Abstract
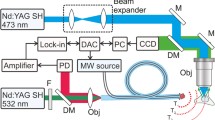
Recently, optical stimulation1,2,3 has begun to unravel the neuronal processing that controls certain animal behaviours4,5. However, optical approaches are limited by the inability of visible light to penetrate deep into tissues. Here, we show an approach based on radio-frequency magnetic-field heating of nanoparticles to remotely activate temperature-sensitive cation channels in cells. Superparamagnetic ferrite nanoparticles were targeted to specific proteins on the plasma membrane of cells expressing TRPV1, and heated by a radio-frequency magnetic field. Using fluorophores as molecular thermometers, we show that the induced temperature increase is highly localized. Thermal activation of the channels triggers action potentials in cultured neurons without observable toxic effects. This approach can be adapted to stimulate other cell types and, moreover, may be used to remotely manipulate other cellular machinery for novel therapeutics.




Similar content being viewed by others
References
Zemelman, B. V., Lee, G. A., Ng, M. & Miesenbock, G. Selective photostimulation of genetically charged neurons. Neuron 33, 15–22 (2002).
Boyden, E. S., Zhang, F., Bamberg, E., Nagel, G. & Deisseroth, K. Millisecond-timescale, genetically targeted optical control of neural activity. Nature Neurosci. 8, 1263–1268 (2005).
Banghart, M., Borges, K., Isacoff, E., Trauner, D. & Kramer, R. H. Light-activated ion channels for remote control of neuronal firing. Nature Neurosci. 7, 1381–1386 (2004).
Tsai, H. C. et al. Phasic firing in dopaminergic neurons is sufficient for behavioral conditioning. Science 324, 1080–1084 (2009).
Gradinaru, V., Mogri, M., Thompson, K. R., Henderson, J. M. & Deisseroth, K. Optical deconstruction of parkinsonian neural circuitry. Science 324, 354–359 (2009).
Wang, N., Butler, J. P. & Ingber, D. E. Mechanotransduction across the cell surface and through the cytoskeleton. Science 260, 1124–1127 (1993).
Hughes, S., McBain, S., Dobson, J. & El Haj, A. J. Selective activation of mechanosensitive ion channels using magnetic particles. J. R. Soc. Interface 5, 855–863 (2008).
Mannix, R. J. et al. Nanomagnetic actuation of receptor-mediated signal transduction. Nature Nanotech. 3, 36–40 (2008).
Caterina, M. J. et al. The capsaicin receptor: a heat-activated ion channel in the pain pathway. Nature 389, 816–824 (1997).
Tominaga, M. et al. The cloned capsaicin receptor integrates multiple pain-producing stimuli. Neuron 21, 531–543 (1998).
Lima, S. Q. & Miesenbock, G. Remote control of behavior through genetically targeted photostimulation of neurons. Cell 121, 141–152 (2005).
Zeng, H., Rice, P. M., Wang, S. X. & Sun, S. Shape-controlled synthesis and shape-induced texture of MnFe2O4 nanoparticles. J. Am. Chem. Soc. 126, 11458–11459 (2004).
Sun, S. et al. Monodisperse MFe2O4 (M = Fe, Co, Mn) nanoparticles. J. Am. Chem. Soc. 126, 273–279 (2004).
Jun, Y.-W. et al. Nanoscale size effect of magnetic nanocrystals and their utilization for cancer diagnosis via magnetic resonance imaging. J. Am. Chem. Soc. 127, 5732–5733 (2005).
Lee, J.-H. et al. Artificially engineered magnetic nanoparticles for ultra-sensitive molecular imaging. Nature Med. 13, 95–99 (2007).
Duhr, S., Arduini, S. & Braun, D. Thermophoresis of DNA determined by microfluidic fluorescence. Eur. Phys. J. E 15, 277–286 (2004).
Kalab, P., Pralle, A., Isacoff, E. Y., Heald, R. & Weis, K. Analysis of a RanGTP-regulated gradient in mitotic somatic cells. Nature 440, 697–701 (2006).
Karstens, T. & Kobs, K. Rhodamine B and Rhodamine 101 as reference substances for fluorescence quantum yield measurements. J. Phys. Chem. 84, 1871–1872 (1980).
Howarth, M., Takao, K., Hayashi, Y. & Ting, A. Y. Targeting quantum dots to surface proteins in living cells with biotin ligase. Proc. Natl Acad. Sci. USA 102, 7583–7588 (2005).
Howarth, M. & Ting, A. Y. Imaging proteins in live mammalian cells with biotin ligase and monovalent streptavidin. Nature Protoc. 3, 534–545 (2008).
Hilger, I. et al. Evaluation of temperature increase with different amounts of magnetite in liver tissue samples. Invest. Radiol. 32, 705–712 (1997).
Hergt, R. et al. Maghemite nanoparticles with very high AC-losses for application in RF-magnetic hyperthermia. J. Magn. Magn. Mater. 270, 345–357 (2004).
Mank, M. et al. A FRET-based calcium biosensor with fast signal kinetics and high fluorescence change. Biophys. J. 90, 1790–1796 (2006).
Hübener, G., Lambacher, A. & Fromherz, P. Anellated hemicyanine dyes with large symmetrical solvatochromism of absorption and fluorescence. J. Phys. Chem. B 107, 7896–7902 (2003).
Wittenburg, N. & Baumeister, R. Thermal avoidance in Caenorhabditis elegans: an approach to the study of nociception. Proc. Natl Acad. Sci. USA 96, 10477–10482 (1999).
Grancharov, S. G. et al. Bio-functionalization of monodisperse magnetic nanoparticles and their use as biomolecular labels in a magnetic tunnel junction based sensor. J. Phys. Chem. B 109, 13030–13035 (2005).
Wang, Y. Y. et al. Addressing the PEG mucoadhesivity paradox to engineer nanoparticles that ‘slip’ through the human mucus barrier. Angew. Chem. Int. Ed. 47, 9726–9729 (2008)
Wood, W. B. The Nematode Caenorhabditis elegans (Cold Spring Harbor Laboratory, 1988).
Acknowledgements
The authors thank F. Qin for the TRPV1 plasmid and initial assistance, A. Ting for the biotin acceptor peptide-cyan fluorescent protein-transmembrane (AP-CFP-TM) and BirA constructs, and O. Griesbeck for the TN-XL plasmid. J. Pazik is acknowledged for technical assistance, and Y. Hsu, V. Rana, M.J. Ezak and M. Zugravu for valuable discussions.
Author information
Authors and Affiliations
Contributions
A.P. designed the study. H.H. carried out nanoparticle coating and cellular measurements. S.D. and H.Z. were responsible for nanoparticle synthesis and characterization, and D.M.F. for the C. elegans experiments. A.P. and H.H. wrote the manuscript. All authors discussed the results and commented on the manuscript. The work was supported by NSF DMR-0547036, UB IRDF, RF and INSIF.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 1161 kb)
Supplementary information
Supplementary movie 1 (MOV 1751 kb)
Supplementary information
Supplementary movie 2 (MOV 897 kb)
Supplementary information
Supplementary movie 3 (MOV 1261 kb)
Supplementary information
Supplementary movie 4 (MOV 573 kb)
Rights and permissions
About this article
Cite this article
Huang, H., Delikanli, S., Zeng, H. et al. Remote control of ion channels and neurons through magnetic-field heating of nanoparticles. Nature Nanotech 5, 602–606 (2010). https://doi.org/10.1038/nnano.2010.125
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nnano.2010.125
- Springer Nature Limited
This article is cited by
-
Self-rectifying magnetoelectric metamaterials for remote neural stimulation and motor function restoration
Nature Materials (2024)
-
Bioinspired nanotransducers for neuromodulation
Nano Research (2024)
-
Functional nanoparticle-enabled non-genetic neuromodulation
Journal of Nanobiotechnology (2023)
-
Magnetothermal-based non-invasive focused magnetic stimulation for functional recovery in chronic stroke treatment
Scientific Reports (2023)
-
Plasmonics for neuroengineering
Communications Materials (2023)