Key Points
-
Synapses of the mammalian CNS are highly complex structures composed of thousands of distinct proteins. Synaptic proteins are often expressed in variable patterns across different brain regions and neuronal populations, suggesting a high degree of molecular diversity in synapses.
-
Synapse types have long been discriminated on the basis of neurotransmitter molecules, but it is now necessary to recognize much deeper diversity of both synaptic protein composition and function within each traditional neurotransmitter type.
-
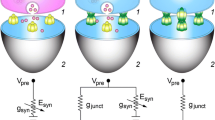
Intra-type synapse diversity reflects many factors, including diversity of presynaptic and postsynaptic parent neurons, activity histories and influences of third-party neurons and glial cells.
-
Intra-type functional diversity is reflected in strength, kinetics, voltage-dependence and plasticity of synaptic transmission. Improving knowledge of the relationships between molecular and functional aspects of diversity is sure to illuminate principals of synaptic circuit function.
-
Proteins identifiable at mature synapses include adhesion molecules that guide selective synaptogenesis during development. Such molecules may encode circuit context and connectivity in the mature brain and aid circuit reconstruction efforts.
-
Many mental and neurological disorders reflect mutations in synaptic proteins expressed in subsets of synapses. A better grasp of synapse molecular diversity is certain to contribute to our understanding of specific brain disorders.
-
New techniques that probe the combinatorial expression of proteins at the level of individual synapses within the brain are bolstering our efforts to link intra-type diversity to distinct physiological and structural properties of synaptic connections and brain circuits.
Abstract
Pioneering studies in the middle of the twentieth century revealed substantial diversity among mammalian chemical synapses and led to a widely accepted classification of synapse type on the basis of neurotransmitter molecule identity. Subsequently, powerful new physiological, genetic and structural methods have enabled the discovery of much deeper functional and molecular diversity within each traditional neurotransmitter type. Today, this deep diversity continues to pose both daunting challenges and exciting new opportunities for neuroscience. Our growing understanding of deep synapse diversity may transform how we think about and study neural circuit development, structure and function.






Similar content being viewed by others
References
Ramón y Cajal, S. The structure and connexions of neurons. Nobelprize.org [online], (1906).
Emes, R. D. et al. Evolutionary expansion and anatomical specialization of synapse proteome complexity. Nature Neurosci. 11, 799–806 (2008).
Takamori, S. et al. Molecular anatomy of a trafficking organelle. Cell 127, 831–846 (2006). A comprehensive study of the protein and lipid composition of synaptic vesicles that culminates in the construction of a detailed structural model of the molecular architecture of these organelles and highlights their molecular complexity.
McPherson, P. S. Proteomic analysis of clathrin-coated vesicles. Proteomics 10, 4025–4039 (2010).
Collins, M. O. et al. Molecular characterization and comparison of the components and multiprotein complexes in the postsynaptic proteome. J. Neurochem. 97 (Suppl. 1), 16–23 (2006).
Dosemeci, A. et al. Composition of the synaptic PSD-95 complex. Mol. Cell Proteomics 6, 1749–1760 (2007).
Peng, J. et al. Semiquantitative proteomic analysis of rat forebrain postsynaptic density fractions by mass spectrometry. J. Biol. Chem. 279, 21003–21011 (2004).
Schrimpf, S. P. et al. Proteomic analysis of synaptosomes using isotope-coded affinity tags and mass spectrometry. Proteomics 5, 2531–2541 (2005).
Filiou, M. D. et al. Profiling of mouse synaptosome proteome and phosphoproteome by IEF. Electrophoresis 31, 1294–1301 (2010).
Cheng, D. et al. Relative and absolute quantification of postsynaptic density proteome isolated from rat forebrain and cerebellum. Mol. Cell. Proteomics 5, 1158–1170 (2006).
Grønborg, M. et al. Quantitative comparison of glutamatergic and GABAergic synaptic vesicles unveils selectivity for few proteins including MAL2, a novel synaptic vesicle protein. J. Neurosci. 30, 2–12 (2010).
Selimi, F., Cristea, I. M., Heller, E., Chait, B. T. & Heintz, N. Proteomic studies of a single CNS synapse type: the parallel fiber/purkinje cell synapse. PLoS Biol. 7, e83 (2009). This study exploited the specific expression of GluRA2 at parallel fibre–Purkinje cell synapses to purify this synapse population for proteomic analysis, which identified dozens of additional proteins highly enriched within this specific anatomical connection.
Gong, S. et al. A gene expression atlas of the central nervous system based on bacterial artificial chromosomes. Nature 425, 917–925 (2003).
Lein, E. S. et al. Genome-wide atlas of gene expression in the adult mouse brain. Nature 445, 168–176 (2007). An extensive study using high-throughput in situ hybridization and image analysis to map the expression of over 20,000 genes across the entire mouse brain.
Fremeau, R. T. Jr et al. The expression of vesicular glutamate transporters defines two classes of excitatory synapse. Neuron 31, 247–260 (2001). A study providing the first evidence that glutamatergic synapses can be divided into two synapse subtypes on the basis of their expression of the vesicular glutamate transporters VGluT1 and VGluT2.
Ferraguti, F. & Shigemoto, R. Metabotropic glutamate receptors. Cell Tissue Res. 326, 483–504 (2006).
Shigemoto, R. et al. Target-cell-specific concentration of a metabotropic glutamate receptor in the presynaptic active zone. Nature 381, 523–525 (1996).
Miyata, M. Distinct properties of corticothalamic and primary sensory synapses to thalamic neurons. Neurosci. Res. 59, 377–382 (2007).
Fioravante, D. & Regehr, W. G. Short-term forms of presynaptic plasticity. Curr. Opin. Neurobiol. 21, 269–274 (2011).
Wang, Y. et al. Heterogeneity in the pyramidal network of the medial prefrontal cortex. Nature Neurosci. 9, 534–542 (2006).
Groh, A. et al. Cell-type specific properties of pyramidal neurons in neocortex underlying a layout that is modifiable depending on the cortical area. Cereb. Cortex 20 826–836 2009).
Blatow, M., Caputi, A., Burnashev, N., Monyer, H. & Rozov, A. Ca2+ buffer saturation underlies paired pulse facilitation in calbindin-D28k-containing terminals. Neuron 38, 79–88 (2003).
Burnashev, N. & Rozov, A. Presynaptic Ca2+ dynamics, Ca2+ buffers and synaptic efficacy. Cell Calcium 37, 489–495 (2005).
Caillard, O. et al. Role of the calcium-binding protein parvalbumin in short-term synaptic plasticity. Proc. Natl Acad. Sci USA 97, 13372–13377 (2000).
Reyes, A. et al. Target-cell-specific facilitation and depression in neocortical circuits. Nature Neurosci. 1, 279–285 (1998).
De Gois, S. et al. Identification of endophilins 1 and 3 as selective binding partners for VGLUT1 and their co-localization in neocortical glutamatergic synapses: implications for vesicular glutamate transporter trafficking and excitatory vesicle formation. Cell. Mol. Neurobiol. 26, 679–693 (2006).
Voglmaier, S. M. et al. Distinct endocytic pathways control the rate and extent of synaptic vesicle protein recycling. Neuron 51, 71–84 (2006).
Mutch, S. A. et al. Protein quantification at the single vesicle level reveals that a subset of synaptic vesicle proteins are trafficked with high precision. J. Neurosci. 31, 1461–1470 (2011).
Bragina, L., Giovedi, S., Barbaresi, P., Benfenati, F. & Conti, F. Heterogeneity of glutamatergic and GABAergic release machinery in cerebral cortex: analysis of synaptogyrin, vesicle-associated membrane protein, and syntaxin. Neuroscience 165, 934–943 (2010).
Bragina, L. et al. Heterogeneity of glutamatergic and GABAergic release machinery in cerebral cortex. Neuroscience 146, 1829–1840 (2007).
Isaac, J. T. R., Ashby, M. C. & McBain, C. J. The role of the GluR2 subunit in AMPA receptor function and synaptic plasticity. Neuron 54, 859–871 (2007).
Lu, W. et al. Subunit composition of synaptic AMPA receptors revealed by a single-cell genetic approach. Neuron 62, 254–268 (2009).
Savtchouk, I. & Liu, S. J. Remodeling of synaptic AMPA receptor subtype alters the probability and pattern of action potential firing. J. Neurosci. 31, 501–511 (2011).
Cull-Candy, S. G. & Leszkiewicz, D. N. Role of distinct NMDA receptor subtypes at central synapses. Sci. STKE, re16 (2004).
Monyer, H., Burnashev, N., Laurie, D. J., Sakmann, B. & Seeburg, P. H. Developmental and regional expression in the rat brain and functional properties of four NMDA receptors. Neuron 12, 529–540 (1994).
Brickley, S. G., Misra, C., Mok, M. H. S., Mishina, M. & Cull-Candy, S. G. NR2B and NR2D subunits coassemble in cerebellar Golgi cells to form a distinct NMDA receptor subtype restricted to extrasynaptic sites. J. Neurosci. 23, 4958–4966 (2003).
Granger, A. J., Gray, J. A., Lu, W. & Nicoll, R. A. Genetic analysis of neuronal ionotropic glutamate receptor subunits. J. Physiol. 589, 4095–4101 (2011).
Luscher, B., Fuchs, T. & Kilpatrick, C. L. GABAA receptor trafficking-mediated plasticity of inhibitory synapses. Neuron 70, 385–409 (2011).
Sassoè-Pognetto, M. Understanding the molecular diversity of GABAergic synapses. Front. Cell. Neurosci. 5, 4 (2011).
Klausberger, T., Roberts, J. D. B. & Somogyi, P. Cell type- and input-specific differences in the number and subtypes of synaptic GABAA receptors in the hippocampus. J. Neurosci. 22, 2513–2521 (2002).
Freund, T. F. & Katona, I. Perisomatic inhibition. Neuron 56, 33–42 (2007).
Rudolph, U., Crestani, F. & Möhler, H. GABAA receptor subtypes: dissecting their pharmacological functions. Trends Pharmacol. Sci. 22, 188–194 (2001).
Hull, C., Isaacson, J. S. & Scanziani, M. Postsynaptic mechanisms govern the differential excitation of cortical neurons by thalamic inputs. J. Neurosci. 29, 9127–9136 (2009). This study demonstrated that thalamic afferents produce distinct effects on different cortical target neurons as a result of differences in receptor isoform expression. This report is one of the rare instances where distinct physiological properties have been convincingly linked to a clear molecular difference between synapse populations.
Burnashev, N., Monyer, H., Seeburg, P. H. & Sakmann, B. Divalent ion permeability of AMPA receptor channels is dominated by the edited form of a single subunit. Neuron 8, 189–198 (1992).
Jonas, P. Differences in Ca2+ permeability of AMPA-type glutamate receptor channels in neocortical neurons caused by differential GluR-B subunit expression. Neuron 12, 1281–1289 (1994).
Hestrin, S. Different glutamate receptor channels mediate fast excitatory synaptic currents in inhibitory and excitatory cortical neurons. Neuron 11, 1083–1091 (1993).
Vicini, S. et al. Functional and pharmacological differences between recombinant N-methyl-D-aspartate receptors. J. Neurophysiol. 79, 555–566 (1998).
Kumar, S. S. & Huguenard, J. R. Pathway-specific differences in subunit composition of synaptic NMDA receptors on pyramidal neurons in neocortex. J. Neurosci. 23, 10074–10083 (2003). This study used careful physiological stimulation of long-distance callosal and local intracortical axons along with extensive pharmacological manipulation to demonstrate that synapses of the same postsynaptic pyramidal neurons express different NMDAR subunit combinations depending on their presynaptic source, and went further to demonstrate important functional ramifications of this pathway-specific difference in receptor subtype expression.
Nyíri, G., Freund, T. F. & Somogyi, P. Input-dependent synaptic targeting of α2-subunit-containing GABAA receptors in synapses of hippocampal pyramidal cells of the rat. Eur. J. Neurosci. 13, 428–442 (2001).
Kato, A. S., Gill, M. B., Yu, H., Nisenbaum, E. S. & Bredt, D. S. TARPs differentially decorate AMPA receptors to specify neuropharmacology. Trends Neurosci. 33, 241–248 (2010).
Béïque, J.-C. et al. Synapse-specific regulation of AMPA receptor function by PSD-95. Proc. Natl Acad. Sci. USA 103, 19535–19540 (2006).
Elias, G. M. & Nicoll, R. A. Synaptic trafficking of glutamate receptors by MAGUK scaffolding proteins. Trends Cell Biol. 17, 343–352 (2007).
Costa-Mattioli, M., Sossin, W. S., Klann, E. & Sonenberg, N. Translational control of long-lasting synaptic plasticity and memory. Neuron 61, 10–26 (2009).
Ho, V. M., Lee, J. & Martin, K. C. The cell biology of synaptic plasticity. Science 334, 623–628 (2011).
Valles, A. et al. Genomewide analysis of rat barrel cortex reveals time- and layer-specific mRNA expression changes related to experience-dependent plasticity. J. Neurosci. 31, 6140–6158 (2011). This genomic study used a combination of microarrays and in situ hybridization to reveal the time course and laminar patterns of gene expression changes in rodent whisker somatosensory cortex following environmental enrichment. This provided a powerful overview of the genetic and molecular basis for experience-dependent learning and memory.
Shepherd, J. D. & Bear, M. F. New views of Arc, a master regulator of synaptic plasticity. Nature Neurosci. 14, 279–284 (2011).
Bliss, T. & Lømo, T. Long-lasting potentiation of synaptic transmission in the dentate gyrus of the rat following selective depletion of monoamines. J. Physiol. 232, 331–356 (1973).
Dudek, S. M. & Bear, M. F. Homosynaptic long-term depression in area CA1 of hippocampus and effects of N-methyl-D-aspartate receptor blockade. Proc. Natl Acad. Sci. USA 89, 4363–4367 (1992).
Malenka, R. C. & Bear, M. F. LTP and LTD: an embarrassment of riches. Neuron 44, 5–21 (2004).
Busetto, G., Higley, M. J. & Sabatini, B. L. Developmental presence and disappearance of postsynaptically silent synapses on dendritic spines of rat layer 2/3 pyramidal neurons. J. Physiol. 586, 1519–1527 (2008).
Ashby, M. C. & Isaac, J. T. R. Maturation of a recurrent excitatory neocortical circuit by experience-dependent unsilencing of newly formed dendritic spines. Neuron 70, 510–521 (2011).
Emond, M. R. et al. AMPA receptor subunits define properties of state-dependent synaptic plasticity. J. Physiol. 588, 1929–1946 (2010).
Kelly, L., Farrant, M. & Cull-Candy, S. G. Synaptic mGluR activation drives plasticity of calcium-permeable AMPA receptors. Nature Neurosci. 12, 593–601 (2009).
Miyashita, T., Kubik, S., Lewandowski, G. & Guzowski, J. F. Networks of neurons, networks of genes: an integrated view of memory consolidation. Neurobiol. Learn. Mem. 89, 269–284 (2008).
Rial Verde, E. M., Lee-Osbourne, J., Worley, P. F., Malinow, R. & Cline, H. T. Increased expression of the immediate-early gene arc/arg3.1 reduces AMPA receptor-mediated synaptic transmission. Neuron 52, 461–474 (2006).
Béïque, J.-C., Na, Y., Kuhl, D., Worley, P. F. & Huganir, R. L. Arc-dependent synapse-specific homeostatic plasticity. Proc. Natl Acad. Sci. USA 108, 816–821 (2011).
Shi, S., Hayashi, Y., Esteban, J. A. & Malinow, R. Subunit-specific rules governing AMPA receptor trafficking to synapses in hippocampal pyramidal neurons. Cell 105, 331–343 (2001).
Li, P., Rudolph, U. & Huntsman, M. M. Long-term sensory deprivation selectively rearranges functional inhibitory circuits in mouse barrel cortex. Proc. Natl Acad. Sci. USA 106, 12156–12161 (2009).
Feldman, D. E. Synaptic mechanisms for plasticity in neocortex. Annu. Rev. Neurosci. 32, 33–55 (2009).
Sjöstrom, P. J., Rancz, E. A., Roth, A. & Häusser, M. Dendritic excitability and synaptic plasticity. Physiol. Rev. 88, 769–840 (2008).
Maffei, A. The many forms and functions of long term plasticity at GABAergic synapses. Neural Plast. 2011, 254724 (2011).
Egger, V., Feldmeyer, D. & Sakmann, B. Coincidence detection and changes of synaptic efficacy in spiny stellate neurons in rat barrel cortex. Nature Neurosci. 2, 1098–1105 (1999).
Lüscher, C. & Huber, K. M. Group 1 mGluR-dependent synaptic long-term depression: mechanisms and implications for circuitry and disease. Neuron 65, 445–459 (2010).
Chevaleyre, V., Takahashi, K.A. & Castillo, P. E. Endocannabinoid-mediated synaptic plasticity in the CNS. Annu. Rev. Neurosci. 29, 37–76 (2006).
Renger, J. J. et al. Experience-dependent plasticity without long-term depression by type 2 metabotropic glutamate receptors in developing visual cortex. Proc. Natl Acad. Sci. USA 99, 1041–1046 (2002).
Crozier, R.A., Wang, Y., Liu, C.-H. & Bear, M. F. Deprivation-induced synaptic depression by distinct mechanisms in different layers of mouse visual cortex. Proc. Natl Acad. Sci. USA 104, 1383–1388 (2007).
Liu, C.-H., Heynen, A. J., Shuler, M. G. H. & Bear, M. F. Cannabinoid receptor blockade reveals parallel plasticity mechanisms in different layers of mouse visual cortex. Neuron 58, 340–345 (2008).
Coba, M. P. et al. Neurotransmitters drive combinatorial multistate postsynaptic density networks. Sci. Signal. 2, ra19 (2009).
Williams, M. E., de Wit, J. & Ghosh, A. Molecular mechanisms of synaptic specificity in developing neural circuits. Neuron 68, 9–18 (2010).
Zipursky, S. L. & Sanes, J. R. Chemoaffinity revisited: dscams, protocadherins, and neural circuit assembly. Cell 143, 343–353 (2010).
Dalva, M. B., McClelland, A. C. & Kayser, M. S. Cell adhesion molecules: signalling functions at the synapse. Nature Rev. Neurosci. 8, 206–220 (2007).
Biederer, T. & Stagi, M. Signaling by synaptogenic molecules. Curr. Opin. Neurobiol. 18, 261–269 (2008).
Sudhof, T. C. Neuroligins and neurexins link synaptic function to cognitive disease. Nature 455, 903–911 (2008).
Takeichi, M. The cadherin superfamily in neuronal connections and interactions. Nature Rev. Neurosci. 8, 11–20 (2007).
Arikkath, J. & Reichardt, L. F. Cadherins and catenins at synapses: roles in synaptogenesis and synaptic plasticity. Trends Neurosci. 31, 487–494 (2008).
Ango, F. et al. Ankyrin-based subcellular gradient of neurofascin, an immunoglobulin family protein, directs GABAergic innervation at Purkinje axon initial segment. Cell 119, 257–272 (2004). A study in which knockout mice and RNA interference were used to demonstrate the role of the adhesion protein neurofascin 186 and the scaffold protein ankyrin G in the formation of a specific synapse that is formed between basket cells and Purkinje cells and is localized to a specific region of the Purkinje cell, the AIS.
Zonta, B. et al. A critical role for Neurofascin in regulating action potential initiation through maintenance of the axon initial segment. Neuron 69, 945–956 (2011).
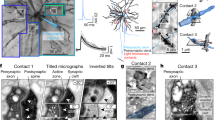
Anderson, J. R. et al. Exploring the retinal connectome. Mol. Vis. 17, 355–379 (2011). This is the first practical connectome data set from a mammalian retina. The data set spans the inner nuclear, inner plexiform and ganglion cell layers and contains six molecular markers, including an in vivo activity marker. Initial exploration of this set confirmed well-known connections and revealed new features of retinal organization, including much higher diversity of synaptic connections.
Anderson, J. R. et al. A computational framework for ultrastructural mapping of neural circuitry. PLoS Biol. 7, e1000074 (2009).
Helmstaedter, M., Briggman, K. L. & Denk, W. High-accuracy neurite reconstruction for high-throughput neuroanatomy. Nature Neurosci. 14, 1081–1088 (2011).
Bock, D. D. et al. Network anatomy and in vivo physiology of visual cortical neurons. Nature 471, 177–182 (2011).
Mishchenko, Y. et al. Ultrastructural analysis of hippocampal neuropil from the connectomics perspective. Neuron 67, 1009–1020 (2010).
Briggman, K. L., Helmstaedter, M. & Denk, W. Wiring specificity in the direction-selectivity circuit of the retina. Nature 471, 183–188 (2011).
Kleinfeld, D. et al. Large-scale automated histology in the pursuit of connectomes. J. Neurosci. 31, 16125–16138 (2011).
Ushkaryov, Y. A. & Sudhof, T. C. Neurexin III α: extensive alternative splicing generates membrane-bound and soluble forms. Proc. Natl Acad. Sci. USA 90, 6410–6414 (1993).
Ushkaryov, Y. A., Petrenko, A. G., Geppert, M. & Sudhof, T. C. Neurexins: synaptic cell surface proteins related to the α-latrotoxin receptor and laminin. Science 257, 50–56 (1992).
Ullrich, B., Ushkaryov, Y. A. & Sudhof, T. C. Cartography of neurexins: more than 1000 isoforms generated by alternative splicing and expressed in distinct subsets of neurons. Neuron 14, 497–507 (1995).
Song, J. Y., Ichtchenko, K., Sudhof, T. C. & Brose, N. Neuroligin 1 is a postsynaptic cell-adhesion molecule of excitatory synapses. Proc. Natl Acad. Sci. USA 96, 1100–1105 (1999).
Varoqueaux, F., Jamain, S. & Brose, N. Neuroligin 2 is exclusively localized to inhibitory synapses. Eur. J. Cell Biol. 83, 449–456 (2004).
Hoon, M. et al. Neuroligin-4 is localized to glycinergic postsynapses and regulates inhibition in the retina. Proc. Natl Acad. Sci. USA 108, 3053–3058 (2011).
Budreck, E. C. & Scheiffele, P. Neuroligin-3 is a neuronal adhesion protein at GABAergic and glutamatergic synapses. Eur. J. Neurosci. 26, 1738–1748 (2007).
Kohmura, N. et al. Diversity revealed by a novel family of cadherins expressed in neurons at a synaptic complex. Neuron 20, 1137–1151 (1998).
Wu, Q. & Maniatis, T. A striking organization of a large family of human neural cadherin-like cell adhesion genes. Cell 97, 779–790 (1999).
Zou, C., Huang, W., Ying, G. & Wu, Q. Sequence analysis and expression mapping of the rat clustered protocadherin gene repertoires. Neuroscience 144, 579–603 (2007).
Junghans, D. et al. Postsynaptic and differential localization to neuronal subtypes of protocadherin β16 in the mammalian central nervous system. Eur. J. Neurosci. 27, 559–571 (2008).
Ascoli, G. A. et al. Petilla terminology: nomenclature of features of GABAergic interneurons of the cerebral cortex. Nature Rev. Neurosci. 9, 557–568 (2008).
Markram, H. et al. Interneurons of the neocortical inhibitory system. Nature Rev. Neurosci. 5, 793–807 (2004).
Miyazaki, T., Fukaya, M., Shimizu, H. & Watanabe, M. Subtype switching of vesicular glutamate transporters at parallel fibre-Purkinje cell synapses in developing mouse cerebellum. Eur. J. Neurosci. 17, 2563–2572 (2003).
Bayes, A. et al. Characterization of the proteome, diseases and evolution of the human postsynaptic density. Nature Neurosci. 14, 19–21 (2010).
Jamain, S., Betancur, C., Giros, B., Leboyer, M. & Bourgeron, T. [Genetics of autism: from genome scans to candidate genes]. Med. Sci. (Paris) 19, 1081–1090 (2003) (in French).
Walsh, C. A., Morrow, E. M. & Rubenstein, J. L. Autism and brain development. Cell 135, 396–400 (2008).
Pinto, D. et al. Functional impact of global rare copy number variation in autism spectrum disorders. Nature 466, 368–372 (2010).
Tabuchi, K. et al. A neuroligin-3 mutation implicated in autism increases inhibitory synaptic transmission in mice. Science 318, 71–76 (2007).
Etherton, M. et al. Autism-linked neuroligin-3 R451C mutation differentially alters hippocampal and cortical synaptic function. Proc. Natl Acad. Sci. USA 108, 13764–13769 (2011).
Jamain, S. et al. Reduced social interaction and ultrasonic communication in a mouse model of monogenic heritable autism. Proc. Natl Acad. Sci. USA 105, 1710–1715 (2008).
Peca, J. et al. Shank3 mutant mice display autistic-like behaviours and striatal dysfunction. Nature 472, 437–442 (2011).
St. Clair, D. et al. Association within a family of a balanced autosomal translocation with major mental illness. Lancet 336, 13–16 (1990).
Millar, J. K. et al. Disruption of two novel genes by a translocation co-segregating with schizophrenia. Hum. Mol. Genet. 9, 1415–1423 (2000).
Brandon, N. J. et al. Understanding the role of DISC1 in psychiatric disease and during normal development. J. Neurosci. 29, 12768–12775 (2009).
Kirkpatrick, B. et al. DISC1 immunoreactivity at the light and ultrastructural level in the human neocortex. J. Comp. Neurol. 497, 436–450 (2006).
Glantz, L. A. & Lewis, D. A. Decreased dendritic spine density on prefrontal cortical pyramidal neurons in schizophrenia. Arch. Gen. Psychiatry 57, 65–73 (2000).
Kvajo, M. et al. A mutation in mouse Disc1 that models a schizophrenia risk allele leads to specific alterations in neuronal architecture and cognition. Proc. Natl Acad. Sci. USA 105, 7076–7081 (2008).
Hayashi-Takagi, A. et al. Disrupted-in-Schizophrenia 1 (DISC1) regulates spines of the glutamate synapse via Rac1. Nature Neurosci. 13, 327–332 (2010).
Schurov, I. L., Handford, E. J., Brandon, N. J. & Whiting, P. J. Expression of disrupted in schizophrenia 1 (DISC1) protein in the adult and developing mouse brain indicates its role in neurodevelopment. Mol. Psychiatry 9, 1100–1110 (2004).
Palade, G. E. Electron microscope observations of internneuronal and neuromuscular synapses. Anat. Rec. 118, 335–336 (1954).
Chen, X. et al. Organization of the core structure of the postsynaptic density. Proc. Natl Acad. Sci. USA 105, 4453–4458 (2008).
Jones, B. W. et al. Retinal remodeling in the Tg P347L rabbit, a large-eye model of retinal degeneration. J. Comp. Neurol. 519, 2713–2733 (2011).
Micheva, K. D. & Smith, S. J. Array tomography: a new tool for imaging the molecular architecture and ultrastructure of neural circuits. Neuron 55, 25–36 (2007). Using high-throughput array tomography, individual cortical synapses in situ were characterized by the presence of 18 molecular markers. An antibody panel was developed to reliably distinguish between glutamatergic and GABAergic synapses and to begin exploring intra-type diversity.
Micheva, K. D., Busse, B., Weiler, N. C., O'Rourke, N. & Smith, S. J. Single-synapse analysis of a diverse synapse population: proteomic imaging methods and markers. Neuron 68, 639–653 (2010).
Hell, S. W. & Wichmann, J. Breaking the diffraction resolution limit by stimulated emission: stimulated-emission-depletion fluorescence microscopy. Opt. Lett. 19, 780–782 (1994).
Rust, M. J., Bates, M. & Zhuang, X. Sub-diffraction-limit imaging by stochastic optical reconstruction microscopy (STORM). Nature Methods 3, 793–795 (2006).
Betzig, E. et al. Imaging intracellular fluorescent proteins at nanometer resolution. Science 313, 1642–1645 (2006).
Hess, S. T., Girirajan, T. P. & Mason, M. D. Ultra-high resolution imaging by fluorescence photoactivation localization microscopy. Biophys. J. 91, 4258–4272 (2006).
Dani, A., Huang, B., Bergan, J., Dulac, C. & Zhuang, X. Superresolution imaging of chemical synapses in the brain. Neuron 68, 843–856 (2010). The distributions of ten pre- and postsynaptic proteins at brain synapses were measured with nanometre precision using three-colour three-dimensional STORM imaging. Variations in neurotransmitter receptor composition and localization were observed among synapses and across different brain regions.
Buckers, J., Wildanger, D., Vicidomini, G., Kastrup, L. & Hell, S. W. Simultaneous multi-lifetime multi-color STED imaging for colocalization analyses. Opt. Express 19, 3130–3143 (2011).
Bates, M., Huang, B., Dempsey, G. T. & Zhuang, X. Multicolor super-resolution imaging with photo-switchable fluorescent probes. Science 317, 1749–1753 (2007).
Sugino, K. et al. Molecular taxonomy of major neuronal classes in the adult mouse forebrain. Nature Neurosci. 9, 99–107 (2006).
Lichtman, J. W. & Denk, W. The big and the small: challenges of imaging the brain's circuits. Science 334, 618–623 (2011).
Jain, V., Seung, H. S. & Turaga, S. C. Machines that learn to segment images: a crucial technology for connectomics. Curr. Opin. Neurobiol. 20, 653–666 (2010).
Ramón y Cajal, S. ¿Neuronismo o reticularismo? Las pruebas objetivas de la unidad anatómica de las células nerviosas. Archivos de Neurobiología 13 1–144 (1933) (in Spanish).
Micheva, K. D. & Beaulieu, C. An anatomical substrate for experience-dependent plasticity of the rat barrel field cortex. Proc. Natl Acad. Sci. USA 92, 11834–11838 (1995).
Stewart, W. W. Functional connections between cells as revealed by dye-coupling with a highly fluorescent naphthalimide tracer. Cell 14, 741–759 (1978).
Grutzendler, J., Kasthuri, N. & Gan, W. B. Long-term dendritic spine stability in the adult cortex. Nature 420, 812–816 (2002).
Trachtenberg, J. T. et al. Long-term in vivo imaging of experience-dependent synaptic plasticity in adult cortex. Nature 420, 788–794 (2002).
Xu, T. et al. Rapid formation and selective stabilization of synapses for enduring motor memories. Nature 462, 915–919 (2009).
Yang, G., Pan, F. & Gan, W. B. Stably maintained dendritic spines are associated with lifelong memories. Nature 462, 920–924 (2009).
Matsuzaki, M. et al. Dendritic spine geometry is critical for AMPA receptor expression in hippocampal CA1 pyramidal neurons. Nature Neurosci. 4, 1086–1092 (2001).
Acknowledgements
The authors are grateful to F. Collman for many helpful discussions. The work was supported by the Gatsby Charitable Trust (London, UK), The Howard Hughes Medical Institute (Maryland, USA) (Collaborative Innovation Award #43667), the Mathers Foundation (New York, USA) and grants from the National Institute of Neurological Diseases and Stroke (Maryland, USA) (1R21NS063210, 1R01NS075252 and 1R01NS077601).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Stanford has licensed a Smith and Micheva array tomography patent to Aratome, LLC, Menlo Park, California, USA. Both Smith and Micheva have founder's equity shares in Aratome. Nancy A. O'Rourke and Nicholas C. Weiler declare no competing interests.
Related links
Glossary
- Proteomic
-
Relating to the study of the proteome, which is the entire set of proteins expressed by an organism, tissue, cell or subcellular organelle. A variety of large-scale techniques are used, such as mass spectrometry, immunolabelling or tagging of proteins and yeast two-hybrid screens.
- Synaptosomes
-
Artificially formed membranous structures that are generated by the subcellular fractionation of brain tissue homogenates. The synaptosome contains most of the presynaptic terminal, including synaptic vesicles and mitochondria, as well as the postsynaptic density and adjacent postsynaptic membrane.
- Isoforms
-
Alternative forms of the same protein generated from either related genes or from alternate splicing of the same gene.
- Short-term facilitation
-
Increase in the amplitude of synaptic transmission over multiple stimuli on the scale of milliseconds, thought to result from frequency-dependent build-up of presynaptic calcium, which increases the release probability for upcoming spikes.
- Short-term depression
-
Decrease in the amplitude of synaptic transmission with repeated stimulation on the scale of milliseconds, thought to result from frequency-dependent depletion of fusion-ready vesicles, which decreases the release probability for upcoming spikes.
- Release probability
-
The probability that a single presynaptic spike will result in the release of a vesicle of neurotransmitter into the synaptic cleft. Release probability is determined by multiple presynaptic factors.
- Long-term potentiation
-
(LTP). Long-lasting increase in synaptic strength between neurons, usually resulting from synchronous or temporally coordinated pre- and postsynaptic activity.
- Long-term depression
-
(LTD). Long-lasting weakening of synaptic strength between neurons, often resulting from asynchronous pre- and postsynaptic activity.
- Pinceau synapses
-
Synapses shaped like a paintbrush ('pinceau' in French) that are formed at the base of the Purkinje cell axon by cerebellar basket cells.
- Autism spectrum disorders
-
(ASDs). A group of complex neurodevelopmental disorders characterized by difficulties in social interaction, poor verbal and non-verbal communication and abnormal repetitive behaviours.
- Golgi stain
-
A technique based on precipitations of metallic salts within cells and used for visualization of sparse subsets of neurons and glial cells in their entirety.
- EM tomography
-
A method for the three-dimensional reconstruction of objects from a series of projection images that are recorded with a transmission electron microscope. It offers the opportunity to obtain spatial information on structural arrangements of cellular components.
- Ultrathin sections
-
Histological sections of resin-embedded or frozen tissue with a thickness of 30–200nm. Such sections are typically used for electron microscopy.
- Confocal microscopy
-
A fluorescence imaging technique that increases resolution through 'optical sectioning'. To attain optical sectioning, excitation light is scanned across an object, illuminating a single point at a time, and the emitted fluorescence light is detected through a pinhole aperture, limiting the light originating from outside the focal plane.
- Two-photon microscopy
-
A form of microscopy in which a fluorochrome that would normally be excited by a single photon is stimulated quasi-simultaneously by two photons of lower energy. Under these conditions, fluorescence increases as a function of the square of the light intensity, and decreases as the fourth power of the distance from the focus. Because of this behaviour, only fluorochrome molecules near the plane of focus are excited, greatly reducing light scattering and photodamage of the sample.
Rights and permissions
About this article
Cite this article
O'Rourke, N., Weiler, N., Micheva, K. et al. Deep molecular diversity of mammalian synapses: why it matters and how to measure it. Nat Rev Neurosci 13, 365–379 (2012). https://doi.org/10.1038/nrn3170
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrn3170
- Springer Nature Limited
This article is cited by
-
Transcriptional diversity in specific synaptic gene sets discriminates cortical neuronal identity
Biology Direct (2023)
-
Determinants of synapse diversity revealed by super-resolution quantal transmission and active zone imaging
Nature Communications (2022)
-
Complex regulation of Gephyrin splicing is a determinant of inhibitory postsynaptic diversity
Nature Communications (2022)
-
(2R,6R)-hydroxynorketamine rapidly potentiates hippocampal glutamatergic transmission through a synapse-specific presynaptic mechanism
Neuropsychopharmacology (2020)
-
Neurexin 3 transmembrane and soluble isoform expression and splicing haplotype are associated with neuron inflammasome and Alzheimer’s disease
Alzheimer's Research & Therapy (2019)