLCδ3 KD did not affect glucagon-induced cytosolic Ca2+ signals. These results indicate that nuclear translocation of PLCδ1 and PLCδ3 by glucagon is important for nuclear Ca2+ signals and hepatic gluconeogenesis.
a Mean densitometric values in Fig. S5B. n = 8 independent experiments. ***P < 0.001. Data are represented as mean ± SEM. Statistics were determined by unpaired t test. b Immunostaining of PLCδ1 and PLCδ3. Glucagon (100 nM) was administered to hepatocytes for 30 sec. Scale bar, 10 μm. Representative images of three independent experiments are shown. c Immunoblot of PLCδ1, PLCδ3, and GAPDH in hepatocytes treated with control scRNA or PLCδ1 or PLCδ3 siRNA. Representative images of three independent experiments are shown. Representative cytosolic and nuclear Ca2+ responses to glucagon in control scRNA-, PLCδ1 siRNA-, or PLCδ3-treated hepatocytes. The time point where 100 nM glucagon (Gcg) is added is indicated by the arrow. n = 20 cells for each condition. d Glucagon-induced mRNA levels of G6pc and Pck1 in control scRNA-, PLCδ1 siRNA-, or PLCδ3-siRNA treated hepatocytes. n = 8 independent experiments. ***P < 0.001. Data are represented as the mean ± SEM. Statistics were determined by one-way ANOVA followed by Tukey’s multiple comparison test.
ADPR-mediated nuclear Ca2+ signaling is required for gluconeogenesis in fasting mice and in a diabetic mouse model
The increase in circulating glucagon promotes hepatic gluconeogenesis in the fasting state and in diabetes5. We also confirmed an increase in plasma glucagon levels in the fasting condition when compared to fed WT mice (Supplementary Fig. 7a). Therefore, we examined the effects of hepatic ADPR-mediated Ca2+ signaling on gluconeogenic activity during fasting and in diabetes. We measured ADPR levels in hepatocytes from m + /db and diabetic animals during fasting as well as in the refed state. Hepatic ADPR levels were significantly higher in db/db mice than in m + /db mice, and ADPR levels were higher during fasting than after refeeding in both m + /db and db/db mice in correlation with plasma glucagon levels (Fig. 7a and Supplementary Fig. 7b, c, e). Hepatocytes prepared from db/db mice showed higher levels of glucagon-induced nuclear Ca2+ signals and gluconeogenic gene expression when compared to hepatocytes from m + /db mice, which was reduced by 8-Br-ADPR (Fig. 7b, c), implicating the potential role of ADPR-mediated nuclear Ca2+ signals in the pathogenesis of diabetes in association with gluconeogenesis. Correspondingly, administering 8-Br-ADPR to db/db mice reduced blood glucose levels in response to pyruvate challenge, CRE luciferase activity, and gluconeogenic gene expression in response to fasting (Fig. 7d–f). However, the administration of 8-Br-ADPR to db/db mice decreased plasma insulin levels and hepatic glycogen content, with no change in plasma glucagon levels (Supplementary Fig. 7d–f). Intriguingly, db/db mice showed upregulated Cx43 phosphorylation compared to normal mice, reinforcing the idea that ADPR production is regulated through Cx43 phosphorylation (Fig. 7g). Furthermore, fasting stimulated Cx43 phosphorylation at Ser368 and gluconeogenic gene expression, whereas feeding reduced Cx43 phosphorylation and gluconeogenic gene expression (Supplementary Fig. 8). These results suggest that ADPR-mediated nuclear Ca2+ signaling is important for glucose production in the fasting state and that the downregulation of ADPR signaling reduces circulating glucose levels in diabetes.
a Hepatic ADPR level of m + /db and db/db mice fed and fasted for 19 h. n = 6 independent experiments. ***P < 0.001. b Effect of 8-Br-ADPR on glucagon-induced nuclear Ca2+ signaling in hepatocytes prepared from m + /db and db/db mice. The time point where 100 nM glucagon (Gcg) is added is indicated by the arrow. n = 20 cells for each condition. c Glucagon-induced mRNA levels of G6pc in hepatocytes prepared from m + /db and db/db mice. Cells were preincubated with 8-Br-ADPR (100 μM) for 30 min before treatment with glucagon (100 nM). n = 6 independent experiments. ***P < 0.001, #P < 0.05; vehicle vs glucagon, ##P < 0.001; glucagon vs 8-Br-ADPR plus glucagon. d–f Effects of 8-Br-ADPR on the pyruvate tolerance test (d, n = 5 mice per group), CRE luciferase activity (e, n = 3 mice per group), and gluconeogenic gene expression (f, n = 7 mice per group) in db/db mice. 8-Br-ADPR (32 mg/kg) was intravenously administered to mice. **P < 0.01, ***P < 0.001. g Immunoblotting for pCx43 (S368), Cx43, and β-actin in the livers of m + /db and db/db mice after fasting for 19 h. n = 4 mice per group. Data are represented as the mean ± SEM. Statistics were determined by unpaired t test (d, f) or one-way ANOVA followed by Tukey’s multiple comparison test (a, c).
Discussion
In the current study, we examined the role of nCD38 in glucagon-induced gluconeogenesis in hepatocytes. Importantly, no previous study has investigated the dependence of gluconeogenic gene transcription on nCD38 in response to glucagon. Nuclear CD38 produces two Ca2+-mobilizing messengers in the nucleus: cADPR and ADPR, each with distinct roles in nuclear Ca2+ signaling. Adebanjo et al.8 previously proposed that CD38, with its catalytic site facing the nucleoplasm, may produce cADPR to activate RyR on the inner nuclear membrane to induce the initial Ca2+ increase. In contrast, we found that CD38 with its catalytic site facing the perinuclear space, forms ADPR to activate TRPM2 for the later phases of nuclear Ca2+ signaling. NAD+ is supplied via Cx43 from the nucleoplasm, and this mechanism is regulated by PKCδ-dependent phosphorylation in response to glucagon. ADPR then activates the TRPM2 channel, after which Ca2+ flows from perinuclear Ca2+ stores into the nucleoplasm, an essential process in regulating gluconeogenic gene expression. Consistently, all molecules involved in this signaling process, CD38, Cx43, TRPM2, and PKCδ, are localized to the inner membrane of the nucleus. Our results suggest that glucagon-induced sustained Ca2+ signals in both the nucleus and the cytosol are dependent on IP3 and cADPR (Supplementary Fig. 3a, d). Dantrolene, an inhibitor of the ryanodine receptor, inhibited glucagon-induced gene expression of G6pc and Pck1 (Supplementary Fig. 9). This is consistent with previous findings that IP3- and cADPR-mediated cytosolic Ca2+ signals are involved in glucagon-induced hepatic gluconeogenesis5,6. In addition, ER Ca2+ depletion results in SOCE41,42. Thus, Ca2+ mobilization from ER Ca2+ stores, triggered by IP3- and cADPR, is critical for the SOCE mechanism, emphasizing the importance of SOCE not only in replenishing perinuclear/ER Ca2+ stores but also for sustaining the Ca2+ signals in the cytosol and the nucleus (Supplementary Fig. 3b, c). Importantly, our results suggest that sufficient Ca2+ repletion of perinuclear Ca2+ stores through SOCE is a prerequisite for ADPR production by nCD38.
Nuclear Ca2+ signaling is regulated by nuclear PtdIns-PLC40. We also found that PLCδ1/δ3 are specifically involved in nuclear, but not cytosolic, Ca2+ signaling in response to glucagon stimulation. Previous findings have demonstrated that PLCδ1 translocates into the nucleus in a Ca2+-dependent manner43. Subsequently, this study demonstrates that IP3- and cADPR-mediated Ca2+ signals in the cytosol, which appear to be essential for the later cytosol and nuclear Ca2+ signals, trigger the nuclear translocation of PLCδ1/δ3. Inhibition of these cytosolic and nuclear Ca2+ signals with 8-Br-cADPR blocked ADPR production by inhibiting Cx43 phosphorylation, affirming that ADPR production is tightly regulated by upstream Ca2+ signaling.
It has been reported that PKCδ KO mice (either global or liver-specific) displayed increased hepatic insulin signaling and reduced expression of gluconeogenic enzymes44. Consistent with this report, we showed that the PKCδ inhibitor or PKCδ knockdown reduced glucagon-induced expression of G6pc and Pck1 (Fig. 2i, j). Our data suggest that PKCδ may be a key regulator of gluconeogenesis. As mentioned earlier, we found that upstream signaling of the nuclear translocation of PLCδ1/δ3 activates nuclear PKCδ, which phosphorylates Cx43 to gate NAD from the nucleoplasm into the perinuclear space (Fig. 2k–m). Given that constitutively active catalytic sites of type II CD38 are compartmentalized with limited substrate availability45, nCD38 can produce ADPR by using the supplied NAD in the perinuclear space. ADPR activates the TRPM2 Ca2+-release channel to increase nuclear Ca2+, ultimately resulting in the expression of G6pc and Pck1 (Fig. 2).
Glucagon regulates glucose homeostasis via a cAMP/PKA-dependent signaling pathway that results in the phosphorylation of CREB, which in turn activates the transcription of gluconeogenic enzymes46. Moreover, PKA mediates the phosphorylation of IP3R to increase cytosolic Ca2+, leading to an increase in calcineurin activity and the subsequent dephosphorylation of the CREB coactivator CRTC25. Based on our results, ADPR-mediated nuclear Ca2+ signaling is essential for CaMKII/CaMKIV/CREB phosphorylation, and nuclear Ca2+ signals are a determinant factor, converging multiple upstream signals, including cytosolic Ca2+ signaling, in gluconeogenic gene transcription in response to glucagon. This conclusion is supported by findings that the upregulation of ADPR-mediated Ca2+ signaling results in increased gluconeogenesis in diabetes.
In summary, this study has identified the essential role of nCD38 in glucagon-treated primary hepatocytes (Fig. 8). The compartmentalization of CD38 in the nucleus increases nuclear Ca2+ influx through TRPM2 activated by ADPR, leading to activation of the CaMKII/CaMKIV/CREB pathway for gluconeogenesis. We suggest that nCD38 plays a critical role in glucose homeostasis under physiological conditions such as fasting.
Glucagon regulates glucose homeostasis by binding to two G protein-coupled receptors (Gq and Gs) to produce IP3 and cADPR, respectively. IP3− and cADPR-mediated Ca2+ mobilization from ER Ca2+ stores induces SOCE, resulting in Ca2+ influx. This activates the nuclear translocation of PLCδ1/3. Nuclear PLCδ1/3 produces IP3 and DAG, activating PKCδ, which phosphorylates Cx43. Phospho-Cx43 gates NAD from the nucleoplasm into the perinuclear space. Constitutively active CD38, with its catalytic site facing the perinuclear space, produces ADPR, which then activates TRPM2 to trigger nucleoplasmic Ca2+ release. Nuclear Ca2+ increases the expression of G6pc and Pck1, which promote gluconeogenesis through the CaMKII/CaMKIV/CREB pathway. Note that CD38, Cx43, TRPM2, and PKCδ are all located on the inner nuclear membrane.
Data and materials availability
All data needed to evaluate the conclusions in the paper are present in the main text or the supplementary materials.
References
Nordlie, R. C., Foster, J. D. & Lange, A. J. Regulation of glucose production by the liver. Annu. Rev. Nutr. 19, 379–406 (1999).
Radziuk, J. & Pye, S. Hepatic glucose uptake, gluconeogenesis and the regulation of glycogen synthesis. Diabetes Metab. Res. Rev. 17, 250–272 (2001).
Lin, H. V. & Accili, D. L. Hormonal regulation of hepatic glucose production in health and disease. Cell Metab. 6, 9–19 (2011).
Wakelam, M. J., Murphy, G. J., Hruby, V. J. & Houslay, M. D. Activation of two signal-transduction systems in hepatocytes by glucagon. Nature 323, 68–71 (1986).
Wang, Y. et al. Inositol-1,4,5-trisphosphate receptor regulates hepatic gluconeogenesis in fasting and diabetes. Nature 485, 128–132 (2012).
Rah, S. Y. & Kim, U. H. CD38-mediated Ca2+ signaling contributes to glucagon-induced hepatic gluconeogenesis. Sci. Rep. 5, 10741 (2015).
Lee, H. C. Cyclic ADP-ribose and nicotinic acid adenine dinucleotide phosphate (NAADP) as messengers for calcium mobilization. J. Biol. Chem. 287, 31633–31640 (2012).
Adebanjo, O. A. et al. A new function for CD38/ADP-ribosyl cyclase in nuclear Ca2+ homeostasis. Nat. Cell Biol. 1, 409–414 (1999).
Park, D. R., Nam, T. S., Kim, Y. W., Bae, Y. S. & Kim, U. H. Oxidative activation of type III CD38 by NADPH oxidase–derived hydrogen peroxide in Ca2+ signaling. FASEB J. 33, 3404–3419 (2019).
Nam, T. S. et al. Interleukin-8 drives CD38 to form NAADP from NADP+ and NAAD in the endolysosomes to mobilize Ca2+ and effect cell migration. FASEB J. 34, 12565–12576 (2020).
Bezin, S. et al. Regulation of nuclear Ca2+ signaling by translocation of the Ca2+ messenger synthesizing enzyme ADP-ribosyl cyclase during neuronal depolarization. J. Biol. Chem. 283, 27859–27870 (2008).
Trubiani, O. et al. Insights into nuclear localization and dynamic association of CD38 in Raji and K562 cells. J. Cell Biochem. 103, 1294–1308 (2008).
Bootman, M. D., Fearnley, C., Smyrnias, I., MacDonald, F. & Roderick, H. L. An update on nuclear calcium signalling. J. Cell Sci. 122, 2337–2350 (2009).
Rodrigues, M. A., Gomes, D. A., Andrade, V. A., Leite, M. F. & Nathabson, M. H. Insulin induces calcium signals in the nucleus of rat hepatocytes. Hepatology 48, 1621–1631 (2008).
Zhang, L. et al. Phospholipase Cε hydrolyzes perinuclear phosphatidylinositol 4-phosphate to regulate cardiac hypertrophy. Cell 153, 216–227 (2013).
Gerasimenko, O. V., Gerasimenko, J. V., Tepikin, A. V. & Petersen, O. H. ATP-dependent accumulation and inositol trisphosphate- or cyclic ADP-ribose-mediated release of Ca2+ from the nuclear envelope. Cell 80, 439–444 (1995).
Humbert, J. P., Matter, N., Artault, J. C. & Malviya, A. N. Inositol 1,4,5-trisphosphate receptor is located to the inner nuclear membrane vindicating regulation of nuclear calcium signaling by inositol 1,4,5-trisphosphate. Discrete distribution of inositol phosphate receptors to inner and outer nuclear membranes. J. Biol. Chem 271, 478–485 (1996).
Echevarria, W., Leite, M. F., Guerra, M. T., Zipfel, W. R. & Nathabson, M. H. Regulation of calcium signals in the nucleus by a nucleoplasmic reticulum. Nat. Cell Biol. 5, 440–446 (2003).
Zhao, Y. et al. An expanded palette of genetically encoded Ca2+ indicators. Science 333, 1888–1891 (2011).
Galijatovic, A. et al. The human CYP1A1 gene is regulated in a developmental and tissue-specific fashion in transgenic mice. J. Biol. Chem. 279, 23969–23976 (2004).
Yoon, J. C. et al. Control of hepatic gluconeogenesis through the transcriptional coactivator PGC-1. Nature 413, 131–138 (2001).
Graeff, R. M., Walseth, T. F., Fryxell, K., Branton, W. D. & Lee, H. C. Enzymatic synthesis and characterizations of cyclic GDP-ribose. A procedure for distinguishing enzymes with ADP-ribosyl cyclase activity. J. Biol. Chem. 269, 30260–30267 (1994).
Quesada, I. et al. Nuclear KATP channels trigger nuclear Ca(2+) transients that modulate nuclear function. Proc. Natl. Acad. Sci. USA 99, 9544–9549 (2002).
Li, B. et al. Nuclear BK channels regulate gene expression via the control of nuclear calcium signaling. Nat. Neurosci. 17, 1055–1063 (2014).
Tong, L., Lee, S. & Denu, J. M. Hydrolase regulates NAD+ metabolites and modulates cellular redox. J. Biol. Chem. 284, 11256–11266 (2009).
Graeff, R. M. & Lee, H. C. A novel cycling assay for cellular cADP-ribose with nanomolar sensitivity. Biochem. J 361, 379–384 (2002).
Khoo, K. M. et al. Localization of the cyclic ADP-ribose-dependent calcium signaling pathway in hepatocyte nucleus. J. Biol. Chem. 275, 24807–24817 (2000).
Hatting, M., Tavares, C. D. J., Sharabi, K., Rines, A. K. & Puigserver, P. Insulin regulation of gluconeogenesis. Ann. N. Y. Acad. Sci. 1411, 21–35 (2018).
Bruzzone, S. et al. A self-restricted CD38-connexin 43 cross-talk affects NAD+ and cyclic ADP-ribose metabolism and regulates intracellular calcium in 3T3 fibroblasts. J. Biol. Chem. 276, 48300–48308 (2001).
Richards, T. S. et al. Protein kinase C spatially and temporally regulates gap junctional communication during human wound repair via phosphorylation of connexin43 on serine368. J. Cell Biol. 167, 555–562 (2004).
Cone, A. C. et al. Protein Kinase Cδ-mediated phosphorylation of connexin43 gap junction channels causes movement within gap junctions followed by vesicle internalization and protein degradation. J. Biol. Chem. 289, 8781–8798 (2014).
Perraud, A. L. et al. ADP-ribose gating of the calcium-permeable LTRPC2 channel revealed by Nudix motif homology. Nature 411, 595–599 (2001).
Lange, I. et al. TRPM2 functions as a lysosomal Ca2+-release channel in beta cells. Sci. Signal. 2, ra23 (2009).
Rah, S. Y., Kwak, J. Y., Chung, Y. J. & Kim, U. H. ADP-ribose/TRPM2-mediated Ca2+ signaling is essential for cytolytic degranulation and antitumor activity of natural killer cells. Sci. Rep. 25, 9482 (2015).
Koo, S. H. et al. The CREB coactivator TORC2 is a key regulator of fasting glucose metabolism. Nature 437, 1109–1111 (2005).
Ozcan, L. et al. Calcium signaling through CaMKII regulates hepatic glucose production in fasting and obesity. Cell Metab. 15, 739–751 (2012).
Visnjic, D. & Banfic, H. Nuclear phospholipid signaling: phosphatidylinositol-specific phospholipase C and phosphoinositide 3-kinase. Pflugers Arch. 455, 19–30 (2007).
Ye, K. & Ahn, J. Y. Nuclear phosphoinositide signaling. Front. Biosci. 13, 540–548 (2008).
Downes, C. P., Gray, A. & Lucocq, J. M. Probing phosphoinositide functions in signaling and membrane trafficking. Trends Cell Biol. 15, 259–268 (2005).
Yagisawa, H. Nucleocytoplasmic shuttling of phospholipase C-delta1: a link to Ca2+. J. Cell Biochem. 97, 233–243 (2006).
Hogan, P. G. & Rao, A. Store-operated calcium entry: Mechanisms and modulation. Biochem. Biophys. Res. Commun. 460, 40–49 (2015).
Lacruz, R. S. & Feske, S. Diseases caused by mutations in ORAI1 and STIM1. Ann. N. Y. Acad. Sci. 1356, 45–79 (2015).
Okada, M., Ishimoto, T., Naito, Y., Hirata, H. & Yagisawa, H. Phospholipase Cdelta1 associates with importin beta1 and translocates into the nucleus in a Ca2+-dependent manner. FEBS Lett. 579, 4949–4954 (2005).
Bezy, O. et al. PKCδ regulates hepatic insulin sensitivity and hepatosteatosis in mice and humans. J. Clin. Invest. 121, 2504–2517 (2011).
Kim, U. H. Multiple enzymatic activities of CD38 for Ca2+ signaling messengers. Messenger 3, 6–14 (2014).
Habegger, K. M. et al. The metabolic actions of glucagon revisited. Nat. Rev. Endocrinol. 6, 689–697 (2010).
Acknowledgements
The authors thank Yasuo Mori (Kyoto University, Japan) for kindly providing TRPM2 KO mice. This work was supported by Korean National Research Foundation Grant 2012R1A3A2026453 to U.-H.K, 2014R1A6A1030318 to H.T.C, and 2021R1I1A1A01047480 to S.-Y.R.
Author information
Authors and Affiliations
Contributions
S.-Y.R. designed the study, performed most experiments, and wrote the manuscript. C.P., Y.J., J.P., and S.W.R. assisted with data analysis and wrote the paper. H.T.C. provided intellectual feedback and support. U.-H.K. conceived the project, supervised the research, and wrote and edited the paper. U.-H.K. is the guarantor of this work.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rah, SY., Joe, Y., Park, J. et al. CD38/ADP-ribose/TRPM2-mediated nuclear Ca2+ signaling is essential for hepatic gluconeogenesis in fasting and diabetes. Exp Mol Med 55, 1492–1505 (2023). https://doi.org/10.1038/s12276-023-01034-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s12276-023-01034-9
- Springer Nature Limited