Abstract
Dicer is a central enzymatic player in RNA-interference pathways that acts to regulate gene expression in nearly all eukaryotes. Although the cytoplasmic function of Dicer is well documented in mammals, its nuclear function remains obscure. Here we show that Dicer is present in both the nucleus and cytoplasm, and its nuclear levels are tightly regulated. Dicer interacts with RNA polymerase II (Pol II) at actively transcribed gene loci. Loss of Dicer causes the appearance of endogenous double-stranded RNA (dsRNA), which in turn leads to induction of the interferon-response pathway and consequent cell death. Our results suggest that Pol II–associated Dicer restricts endogenous dsRNA formation from overlapping noncoding-RNA transcription units. Failure to do so has catastrophic effects on cell function.








Similar content being viewed by others
References
Grewal, S.I. RNAi-dependent formation of heterochromatin and its diverse functions. Curr. Opin. Genet. Dev. 20, 134–141 (2010).
Bartel, D.P. MicroRNAs: target recognition and regulatory functions. Cell 136, 215–233 (2009).
Baulcombe, D. RNA silencing in plants. Nature 431, 356–363 (2004).
Gullerova, M. & Proudfoot, N.J. Cohesin complex promotes transcriptional termination between convergent genes in S. pombe. Cell 132, 983–995 (2008).
Gullerova, M. & Proudfoot, N.J. Convergent transcription induces transcriptional gene silencing in fission yeast and mammalian cells. Nat. Struct. Mol. Biol. 19, 1193–1201 (2012).
Minchiotti, G. & Di Nocera, P.P. Convergent transcription initiates from oppositely oriented promoters within the 5′ end regions of Drosophila melanogaster F elements. Mol. Cell. Biol. 11, 5171–5180 (1991).
Faghihi, M.A. & Wahlestedt, C. Regulatory roles of natural antisense transcripts. Nat. Rev. Mol. Cell Biol. 10, 637–643 (2009).
Samuel, C.E. Antiviral actions of interferons. Clin. Microbiol. Rev. 14, 778–809 (2001).
García, M.A., Meurs, E.F. & Esteban, M. The dsRNA protein kinase PKR: virus and cell control. Biochimie 89, 799–811 (2007).
Han, J. et al. Molecular basis for the recognition of primary microRNAs by the Drosha-DGCR8 complex. Cell 125, 887–901 (2006).
Yates, L.A., Norbury, C.J. & Gilbert, R.J. The long and short of microRNA. Cell 153, 516–519 (2013).
Jaskiewicz, L. & Filipowicz, W. Role of Dicer in posttranscriptional RNA silencing. Curr. Top. Microbiol. Immunol. 320, 77–97 (2008).
Bühler, M., Verdel, A. & Moazed, D. Tethering RITS to a nascent transcript initiates RNAi- and heterochromatin-dependent gene silencing. Cell 125, 873–886 (2006).
Bühler, M., Haas, W., Gygi, S.P. & Moazed, D. RNAi-dependent and -independent RNA turnover mechanisms contribute to heterochromatic gene silencing. Cell 129, 707–721 (2007).
Moazed, D. et al. Studies on the mechanism of RNAi-dependent heterochromatin assembly. Cold Spring Harb. Symp. Quant. Biol. 71, 461–471 (2006).
Billy, E., Brondani, V., Zhang, H., Muller, U. & Filipowicz, W. Specific interference with gene expression induced by long, double-stranded RNA in mouse embryonal teratocarcinoma cell lines. Proc. Natl. Acad. Sci. USA 98, 14428–14433 (2001).
Provost, P. et al. Ribonuclease activity and RNA binding of recombinant human Dicer. EMBO J. 21, 5864–5874 (2002).
Kotaja, N. et al. The chromatoid body of male germ cells: similarity with processing bodies and presence of Dicer and microRNA pathway components. Proc. Natl. Acad. Sci. USA 103, 2647–2652 (2006).
Jakymiw, A. et al. Overexpression of dicer as a result of reduced let-7 microRNA levels contributes to increased cell proliferation of oral cancer cells. Genes Chromosom. Cancer 49, 549–559 (2010).
Doyle, M. et al. The double-stranded RNA binding domain of human Dicer functions as a nuclear localization signal. RNA 19, 1238–1252 (2013).
Haussecker, D. & Proudfoot, N.J. Dicer-dependent turnover of intergenic transcripts from the human β-globin gene cluster. Mol. Cell. Biol. 25, 9724–9733 (2005).
Morris, K.V., Chan, S.W., Jacobsen, S.E. & Looney, D.J. Small interfering RNA-induced transcriptional gene silencing in human cells. Science 305, 1289–1292 (2004).
Janowski, B.A. et al. Inhibiting gene expression at transcription start sites in chromosomal DNA with antigene RNAs. Nat. Chem. Biol. 1, 216–222 (2005).
Saint-André, V., Batsche, E., Rachez, C. & Muchardt, C. Histone H3 lysine 9 trimethylation and HP1γ favor inclusion of alternative exons. Nat. Struct. Mol. Biol. 18, 337–344 (2011).
Alló, M. et al. Control of alternative splicing through siRNA-mediated transcriptional gene silencing. Nat. Struct. Mol. Biol. 16, 717–724 (2009).
Ameyar-Zazoua, M. et al. Argonaute proteins couple chromatin silencing to alternative splicing. Nat. Struct. Mol. Biol. 19, 998–1004 (2012).
Schmitter, D. et al. Effects of Dicer and Argonaute down-regulation on mRNA levels in human HEK293 cells. Nucleic Acids Res. 34, 4801–4815 (2006).
Nesterova, T.B. et al. Dicer regulates Xist promoter methylation in ES cells indirectly through transcriptional control of Dnmt3a. Epigenetics Chromatin 1, 2 (2008).
Morris, D.P., Michelotti, G.A. & Schwinn, D.A. Evidence that phosphorylation of the RNA polymerase II carboxyl-terminal repeats is similar in yeast and humans. J. Biol. Chem. 280, 31368–31377 (2005).
Zamore, P.D. & Haley, B. Ribo-gnome: the big world of small RNAs. Science 309, 1519–1524 (2005).
Kim, D.H., Villeneuve, L.M., Morris, K.V. & Rossi, J.J. Argonaute-1 directs siRNA-mediated transcriptional gene silencing in human cells. Nat. Struct. Mol. Biol. 13, 793–797 (2006).
Affymetrix ENCODE Transcriptome Project; Cold Spring Harbor Laboratory ENCODE Transcriptome Project. Post-transcriptional processing generates a diversity of 5′-modified long and short RNAs. Nature 457, 1028–1032 (2009).
Wang, R. et al. Mouse embryonic stem cells are deficient in type I interferon expression in response to viral infections and double-stranded RNA. J. Biol. Chem. 288, 15926–15936 (2013).
Stetson, D.B. & Medzhitov, R. Type I interferons in host defense. Immunity 25, 373–381 (2006).
Kawai, T. & Akira, S. Toll-like receptors and their crosstalk with other innate receptors in infection and immunity. Immunity 34, 637–650 (2011).
Wilson, R.C. & Doudna, J.A. Molecular mechanisms of RNA interference. Annu. Rev. Biophys. 42, 217–239 (2013).
Gagnon, K.T., Li, L., Chu, Y., Janowski, B.A. & Corey, D.R. RNAi factors are present and active in human cell nuclei. Cell Reports 6, 211–221 (2014).
Lee, J.T. Epigenetic regulation by long noncoding RNAs. Science 338, 1435–1439 (2012).
Kaneko, H. et al. DICER1 deficit induces Alu RNA toxicity in age-related macular degeneration. Nature 471, 325–330 (2011).
Passon, N. et al. Expression of Dicer and Drosha in triple-negative breast cancer. J. Clin. Pathol. 65, 320–326 (2012).
Castel, S.E. & Martienssen, R.A. RNA interference in the nucleus: roles for small RNAs in transcription, epigenetics and beyond. Nat. Rev. Genet. 14, 100–112 (2013).
Peters, A.H. et al. Loss of the Suv39h histone methyltransferases impairs mammalian heterochromatin and genome stability. Cell 107, 323–337 (2001).
Fodor, B.D. et al. Jmjd2b antagonizes H3K9 trimethylation at pericentric heterochromatin in mammalian cells. Genes Dev. 20, 1557–1562 (2006).
Bulut-Karslioglu, A. et al. A transcription factor–based mechanism for mouse heterochromatin formation. Nat. Struct. Mol. Biol. 19, 1023–1030 (2012).
Martens, J.H. et al. The profile of repeat-associated histone lysine methylation states in the mouse epigenome. EMBO J. 24, 800–812 (2005).
Shinkai, Y. & Tachibana, M. H3K9 methyltransferase G9a and the related molecule GLP. Genes Dev. 25, 781–788 (2011).
Fagegaltier, D. et al. The endogenous siRNA pathway is involved in heterochromatin formation in Drosophila. Proc. Natl. Acad. Sci. USA 106, 21258–21263 (2009).
Aagaard, L. et al. A facile lentiviral vector system for expression of doxycycline-inducible shRNAs: knockdown of the pre-miRNA processing enzyme Drosha. Mol. Ther. 15, 938–945 (2007).
Landt, S.G. et al. ChIP-seq guidelines and practices of the ENCODE and modENCODE consortia. Genome Res. 22, 1813–1831 (2012).
Pall, G.S., Codony-Servat, C., Byrne, J., Ritchie, L. & Hamilton, A. Carbodiimide-mediated cross-linking of RNA to nylon membranes improves the detection of siRNA, miRNA and piRNA by northern blot. Nucleic Acids Res. 35, e60 (2007).
Acknowledgements
We are grateful to E. Johnson for help with transmission electron microscopy and scanning electron microscopy experiments. This work was supported by grants from the Wellcome Trust (091805/Z/10/Z to N.J.P.) and E.P. Abraham Trust (to N.J.P.) and by a Medical Research Council Career Development Award (MR/K006606/1 to M.G.) and a L'Oreal-UNESCO Woman in Science UK and Ireland award (to M.G.).
Author information
Authors and Affiliations
Contributions
E.W. and M.G. performed all the experimental analyses. M.S. and K.K.-G. performed bioinformatics analyses of the ChIP-seq and RNA-seq data. E.W., M.G. and N.J.P. designed the experiments and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Dicer localizes in both nucleus and cytoplasm of HEK293 cells.
a) Western blot analysis of protein extracts from HEK293 cells with inducible integrated shRNA transgene. Induction by doxycycline leads to Dicer-specific shRNA production and consequent Dicer knockdown. Antibodies specific to Dicer and γ-actin were employed. b) Western blot analysis of Dicer in nuclear and cytoplasmic extracts from HEK293 cells transfected with episomal GFP-Dicer or RFP-Dicer expressing plasmids. Grp75 and γ-actin were used as controls. c) Expression of GFP-Dicer transgene (green) in normal and Dicer knockout murine embryonic stem cells as in Figure 1b. d) Control FRAP experiment shows Dicer-GFP (green) recovery in nucleus of HEK293 cells (lacking GFP signal) after bleaching. This provides a background control to FRAP analysis of GFP-Dicer expression in the nucleus of Dicer knockdown cells. Here, only background signals can be detected. The cell is shown in the left hand panel before bleaching. In the middle panel, the red circle corresponds to bleached area in nucleus. Fluorescent recovery was measured in the green circle. Fluorescent background values were measured in the blue circle. Fluorescent recovery was measured in 20 frames, bleaching was initiated at frame 5. Right-hand panel shows the cell after recovery. Absolute levels of fluorescence and relative levels of recovery (background fluorescence subtracted from fluorescence levels in measured region) are shown in the left and right panels, respectively. All experiments described in Supplementary Fig. 1a-c, were replicated three times.
Supplementary Figure 2 Genomic annotation of Dicer ChIP-seq peaks.
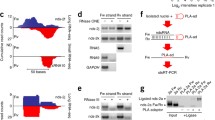
a) Table showing the top 12 peaks of Dicer binding loci according to the ChIP sequencing data. Nearest gene identity is shown. b) The 4 additional loci chosen for further analysis, showing a range of Dicer binding c) Metagene analysis of top 118 Dicer binding sites (ChIP-seq peaks) showing their location distribution around TSS and PAS of coding genes in the human genome. Data were grouped into 250 nt bins. Each bin-midpoint was plotted on the graph. d) Table summarising the enrichment of repetitive DNA sequence elements in the Dicer ChIP-seq data. The RepeatMasker hg19 table was used to extract genomic repetitive elements. Repetitive element overlapping a Dicer peak with 5 nt or more was included in the analysis. Each category of repetitive elements as presented, was analysed separately. p-values are based on a background distribution of sampling 118 random genomic sequences of the same length as Dicer peaks. Sampling was performed more than 1000 times. e) H3 control ChIP experiment as in Figure 2e. f) Top panel: Diagram of the MTRNR2L6. The start site of transcription (arrow) and the position of the PCR amplicons (bold) are indicated. Bottom panels: Pol II, Dicer and H3 ChIP experiments as in Figure 2e.
Supplementary Figure 4 Loss of Drosha has no effect on the accumulation of long dsRNA.
a) IF as in Fig. 1a using anti-Drosha and J2 antibodies on normal and Drosha knockdown HEK293 cells. b) Levels of pri-mRNA15a transcripts as determined by qRT-PCR analysis using a specific primer for reverse transcription. RNA was isolated from normal HEK293 cells and cells treated with an shRNA against Drosha for 10 days. Signals are based on average values ± s.d. from three independent biological experiments and are normalised to the levels of transcripts in normal cells, set as 1.
Supplementary Figure 5 Loss of Dicer leads to accumulation of long dsRNA.
IF as in Fig. 1a using anti-Dicer and J2 antibodies on Dicer knockout murine embryonic stem cells.
Supplementary Figure 6 Loss of Dicer leads to accumulation of dsRNA.
a) Histogram of peak summit ratio of dsRNA in Dicer knockdown cells to dsRNA in normal cells. Any dsRNA signal >10 in both uninduced and induced cells was considered. Value higher than 1 indicates accumulation of dsRNA and value lower than 1 indicates a decrease in dsRNA levels in Dicer knockdown cells. The black line marks the value of 1. Dark blue bins indicate the number of times a ratio falls in value span marked on the horizontal axis. Light blue bins represent a cumulative number for the values under 1 or over 1 separately. Centromeric repeat regions were excluded from analysis. b) Diagram of the change level in dsRNA (according to peak summit value) accumulation in normal and Dicer knockdown cells. Any dsRNA signal >10 in both uninduced and induced cells was considered. Plotted levels from normal cells are subtracted Dicer knockdown cell levels. Value less than 0 (marked by a black vertical line) indicates the absolute value of the decrease in dsRNA, while a value greater than 0 indicates an increase. Each blue point depicts one locus of Dicer-dsRNA co-localization. The loci were ordered according to the difference value in ascending order. One point is too large to be depicted in the plot, it has value 2432530. Note the scale of the graph is (x104). Centromeric repeat regions were excluded from analysis. c) Box Plot of change in dsRNA levels (according to peak summit value) between Dicer knockdown and normal cells. Any dsRNA signal >10 in both uninduced and induced cells was considered. The red mark indicated the median, at a value of 855. The lower quartile q1 (25th percentile) is at -19 and the upper quartile q3 (75th percentile) at 3992 (blue box). The shift in the positive direction of the box shows a general increase in dsRNA at Dicer loci. 20 outlier values were excluded from the plot. Centromeric repeat regions were excluded from analysis. d) Tabular summary of enrichment of repetitive elements in dsRNA-seq data. The RepeatMasker hg19 table was used to extract genomic repetitive elements. Repetitive element overlapping a dsRNA peak with 5 nt or more was included in the analysis. Each category of repetitive elements as presented, was analysed separately. p-values are based on a background distribution of sampling 118 random genomic sequences of the same length as Dicer peaks. Sampling was performed more than 1000 times. e) Metagene analysis of top dsRNA peaks (see Online Methods) showing location distribution around TSS and PAS of coding genes in the human genome. Data were grouped into 250 nt bins. Each bin-midpoint was plotted on graph.
Supplementary Figure 7 Loss of Dicer triggers the interferon response.
a) Quantitation of Western blot analysis of cell extracts isolated from normal and Dicer knockdown HEK293 cells, growing in tissue culture for 1 or 2 weeks using antibodies specific to Dicer, TLR3, PKR1 and γ-actin (see Figure 7a). Signals were measured by image-Quant software, are expressed as a % of input and are expressed as an average of three independent experiments, normalised to normal HEK293 cell levels, set as 1. * indicates statistical significance (p < 0.05), based on unpaired, two-tailed distribution Student's t-test. b) IF as in Figure 4c using anti-PKR antibody (green).
Supplementary Figure 8 Loss of Dicer triggers the interferon response.
a) Morphological changes of normal and Dicer knockdown HEK293 cells using Electron Microscopy (SEM) at 3000-fold magnification. b) TEM analysis of nuclear structure in normal and Dicer knockdown HEK293. c) TEM as in middle panel focusing on mitochondria. Representative images from 20 cells are presented.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–9 and Supplementary Table 1 (PDF 6583 kb)
Rights and permissions
About this article
Cite this article
White, E., Schlackow, M., Kamieniarz-Gdula, K. et al. Human nuclear Dicer restricts the deleterious accumulation of endogenous double-stranded RNA. Nat Struct Mol Biol 21, 552–559 (2014). https://doi.org/10.1038/nsmb.2827
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.2827
- Springer Nature America, Inc.
This article is cited by
-
Dicer promotes genome stability via the bromodomain transcriptional co-activator BRD4
Nature Communications (2022)
-
Conserved chromosomal functions of RNA interference
Nature Reviews Genetics (2020)
-
Genetic variants in humanin nuclear isoform gene regions show no association with coronary artery disease
BMC Research Notes (2019)
-
Dgcr8 knockout approaches to understand microRNA functions in vitro and in vivo
Cellular and Molecular Life Sciences (2019)
-
Protective paraspeckle hyper-assembly downstream of TDP-43 loss of function in amyotrophic lateral sclerosis
Molecular Neurodegeneration (2018)