Abstract
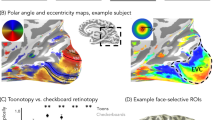
A region in ventral human cortex (fusiform face area, FFA) thought to be important for face perception responds strongly to faces and less strongly to nonface objects. This pattern of response may reflect a uniform face-selective neural population or activity averaged across populations with heterogeneous selectivity. Using high-resolution functional magnetic resonance imaging (MRI), we found that the FFA has a reliable heterogeneous structure: localized subregions within the FFA highly selective to faces are spatially interdigitated with localized subregions highly selective to different object categories. We found a preponderance of face-selective responses in the FFA, but no difference in selectivity to faces compared to nonfaces. Thus, standard fMRI of the FFA reflects averaging of heterogeneous highly selective neural populations of differing sizes, rather than higher selectivity to faces. These results suggest that visual processing in this region is not exclusive to faces. Overall, our approach provides a framework for understanding the fine-scale structure of neural representations in the human brain.








Similar content being viewed by others
Change history
13 December 2006
PDF replaced
References
Kanwisher, N., McDermott, J. & Chun, M.M. The fusiform face area: a module in human extrastriate cortex specialized for face perception. J. Neurosci. 17, 4302–4311 (1997).
George, N. et al. Contrast polarity and face recognition in the human fusiform gyrus. Nat. Neurosci. 2, 574–580 (1999).
Tong, F., Nakayama, K., Vaughan, J.T. & Kanwisher, N. Binocular rivalry and visual awareness in human extrastriate cortex. Neuron 21, 753–759 (1998).
Hasson, U., Hendler, T., Ben Bashat, D. & Malach, R. Vase or face? A neural correlate of shape-selective grouping processes in the human brain. J. Cogn. Neurosci. 13, 744–753 (2001).
Grill-Spector, K., Knouf, N. & Kanwisher, N. The fusiform face area subserves face perception, not generic within-category identification. Nat. Neurosci. 7, 555–562 (2004).
Kanwisher, N. Domain specificity in face perception. Nat. Neurosci. 3, 759–763 (2000).
Schwarzlose, R.F., Baker, C.I. & Kanwisher, N.K. Separate face and body selectivity on the fusiform gyrus. J. Neurosci. 25, 11055–11059 (2005).
Winston, J.S., Henson, R.N., Fine-Goulden, M.R. & Dolan, R.J. fMRI-adaptation reveals dissociable neural representations of identity and expression in face perception. J. Neurophysiol. 92, 1830–1839 (2004).
Spiridon, M. & Kanwisher, N. How distributed is visual category information in human occipito-temporal cortex? An fMRI study. Neuron 35, 1157–1165 (2002).
Edelman, S., Grill-Spector, K., Kushnir, T. & Malach, R. Toward direct visualization of the internal shape representation space by fMRI. Psychobiology 26, 309–321 (1998).
Haxby, J.V. et al. Distributed and overlapping representations of faces and objects in ventral temporal cortex. Science 293, 2425–2430 (2001).
Ishai, A., Ungerleider, L.G., Martin, A., Schouten, J.L. & Haxby, J.V. Distributed representation of objects in the human ventral visual pathway. Proc. Natl. Acad. Sci. USA 96, 9379–9384 (1999).
Avidan, G., Hasson, U., Hendler, T., Zohary, E. & Malach, R. Analysis of the neuronal selectivity underlying low fMRI signals. Curr. Biol. 12, 964–972 (2002).
Desimone, R., Albright, T.D., Gross, C.G. & Bruce, C. Stimulus-selective properties of inferior temporal neurons in the macaque. J. Neurosci. 4, 2051–2062 (1984).
Tsao, D.Y., Freiwald, W.A., Tootell, R.B. & Livingstone, M.S. A cortical region consisting entirely of face-selective cells. Science 311, 670–674 (2006).
Grill-Spector, K., Kourtzi, Z. & Kanwisher, N. The lateral occipital complex and its role in object recognition. Vision Res. 41, 1409–1422 (2001).
Duda, R.O., Hart, P.E. & Stork, D.G. Pattern Classification (John Wiley & Sons, New York, 2001).
Malach, R. et al. Object-related activity revealed by functional magnetic resonance imaging in human occipital cortex. Proc. Natl. Acad. Sci. USA 92, 8135–8139 (1995).
Grill-Spector, K. The neural basis of object perception. Curr. Opin. Neurobiol. 13, 159–166 (2003).
Sayres, R. & Grill-Spector, K. Object-selective cortex exhibits performance-independent repetition suppression. J. Neurophysiol. 95, 995–1007 (2006).
Peelen, M.V. & Downing, P.E. Selectivity for the human body in the fusiform gyrus. J. Neurophysiol. 93, 603–608 (2005).
Saleem, K.S. et al. Magnetic resonance imaging of neuronal connections in the macaque monkey. Neuron 34, 685–700 (2002).
Tolias, A.S. et al. Mapping cortical activity elicited with electrical microstimulation using fMRI in the macaque. Neuron 48, 901–911 (2005).
Tsao, D.Y., Freiwald, W.A., Knutsen, T.A., Mandeville, J.B. & Tootell, R.B. Faces and objects in macaque cerebral cortex. Nat. Neurosci. 6, 989–995 (2003).
Pinsk, M.A., DeSimone, K., Moore, T., Gross, C.G. & Kastner, S. Representations of faces and body parts in macaque temporal cortex: a functional MRI study. Proc. Natl. Acad. Sci. USA 102, 6996–7001 (2005).
Gauthier, I., Tarr, M.J., Anderson, A.W., Skudlarski, P. & Gore, J.C. Activation of the middle fusiform 'face area' increases with expertise in recognizing novel objects. Nat. Neurosci. 2, 568–573 (1999).
Tarr, M.J. & Gauthier, I. FFA: a flexible fusiform area for subordinate-level visual processing automatized by expertise. Nat. Neurosci. 3, 764–769 (2000).
Levy, I., Hasson, U., Avidan, G., Hendler, T. & Malach, R. Center-periphery organization of human object areas. Nat. Neurosci. 4, 533–539 (2001).
Malach, R., Levy, I. & Hasson, U. The topography of high-order human object areas. Trends Cogn. Sci. 6, 176–184 (2002).
Wang, G., Tanaka, K. & Tanifuji, M. Optical imaging of functional organization in the monkey inferotemporal cortex. Science 272, 1665–1668 (1996).
Grill-Spector, K. et al. Differential processing of objects under various viewing conditions in the human lateral occipital complex. Neuron 24, 187–203 (1999).
Tsunoda, K., Yamane, Y., Nishizaki, M. & Tanifuji, M. Complex objects are represented in macaque inferotemporal cortex by the combination of feature columns. Nat. Neurosci. 4, 832–838 (2001).
Gauthier, I. & Nelson, C.A. The development of face expertise. Curr. Opin. Neurobiol. 11, 219–224 (2001).
Golby, A.J., Gabrieli, J.D., Chiao, J.Y. & Eberhardt, J.L. Differential responses in the fusiform region to same-race and other-race faces. Nat. Neurosci. 4, 845–850 (2001).
Fujita, I., Tanaka, K., Ito, M. & Cheng, K. Columns for visual features of objects in monkey inferotemporal cortex. Nature 360, 343–346 (1992).
Olshausen, B.A. & Field, D.J. Sparse coding of sensory inputs. Curr. Opin. Neurobiol. 14, 481–487 (2004).
Albright, T.D., Desimone, R. & Gross, C.G. Columnar organization of directionally selective cells in visual area MT of the macaque. J. Neurophysiol. 51, 16–31 (1984).
DeAngelis, G.C. & Newsome, W.T. Organization of disparity-selective neurons in macaque area MT. J. Neurosci. 19, 1398–1415 (1999).
Coppola, D.M., White, L.E., Fitzpatrick, D. & Purves, D. Unequal representation of cardinal and oblique contours in ferret visual cortex. Proc. Natl. Acad. Sci. USA 95, 2621–2623 (1998).
Chapman, B. & Bonhoeffer, T. Overrepresentation of horizontal and vertical orientation preferences in developing ferret area 17. Proc. Natl. Acad. Sci. USA 95, 2609–2614 (1998).
Acknowledgements
We would like to thank B. Wandell, Y. Spector, N. Kanwisher and C. Baker for fruitful discussions; G. Golarai, D. Remus and D. Yoon for their comments on the manuscript; B. Dougherty for his help in developing software to project ROIs across gray layers; and J. Vinberg for his help in scanning subjects. This research was funded by grants from the National Eye Institute (1 R21EY016199-0) and the Whitehall Foundation (2005-05-111-RES) to K.G.-S. R.S. was supported by the National Eye Institute (5 F31 EY015937).
Author information
Authors and Affiliations
Contributions
K.G.-S. contributed to all aspects of this experiment: design, data collection, development of data analysis methods, code development, and data analyses. R.S. contributed to the design, data collection, code and data analysis development, and data analyses. D.R. developed the surface coil used in these experiments and implemented and developed the HR-fMRI protocols used in these experiments. All authors contributed to understanding the implications of our results and to preparing the manuscript.
Note: Supplementary information is available on the Nature Neuroscience website.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Examples of stimuli used in the experiment. (PDF 339 kb)
Supplementary Fig. 2
Reliability of HR-fMRI. (PDF 396 kb)
Supplementary Fig. 3
Individual subject selectivity maps on inplane slices. (PDF 378 kb)
Supplementary Fig. 4
Individual subject category selective subregions on the inflated cortical surface. (PDF 236 kb)
Supplementary Fig. 5
Decoding distributed FFA activation patterns (PDF 825 kb)
Supplementary Fig. 6
HR-FFA responses to whole and scrambled images versus fixation. (PDF 675 kb)
Supplementary Table 1
Reliability Analyses of HR-fMRI. (PDF 60 kb)
Rights and permissions
About this article
Cite this article
Grill-Spector, K., Sayres, R. & Ress, D. High-resolution imaging reveals highly selective nonface clusters in the fusiform face area. Nat Neurosci 9, 1177–1185 (2006). https://doi.org/10.1038/nn1745
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn1745
- Springer Nature America, Inc.
This article is cited by
-
Bidirectional and parallel relationships in macaque face circuit revealed by fMRI and causal pharmacological inactivation
Nature Communications (2022)
-
Implicit evidence on the dissociation of identity and emotion recognition
Cognitive Processing (2022)
-
Effects of Mild Traumatic Brain Injury on Resting State Brain Network Connectivity in Older Adults
Brain Imaging and Behavior (2022)
-
Higher visual responses in the temporal cortex of mice
Scientific Reports (2018)
-
Sprachdiagnostik bei Patienten mit unilateraler Temporallappenepilepsie
Zeitschrift für Epileptologie (2010)