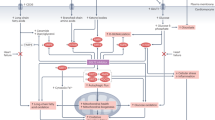
Key Points
-
Nicotinamide adenine dinucleotide (NAD+) is a coenzyme with roles in several cardiac and renal metabolic processes
-
NAD+ depletion is emerging as a major contributor to the pathogenesis of cardiac and renal disease
-
Preclinical data suggest that NAD+ repletion strategies have the potential to restore healthy renal and cardiac metabolism and physiology
-
The mitochondrial sirtuins mediate some of the beneficial effects of NAD+ supplementation
-
NAD+ supplementation can directly enhance metabolism and improve cellular redox reactions in the setting of cardiac and renal disease
-
NAD+ is also a substrate for enzymes involved in DNA damage repair and calcium signalling pathways; NAD+ supplementation could alter these pathways to influence cell viability, organ function and disease outcomes
Abstract
The coenzyme nicotinamide adenine dinucleotide (NAD+) has key roles in the regulation of redox status and energy metabolism. NAD+ depletion is emerging as a major contributor to the pathogenesis of cardiac and renal diseases and NAD+ repletion strategies have shown therapeutic potential as a means to restore healthy metabolism and physiological function. The pleotropic roles of NAD+ enable several possible avenues by which repletion of this coenzyme could have therapeutic efficacy. In particular, NAD+ functions as a co-substrate in deacylation reactions carried out by the sirtuin family of enzymes. These NAD+-dependent deacylases control several aspects of metabolism and a wealth of data suggests that boosting sirtuin activity via NAD+ supplementation might be a promising therapy for cardiac and renal pathologies. This Review summarizes the role of NAD+ metabolism in the heart and kidney, and highlights the mitochondrial sirtuins as mediators of some of the beneficial effects of NAD+-boosting therapies in preclinical animal models. We surmise that modulating the NAD+–sirtuin axis is a clinically relevant approach to develop new therapies for cardiac and renal diseases.


Similar content being viewed by others
References
Forbes, J. M. Mitochondria-power players in kidney function? Trends Endocrinol. Metab. 27, 441–442 (2016).
Tran, M. T. et al. PGC1α drives NAD biosynthesis linking oxidative metabolism to renal protection. Nature 531, 528–532 (2016). This study demonstrates that NAM treatment improves renal function in a mouse model of AKI.
Cantó, C., Menzies, K. J. & Auwerx, J. NAD+ metabolism and the control of energy homeostasis: a balancing act between mitochondria and the nucleus. Cell Metab. 22, 31–53 (2015).
He, W., Newman, J. C., Wang, M. Z., Ho, L. & Verdin, E. Mitochondrial sirtuins: regulators of protein acylation and metabolism. Trends Endocrinol. Metab. 23, 467–476 (2012).
Mericskay, M. Nicotinamide adenine dinucleotide homeostasis and signalling in heart disease: pathophysiological implications and therapeutic potential. Arch. Cardiovasc. Dis. 109, 207–215 (2015).
Ebrahimkhani, M. R. et al. Aag-initiated base excision repair promotes ischemia reperfusion injury in liver, brain, and kidney. Proc. Natl Acad. Sci. USA 111, E4878–E4886 (2014).
Braidy, N. et al. Age related changes in NAD+ metabolism oxidative stress and Sirt1 activity in wistar rats. PLoS ONE 6, e19194 (2011).
Mori, V. et al. Metabolic profiling of alternative NAD biosynthetic routes in mouse tissues. PLoS ONE 9, e113939 (2014).
Lin, L. F. & Henderson, L. M. Pyridinium precursors of pyridine nucleotides in perfused rat kidney and in the testis. J. Biol. Chem. 247, 8023–8030 (1972).
Ikeda, M. et al. Studies on the biosynthesis of nicotinamide adenine dinucleotide. II. A role of picolinic carboxylase in the biosynthesis of nicotinamide adenine dinucleotide from tryptophan in mammals. J. Biol. Chem. 240, 1395–1401 (1965).
Shibata, K., Morita, N., Shibata, Y. & Fukuwatari, T. Enzymes that control the conversion of L-tryptophan-nicotinamide and the urinary excretion ratio (N1-methyl-2-pyridone-5-carboxamide + N1-methyl-4-pyridone-3-carboxamide)/N1-methylnicotinamide in mice. Biosci. Biotechnol. Biochem. 77, 2105–2111 (2013).
Eto, N., Miyata, Y., Ohno, H. & Yamashita, T. Nicotinamide prevents the development of hyperphosphataemia by suppressing intestinal sodium-dependent phosphate transporter in rats with adenine-induced renal failure. Nephrol. Dial. Transplant. 20, 1378–1384 (2005).
Kempson, S. A., Colon-Otero, G., Ou, S. Y., Turner, S. T. & Dousa, T. P. Possible role of nicotinamide adenine dinucleotide as an intracellular regulator of renal transport of phosphate in the rat. J. Clin. Invest. 67, 1347–1360 (1981).
Gopal, E. et al. Sodium-coupled and electrogenic transport of B-complex vitamin nicotinic acid by slc5a8, a member of the Na/glucose co-transporter gene family. Biochem. J. 388, 309–316 (2005).
Trueblood, N. A., Ramasamy, R., Wang, L. F. & Schaefer, S. Niacin protects the isolated heart from ischemia-reperfusion injury. Am. J. Physiol. Heart Circ. Physiol. 279, H764–H771 (2000).
Collins, P. B. & Chaykin, S. The management of nicotinamide and nicotinic acid in the mouse. J. Biol. Chem. 247, 778–783 (1972).
Corr, P. B. & May, D. G. Renal mechanisms for the excretion of nicotinic acid. J. Pharmacol. Exp. Ther. 192, 195–200 (1975).
Nomura, K. et al. Hepatectomy-related hypophosphatemia: a novel phosphaturic factor in the liver–kidney axis. J. Am. Soc. Nephrol. 25, 761–772 (2014).
Grozio, A. et al. CD73 protein as a source of extracellular precursors for sustained NAD+ biosynthesis in FK866-treated tumor cells. J. Biol. Chem. 288, 25938–25949 (2013).
Ratajczak, J. et al. NRK1 controls nicotinamide mononucleotide and nicotinamide riboside metabolism in mammalian cells. Nat. Commun. 7, 13103 (2016).
Trammell, S. A. J. et al. Nicotinamide riboside is uniquely and orally bioavailable in mice and humans. Nat. Commun. 7, 12948 (2016). The first clinical study of nicotinamide riboside supplementation; shows a dose-dependent increase in NAD+ with nicotinamide riboside, demonstrating the potential of NAD+ boosting therapies in a clinical setting.
Karamanlidis, G. et al. Mitochondrial complex I deficiency increases protein acetylation and accelerates heart failure. Cell. Metab. 18, 239–250 (2013).
Lee, C. F. et al. Normalization of NAD+ redox balance as a therapy for heart failure. Circulation 134, 883–894 (2016). This study demonstrates that NMN and cardiac NAMPT increase NAD+, reduce protein hyperacetylation, and improve cardiac function in a mouse model of heart failure.
Yamamoto, T. et al. Nicotinamide mononucleotide, an intermediate of NAD+ synthesis, protects the heart from ischemia and reperfusion. PLoS ONE 9, e98972 (2014).
Berger, F., Lau, C., Dahlmann, M. & Ziegler, M. Subcellular compartmentation and differential catalytic properties of the three human nicotinamide mononucleotide adenylyltransferase isoforms. J. Biol. Chem. 280, 36334–36341 (2005).
Yamamoto, M. et al. Nmnat3 is dispensable in mitochondrial NAD level maintenance in vivo. PLoS ONE 11, e0147037 (2016).
Hsu, C.-P., Yamamoto, T., Oka, S. & Sadoshima, J. The function of nicotinamide phosphoribosyltransferase in the heart. DNA Repair (Amst.) 23, 64–68 (2014).
Wang, P. & Miao, C.-Y. NAMPT as a therapeutic target against stroke. Trends Pharmacol. Sci. 36, 891–905 (2015).
Aboud, O. A. et al. Dual and specific inhibition of NAMPT and PAK4 by KPT-9274 decreases kidney cancer growth. Mol. Cancer Ther. 15, 2119–2129 (2016).
Benito-Martin, A. et al. Endogenous NAMPT dampens chemokine expression and apoptotic responses in stressed tubular cells. Biochim. Biophys. Acta 1842, 293–303 (2014).
Pillai, V. B. et al. Exogenous NAD blocks cardiac hypertrophic response via activation of the SIRT3–LKB1–AMP-activated kinase pathway. J. Biol. Chem. 285, 3133–3144 (2010). Study showing that exogenous NAD+ supplementation blocked agonist-induced hypertrophic responses in cardiomyocytes and mouse models; implicates SIRT3 deacetylase activity in the mechanism of this cardioprotection.
Hsu, C.-P., Oka, S., Shao, D., Hariharan, N. & Sadoshima, J. Nicotinamide phosphoribosyltransferase regulates cell survival through NAD+ synthesis in cardiac myocytes. Circ. Res. 105, 481–491 (2009).
Zhuo, L. et al. NAD blocks high glucose induced mesangial hypertrophy via activation of the sirtuins–AMPK–mTOR pathway. Cell. Physiol. Biochem. 27, 681–690 (2011). This study shows that exogenous NAD+ maintains SIRT1 and SIRT3 activity in the setting of high-glucose-induced mesangial hypertrophy and provides a mechanism for how sirtuin activity protects against mesangial hypertrophy.
Morigi, M. et al. Sirtuin 3-dependent mitochondrial dynamic improvements protect against acute kidney injury. J. Clin. Invest. 125, 715–726 (2015).
Anderson, K. A., Green, M. F., Huynh, F. K., Wagner, G. R. & Hirschey, M. D. SnapShot: mammalian sirtuins. Cell 159, 956–956.e1 (2014).
Denu, J. M. The Sir2 family of protein deacetylases. Curr. Opin. Chem. Biol. 9, 431–440 (2005).
Peng, C. et al. The first identification of lysine malonylation substrates and its regulatory enzyme. Mol. Cell. Proteomics 10, M111.012658 (2011).
Du, J. et al. Sirt5 is a NAD-dependent protein lysine demalonylase and desuccinylase. Science 334, 806–809 (2011).
Tan, M. et al. Lysine glutarylation is a protein posttranslational modification regulated by SIRT5. Cell. Metab. 19, 605–617 (2014).
Feldman, J. L., Baeza, J. & Denu, J. M. Activation of the protein deacetylase SIRT6 by long-chain fatty acids and widespread deacylation by mammalian sirtuins. J. Biol. Chem. 288, 31350–31356 (2013).
Sundaresan, N. R. et al. The sirtuin SIRT6 blocks IGF–Akt signaling and development of cardiac hypertrophy by targeting c-Jun. Nat. Med. 18, 1643–1650 (2012).
Gupta, M., Samant, S., Bao, R. & Pillai, V. The sirtuin SIRT6 represses expression of cachexia-associated cytokine myostatin by blocking its NF-kB-dependent gene transcription. FASEB J. 30, 1009.11 (2016).
Hirschey, M. D. et al. SIRT3 regulates mitochondrial fatty-acid oxidation by reversible enzyme deacetylation. Nature 464, 121–125 (2010).
Haigis, M. C. et al. SIRT4 inhibits glutamate dehydrogenase and opposes the effects of calorie restriction in pancreatic beta cells. Cell 126, 941–954 (2006).
Michishita, E., Park, J. Y. & Burneskis, J. M. Evolutionarily conserved and nonconserved cellular localizations and functions of human SIRT proteins. Mol. Biol. Cell 16, 4623–4635 (2005).
Mathias, R. A. et al. Sirtuin 4 is a lipoamidase regulating pyruvate dehydrogenase complex activity. Cell 159, 1615–1625 (2014).
Jeong, S. M. et al. SIRT4 has tumor-suppressive activity and regulates the cellular metabolic response to DNA damage by inhibiting mitochondrial glutamine metabolism. Cancer Cell 23, 450–463 (2013).
Wagner, G. R. & Payne, R. M. Mitochondrial acetylation and diseases of aging. J. Aging Res. 2011, 1–13 (2011).
Wagner, G. R. & Hirschey, M. D. Nonenzymatic protein acylation as a carbon stress regulated by sirtuin deacylases. Mol. Cell 54, 5–16 (2014).
Ghanta, S., Grossmann, R. E. & Brenner, C. Mitochondrial protein acetylation as a cell-intrinsic, evolutionary driver of fat storage: biocehmical and metabolic logic of acetyl-lysine modifications. Crit. Rev. Biochem. Mol. Biol. 48, 1–42 (2013).
Chhoy, P. et al. in Sirtuins Vol. 10 (ed. Houtkooper, R.) 105–138 (Sirtuins, 2016).
Park, J. et al. SIRT5-mediated lysine desuccinylation impacts diverse metabolic pathways. Mol. Cell 50, 919–930 (2013).
Imai, S., Armstrong, C. M., Kaeberlein, M. & Guarente, L. Transcriptional silencing and longevity protein Sir2 is an NAD-dependent histone deacetylase. Nature 403, 795–800 (2000).
Anderson, R. M. et al. Manipulation of a nuclear NAD+ salvage pathway delays aging without altering steady-state NAD+ levels. J. Biol. Chem. 277, 18881–18890 (2002).
Belenky, P. et al. Nicotinamide riboside promotes Sir2 silencing and extends lifespan via Nrk and Urh1/Pnp1/Meu1 pathways to NAD+. Cell 129, 473–484 (2007).
Bitterman, K. J., Anderson, R. M., Cohen, H. Y., Latorre-Esteves, M. & Sinclair, D. A. Inhibition of silencing and accelerated aging by nicotinamide, a putative negative regulator of yeast sir2 and human SIRT1. J. Biol. Chem. 277, 45099–45107 (2002).
Mouchiroud, L. et al. The NAD+/sirtuin pathway modulates longevity through activation of mitochondrial UPR and FOXO signaling. Cell 154, 430–441 (2013).
Yoshino, J., Mills, K. F., Yoon, M. J. & Imai, S.-I. Nicotinamide mononucleotide, a key NAD+ intermediate, treats the pathophysiology of diet- and age-induced diabetes in mice. Cell 14, 528–536 (2011).
Imai, S. & Guarente, L. NAD+ and sirtuins in aging and disease. Trends Cell Biol. 24, 464–471 (2014).
Giblin, W., Skinner, M. E. & Lombard, D. B. Sirtuins: guardians of mammalian healthspan. Trends Genet. 30, 271–286 (2014).
Ma, L. & Li, Y. SIRT1: role in cardiovascular biology. Clin. Chim. Acta 440, 8–15 (2015).
Winnik, S., Auwerx, J., Sinclair, D. A. & Matter, C. M. Protective effects of sirtuins in cardiovascular diseases: from bench to bedside. Eur. Heart J. 36, 3404–3412 (2015).
Kong, L. et al. Sirtuin 1: a target for kidney diseases. Mol. Med. 21, 87–97 (2015).
Dong, Y.-J. et al. Renal protective effect of sirtuin 1. J. Diabetes Res. 2014, 843786 (2014).
Kitada, M., Kume, S. & Koya, D. Role of sirtuins in kidney disease. Curr. Opin. Nephrol. Hypertens. 23, 75–79 (2014).
Ahn, B.-H. et al. A role for the mitochondrial deacetylase Sirt3 in regulating energy homeostasis. Proc. Natl Acad. Sci. USA 105, 14447–14452 (2008).
Sundaresan, N. R. et al. Sirt3 blocks the cardiac hypertrophic response by augmenting Foxo3a-dependent antioxidant defense mechanisms in mice. J. Clin. Invest. 119, 2758–2771 (2009).
Hafner, A. V. et al. Regulation of the mPTP by SIRT3-mediated deacetylation of CypD at lysine 166 suppresses age-related cardiac hypertrophy. Aging (Albany NY) 2, 914–923 (2010).
Porter, G., Urciuoli, W. R., Brookes, P. S. & Nadtochiy, S. M. SIRT3 deficiency exacerbates ischemia-reperfusion injury: implication for aged hearts. Am. J. Physiol. Heart Circ. Physiol. 306, H1602–H1609 (2014).
Horton, J. L. et al. Mitochondrial protein hyperacetylation in the failing heart. JCI Insight 1, 1–14 (2016).
Alrob, O. A. et al. Obesity-induced lysine acetylation increases cardiac fatty acid oxidation and impairs insulin signalling. Cardiovasc. Res. 103, 485–497 (2014).
Cantó, C. et al. The NAD+ precursor nicotinamide riboside enhances oxidative metabolism and protects against high-fat diet-induced obesity. Cell. Metab. 15, 838–847 (2012).
Liu, B. et al. SIRT4 prevents hypoxia-induced apoptosis in H9c2 cardiomyoblast cells. Cell. Physiol. Biochem. 32, 655–662 (2013).
Luo, Y.-X. et al. Sirt4 accelerates Ang II-induced pathological cardiac hypertrophy by inhibiting manganese superoxide dismutase activity. Eur. Heart J. http://dx.doi.org/10.1093/eurheartj/ehw138 (2016).
Yu, J. et al. Metabolic characterization of a Sirt5 deficient mouse model. Sci. Rep. 3, 2806 (2013).
Boylston, J. A. et al. Characterization of the cardiac succinylome and its role in ischemia-reperfusion injury. J. Mol. Cell. Cardiol. 88, 73–81 (2015). The first publication of the cardiac succinylome; the data suggest a role of SIRT5 in the response to ischaemia–reperfusion injury.
Nishida, Y. et al. SIRT5 regulates both cytosolic and mitochondrial protein malonylation with glycolysis as a major target. Mol. Cell 59, 321–332 (2015).
Sadhukhan, S. et al. Metabolomics-assisted proteomics identifies succinylation and SIRT5 as important regulators of cardiac function. Proc. Natl Acad. Sci. USA 113, 4320–4325 (2016). This study shows that SIRT5-knockout hearts have impaired fatty acid oxidation that contributes to cardiac hypertrophy with ageing.
Koyama, T. et al. SIRT3 attenuates palmitate-induced ROS production and inflammation in proximal tubular cells. Free Radic. Biol. Med. 51, 1258–1267 (2011).
Yang, H. et al. Green tea polyphenols attenuate high-fat diet-induced renal oxidative stress through SIRT3-dependent deacetylation. Biomed. Environ. Sci. 28, 455–459 (2015).
Ugur, S. et al. The renoprotective effect of curcumin in cisplatin-induced nephrotoxicity. Renal Fail. 37, 332–336 (2014).
Nakagawa, T., Lomb, D. J., Haigis, M. C. & Guarente, L. SIRT5 deacetylates carbamoyl phosphate synthetase 1 and regulates the urea cycle. Mol. Cell 137, 560–570 (2009).
Polletta, L. et al. SIRT5 regulation of ammonia-induced autophagy and mitophagy. Autophagy 11, 253–270 (2015).
Hall, A. M. & Unwin, R. J. The not so 'mighty chondrion': emergence of renal diseases due to mitochondrial dysfunction. Nephron Physiol. 105, 1–10 (2007).
Fosslien, E. Mitochondrial medicine — molecular pathology of defective oxidative phosphorylation. Ann. Clin. Lab. Sci. 31, 25–67 (2001).
Fung Lee, C., Garcia-Menendez, L., Karamanlidis, G. & Tian, R. Restoration of NAD redox balance ameliorates pressure overload-induced cardiac hypertrophy and dysfunction via regulation of mitochondrial protein acetylation and permeability transition. Free Radic. Biol. Med. 65, S75 (2013).
Madsen, A. S. et al. Investigating the sensitivity of NAD+-dependent sirtuin deacylation activities to NADH. J. Biol. Chem. 291, 7128–7141 (2016).
Aksoy, P., White, T. A., Thompson, M. & Chini, E. N. Regulation of intracellular levels of NAD: a novel role for CD38. Biochem. Biophys. Res. Commun. 345, 1386–1392 (2006).
Rouleau, M., Patel, A., Hendzel, M. J., Kaufmann, S. H. & Poirier, G. G. PARP inhibition: PARP1 and beyond. Nat. Rev. Cancer 10, 293–301 (2010).
Malavasi, F. et al. Evolution and function of the ADP ribosyl cyclase/CD38 gene family in physiology and pathology. Physiol. Rev. 88, 841–886 (2008).
Xiao, C.-Y. et al. Poly(ADP-ribose) polymerase promotes cardiac remodeling, contractile failure, and translocation of apoptosis-inducing factor in a murine experimental model of aortic banding and heart failure. J. Pharmacol. Exp. Ther. 312, 891–898 (2005).
Andrabi, S. A. et al. Poly(ADP-ribose) polymerase-dependent energy depletion occurs through inhibition of glycolysis. Proc. Natl Acad. Sci. USA 111, 10209–10214 (2014).
Devalaraja-Narashimha, K. & Padanilam, B. J. PARP-1 inhibits glycolysis in ischemic kidneys. J. Am. Soc. Nephrol. 20, 95–103 (2009).
Pirinen, E. et al. Pharmacological Inhibition of poly(ADP-ribose) polymerases improves fitness and mitochondrial function in skeletal muscle. Cell. Metab. 19, 1034–1041 (2014).
Ying, W. NAD+/NADH and NADP+/NADPH in cellular functions and cell death: regulation and biological consequences. Antioxid. Redox Signal. 10, 179–206 (2008).
Rizzuto, R., De Stefani, D., Raffaello, A. & Mammucari, C. Mitochondria as sensors and regulators of calcium signalling. Nat. Rev. Mol. Cell Biol. 13, 566–578 (2012).
Kim, S.-Y., Park, K.-H., Gul, R., Jang, K. Y. & Kim, U.-H. Role of kidney ADP-ribosyl cyclase in diabetic nephropathy. Am. J. Physiol. Renal Physiol. 296, F291–F297 (2009).
Stockand, J. D. & Sansom, S. C. Glomerular mesangial cells: electrophysiology and regulation of contraction. Physiol. Rev. 78, 723–744 (1998).
Thai, T. L. & Arendshorst, W. J. Mice lacking the ADP ribosyl cyclase CD38 exhibit attenuated renal vasoconstriction to angiotensin II, endothelin-1, and norepinephrine. Am. J. Physiol. Renal Physiol. 297, F169–F176 (2009).
Kannt, A., Sicka, K., Kroll, K., Kadereit, D. & Gögelein, H. Selective inhibitors of cardiac ADPR cyclase as novel anti-arrhythmic compounds. Naunyn Schmiedebergs Arch. Pharmacol. 385, 717–727 (2012).
Gan, L. et al. Disruption of CD38 gene enhances cardiac functions by elevating serum testosterone in the male null mice. Life Sci. 89, 491–497 (2011).
Rajakumar, S. V. et al. Deficiency or inhibition of CD73 protects in mild kidney ischemia-reperfusion injury. Transplantation 90, 1260–1264 (2010).
Young, G. S., Choleris, E., Lund, F. E. & Kirkland, J. B. Decreased cADPR and increased NAD+ in the Cd38−/− mouse. Biochem. Biophys. Res. Commun. 346, 188–192 (2006).
Guan, X.-H. et al. CD38 deficiency protects the heart from ischemia/reperfusion injury through activating SIRT1/FOXOs-mediated antioxidative stress pathway. Oxid. Med. Cell. Longev. 2016, 7410257 (2016).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT02300740 (2015).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT02191462 (2014).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT02689882 (2016).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT02712593 (2017).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT02303483 (2016).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT02835664 (2016).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT02812238 (2016).
Harden, A. & Young, W. J. The alcoholic ferment of yeast-juice. Part II. — the conferment of yeast-juice. Proc. R. Soc. Lond. B 78, 369–375 (1906).
Acknowledgements
We acknowledge the scientists whose discoveries were the basis for this review, thank the anonymous peer-reviewers for helpful comments, and apologize to our colleagues whose work we could not cite. We acknowledge funding support from the American Heart Association grants 12SDG8840004 and 12IRG9010008, The Ellison Medical Foundation, Friedreich's Ataxia Research Alliance, the NIH and the NIA grant R01AG045351, the NIH and the NIAAA grant R01AA022146, the Duke Pepper Older Americans Independence Center (OAIC) Program in Ageing Research supported by the National Institute of Ageing (P30AG028716-01), the Duke O'Brien Center for Kidney Research (5P30DK096493-02). K.A.H. was supported by an NIH/NIGMS training grant to Duke University Pharmacological Sciences Training Program (5T32GM007105-40) and is supported by an NIH pre-doctoral fellowship 1F31HL127959. A.S.M. is supported by an NIH pre-doctoral fellowship 1F31HL123275-31.
Author information
Authors and Affiliations
Contributions
All authors researched the data, discussed the content, wrote the text and reviewed or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- Nicotinamide adenine dinucleotide
-
(NAD+). A pyridine dinucleotide and important metabolic cofactor.
- ADPR cyclases
-
Effector molecules that generate calcium-mobilizing second messengers.
- ADP ribosyltransferases
-
Enzymes that transfer the ADPR group of NAD+ as a signal to repair damaged DNA.
- Sirtuins
-
NAD+-dependent protein deacylases that consume NAD+ to remove post-translational acyl modifications from proteins.
- Biosynthetic precursors
-
The biosynthetic precursors of NAD+ are dietary vitamin B3 compounds, including nicotinic acid, nicotinamide, and nicotinamide riboside. These precursors are recycled from the diet and used by tissues to generate NAD+.
- Nicotinamide phosphoribosyltransferase
-
(NAMPT). An enzyme that converts nicotinamide into NMN in the NAD+ salvage pathway.
- Oxidative phosphorylation
-
(OXPHOS). The electron transport pathway that is used by cells to generate ATP.
- Parthanatos
-
A form of programmed cell death that is induced by accumulation of poly(ADP)ribose and the nuclear translocation of apoptosis-inducing factor from mitochondria.
Rights and permissions
About this article
Cite this article
Hershberger, K., Martin, A. & Hirschey, M. Role of NAD+ and mitochondrial sirtuins in cardiac and renal diseases. Nat Rev Nephrol 13, 213–225 (2017). https://doi.org/10.1038/nrneph.2017.5
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneph.2017.5
- Springer Nature Limited
This article is cited by
-
Circular RNA FEACR inhibits ferroptosis and alleviates myocardial ischemia/reperfusion injury by interacting with NAMPT
Journal of Biomedical Science (2023)
-
PBX1-SIRT1 Positive Feedback Loop Attenuates ROS-Mediated HF-MSC Senescence and Apoptosis
Stem Cell Reviews and Reports (2023)
-
Technology and functional insights into the nicotinamide mononucleotide for human health
Applied Microbiology and Biotechnology (2023)
-
Immune checkpoint modulators in cancer immunotherapy: recent advances and emerging concepts
Journal of Hematology & Oncology (2022)
-
Lnc Tmem235 promotes repair of early steroid-induced osteonecrosis of the femoral head by inhibiting hypoxia-induced apoptosis of BMSCs
Experimental & Molecular Medicine (2022)