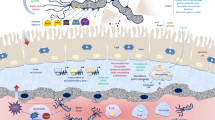
Abstract
Some bacterial pathogens can establish life-long chronic infections in their hosts. Persistence is normally established after an acute infection period involving activation of both the innate and acquired immune systems. Bacteria have evolved specific pathogenic mechanisms and harbor sets of genes that contribute to the establishment of a persistent lifestyle that leads to chronic infection. Persistent bacterial infection may involve occupation of a particular tissue type or organ or modification of the intracellular environment within eukaryotic cells. Bacteria appear to adapt their immediate environment to favor survival and may hijack essential immunoregulatory mechanisms designed to minimize immune pathology or the inappropriate activation of immune effectors.


Similar content being viewed by others
References
Hijjar, A.M., Ernst, R.K., Tsai, J.H., Wilson, C.B. & Miller, S.I. Human Toll-like receptor 4 recognizes host-specific LPS modifications. Nature Immunol. 3, 354–359 (2002).
Guo, L. et al. Regulation of lipid A modifications by Salmonella typhimurium virulence genes phoP-phoQ. Science 276, 250–253 (1997).
Stockinger, B., Barthlott, T. & Kassiotis, G. T cell regulation: a special job or everyone's responsibility? Nature Immunol. 2, 757–758 (2001).
Krinos, C.M., Coyne, M.J., Weinacht, K.G., Tzianabos, A.O., Kasper, D.L. & Comstock, L.E. Extensive surface diversity of a commensal microorganism by multiple DNA inversions. Nature 414, 555–558 (2001).
Virji, M., Makepeace, K., Peak, I.R., Ferguson, D.J. & Moxon, E.R. Pathogenic mechanisms of Neisseria meningitides. Ann. NY Acad. Sci. 797, 273–276 (1996).
Ma, A., Datta, M., Margosian, E., Chen, J. & Horak, I. T cells, but not B cells, are required for bowel inflammation in interleukin 2-deficient mice. J. Exp. Med. 182, 1567–1572 (1995).
Kuhn, R., Lohler, J., Rennick, D., Rajewsky, K. & Muller, W. Interleukin-10-deficient mice develop chronic enterocolitis. Cell 75, 263–274 (1993).
Sellon, R.K. et al. Resident enteric bacteria are necessary for development of spontaneous colitis and immune system activation in interleukin-10-deficient mice. Infect. Immun. 66, 5224–5231 (1998).
Simpson, S.J. et al. T cell-mediated pathology in two models of experimental colitis depends predominantly on the interleukin 12/signal transducer and activator of transcription (Stat)-4 pathway, but is not conditional on interferon γ expression by T cells. J. Exp. Med. 187, 1225–1234 (1998).
Takeda, K. et al. Enhanced Th1 activity and development of chronic enterocolitis in mice devoid of Stat3 in macrophages and neutrophils. Immunity 10, 39–49 (1999).
Strober, W., Nakamura, K. & Kitani, A. The SAMP1/Yit mouse: another step closer to modeling human inflammatory bowel disease. J. Clin. Invest. 107, 667–670 (2001).
Panwala, C.M., Jones, J.C. & Viney, J.L. A novel model of inflammatory bowel disease: mice deficient for the multiple drug resistance gene, mdr1a, spontaneously develop colitis. J. Immunol. 161, 5733–5744 (1998).
Hermiston, M.L. & Gordon, J.I. Inflammatory bowel disease and adenomas in mice expressing a dominant negative N-cadherin. Science 270, 1203–1207 (1995).
Madsen, K.L. et al. Antibiotic therapy attenuates colitis in interleukin 10 gene-deficient mice. Gastroenterology 118, 1094–1105 (2000).
Nagler-Anderson, C. Man the barrier! Strategic defences in the intestinal mucosa. Nature Rev. Immunol. 1, 59–67 (2001).
Strobel, S. & Mowat, A.M. Immune responses to dietary antigens: oral tolerance. Immunol. Today 19, 173–181 (1998).
Macpherson, A.J. et al. IgA production without μ or δ chain expression in developing B cells. Nature Immunol. 2, 625–631 (2001).
Hooper, L.V. & Gordon, J.I. Commensal host-bacterial relationships in the gut. Science 292, 1115–1118 (2001).
Cario, E. & Podolsky, D.K. Differential alteration in intestinal epithelial cell expression of toll-like receptor 3 (TLR3) and TLR4 in inflammatory bowel disease. Infect. Immun. 68, 7010–7017 (2000).
Smith, P.D. et al. Intestinal macrophages lack CD14 and CD89 and consequently are down–regulated for LPS- and IgA-mediated activities. J. Immunol. 167, 2651–2656 (2001).
Blumberg, R.S., Saubermann, L.J. & Strober, W. Animal models of mucosal inflammation and their relation to human inflammatory bowel disease. Curr. Opin. Immunol. 11, 648–656 (1999).
Groux, H. et al. A CD4+ T-cell subset inhibits antigen-specific T-cell responses and prevents colitis. Nature 389, 737–742 (1997).
Newberry, R.D., McDonough, J.S., Stenson, W.F. & Lorenz, R.G. Spontaneous and continuous cyclooxygenase-2-dependent prostaglandin E2 production by stromal cells in the murine small intestine lamina propria: directing the tone of the intestinal immune response. J. Immunol. 166, 4465–4472 (2001).
Maloy, K.J. & Powrie, F. Regulatory T cells in the control of immune pathology. Nature Immunol. 2, 816–822 (2001).
Zabel, B.A. et al. Human G protein-coupled receptor GPR-9-6/CC chemokine receptor 9 is selectively expressed on intestinal homing T lymphocytes, mucosal lymphocytes, and thymocytes and is required for thymus-expressed chemokine-mediated chemotaxis. J. Exp. Med. 190, 1241–1256 (1999).
Rappuoli, R., Pizza, M., Douce, G. & Dougan, G. Structure and mucosal adjuvanticity of cholera and Escherichia coli heat-labile enterotoxins. Immunol. Today 20, 493–500 (1999).
Macdonald, T.T. & Monteleone, G. IL-12 and Th1 immune responses in human Peyer's patches. Trends. Immunol 22, 244–247 (2001).
Wain, J. et al. Molecular typing of multiple-antibiotic-resistant Salmonella enterica serovar Typhi from Vietnam: application to acute and relapse cases of typhoid fever. J. Clin. Microbiol. 37, 2466–2472 (1999).
Wain, J. et al. Quantitation of bacteria in bone marrow from patients with typhoid fever: relationship between counts and clinical features. J. Clin. Microbiol. 39, 1571–1576 (2001).
Parkhill, J. et al. genome sequence of a multiple drug resistant Salmonella enterica serovar Typhi CT18. Nature 413, 848–852 (2001).
McClelland, M. et al. Complete genome sequence of Salmonella enterica serovar Typhimurium LT2. Nature 413, 852–856 (2001).
Townsend, S.M. et al. Salmonella enterica serovar Typhi possesses a unique repertoire of fimbrial gene sequences. Infect. Immun. 69, 2894–2901 (2001).
Kingsley, R.A., Santos, R.L.K.A.M., Adams, L.G. & Baumler, A.J. Salmonella enteritica serotype Typhimurium ShdA is an outer membrane fibronectin-binding protein that is expressed in the intestine. Mol. Microbiol. 43, 895–905 (2002).
Hughes, E.A. & Galan, J.E. Immune response to Salmonella: location, location, location? Immunity 16, 325–328 (2002).
Parkhill, J. et al. Genome sequence of Yersinia pestis, the causative agent of plague. Nature 413, 523–527 (2001).
Cole, S.T. et al. Massive gene decay in the leprosy bacillus. Nature 409, 1007–1011 (2001).
Rescigno, M. et al. Dendritic cells express tight junction proteins and penetrate gut epithelial monolayers to sample bacteria. Nature Immunol. 2, 361–367 (2001).
Vazquez-Torres, A. et al. Extraintestinal dissemination of Salmonella by CD18-expressing phagocytes. Nature 401, 804–808 (1999).
Hindle, Z. et al. Characterisation in volunteers of Salmonella enteritica derivatives harboring defined aroC and SPI-2 type III secretion system (ssaV) mutations. Infect. Immun. 70, 3457–3467 (2002).
Clements, M.O. et al. Polynucleotide phosphorylase is a global regulator of virulence and persistency in Salmonella enterica. Proc. Natl. Acad. Sci. USA 99, 8784–8789 (2002).
O'Callaghan, D., Maskell, D., Liew, F.Y., Easmon, C.S. & Dougan, G. Characterization of aromatic- and purine-dependent Salmonella typhimurium: attention, persistence, and ability to induce protective immunity in BALB/c mice. Infect. Immun. 56, 419–423 (1988).
Warren, J. et al. Increased susceptibility of C1q-deficient mice to Salmonella enterica serovar Typhimurium infection. Infect. Immun. 70, 551–557 (2002).
Mastroeni, P., Simmons, C., Fowler, R., Hormaeche, C.E. & Dougan, G. Igh-6−/− (B-cell-deficient) mice fail to mount solid acquired resistance to oral challenge with virulent Salmonella enterica serovar typhimurium and show impaired Th1 T-cell responses to Salmonella antigens. Infect. Immun. 68, 46–53 (2000).
Salcedo, S.P., Noursadeghi, M., Cohen, J. & Holden, D.W. Intracellular replication of Salmonella typhimurium strains in specific subsets of splenic macrophages in vivo. Cell Microbiol. 3, 587–597 (2001).
Dunstan, S.J. et al. fever and genetic polymorphisms at the natural resistance-associated macrophage protein 1. J. Infect. Dis. 183, 1156–1160 (2001).
Hess, J., Ladel, C., Miko, D. & Kaufmann, S.H. Salmonella typhimurium aroA- infection in gene-targeted immunodeficient mice: major role of CD4+ TCR-αβ cells and IFN-γ in bacterial clearance independent of intracellular location. J. Immunol. 156, 3321–3326 (1996).
Nauciel, C. Role of CD4+ T cells and T-independent mechanisms in acquired resistance to Salmonella typhimurium infection. J. Immunol. 145, 1265–1269 (1990).
O'Brien, A.D. & Metcalf, E.S. Control of early Salmonella typhimurium growth in innately Salmonella-resistant mice does not require functional T lymphocytes. J. Immunol. 129, 1349–1351 (1982).
Mittrucker, H.W., Kohler, A., Mak, T.W. & Kaufmann, S.H. Critical role of CD28 in protective immunity against Salmonella typhimurium. J. Immunol. 163, 6769–6776 (1999).
McSorley, S.J. & Jenkins, M.K. Antibody is required for protection against virulent but not attenuated Salmonella enterica serovar typhimurium. Infect. Immun. 68, 3344–3348 (2000).
Dunstan, S.J. et al. Genes of the class II and class III major histocompatibility complex are associated with typhoid fever in Vietnam. J. Infect. Dis. 183, 261–268 (2001).
Russell, D.G. Mycobacterium tuberculosis: here today, and here tomorrow. Nature Rev. Mol. Cell Biol. 2, 569–577 (2001).
Brennan, P.J. & Nikaido, H. The envelope of mycobacteria. Annu. Rev. Biochem. 64, 29–63 (1995).
Cole, S.T. et al. Deciphering the biology of Mycobacterium tuberculosis from the complete genome sequence. Nature 393, 537–544 (1998).
Barry, C.E. Interpreting cell wall 'virulence factors' of Mycobacterium tuberculosis. Trends. Microbiol. 9, 237–241 (2001).
Bryk, R., Lima, C.D., Erdjument-Bromage, H., Tempst, P. & Nathan, C. Metabolic enzymes of mycobacteria linked to antioxidant defense by a thioredoxin-like protein. Science 295, 1073–1077 (2002).
Hondalus, M.K. et al. Attenuation of and protection induced by a leucine auxotroph of Mycobacterium tuberculosis. Infect. Immun. 68, 2888–2898 (2000).
Via, L.E. et al. Effects of cytokines on mycobacterial phagosome maturation. J. Cell Sci. 111, 897–905 (1998).
Casanova, J.L. & Abel, L. Genetic dissection of immunity to mycobacteria: the human model. Annu. Rev. Immunol. 20, 581–620 (2002).
Flynn, J.L. & Chan, J. Immunology of tuberculosis. Annu. Rev. Immunol. 19, 93–129 (1902).
Brightbill, H.D. et al. Host defense mechanisms triggered by microbial lipoproteins through toll-like receptors. Science 285, 732–736 (1999).
Underhill, D.M., Ozinsky, A., Smith, K.D. & Aderem, A. Toll-like receptor-2 mediates mycobacteria-induced proinflammatory signaling in macrophages. Proc. Natl. Acad. Sci. USA 96, 14459–14463 (1999).
Nau, G.J. et al. Human macrophage activation programs induced by bacterial pathogens. Proc. Natl. Acad. Sci. USA 99, 1503–1508 (2002).
Constant, P. et al. Stimulation of human γδ T cells by nonpeptidic mycobacterial ligands. Science 264, 267–270 (1994).
Ulrichs, T. & Porcelli, S.A. CD1 proteins: targets of T cell recognition in innate and adaptive immunity. Rev. Immunogenet. 2, 416–432 (2000).
Schaible, U.E., Hagens, K., Fischer, K., Collins, H.L. & Kaufmann, S.H. Intersection of group I CD1 molecules and mycobacteria in different intracellular compartments of dendritic cells. J. Immunol. 164, 4843–4852 (2000).
Neyrolles, O. et al. Lipoprotein access to MHC class I presentation during infection of murine macrophages with live mycobacteria. J. Immunol. 166, 447–457 (2001).
Stenger, S. et al. Differential effects of cytolytic T cell subsets on intracellular infection. Science 276, 1684–1687 (1997).
Noss, E.H. et al. Toll-like receptor 2-dependent inhibition of macrophage class II MHC expression and antigen processing by 19-kDa lipoprotein of Mycobacterium tuberculosis. J. Immunol. 167, 910–918 (2001).
Seah, G.T., Scott, G.M. & Rook, G.A. Type 2 cytokine gene activation and its relationship to extent of disease in patients with tuberculosis. J. Infect. Dis. 181, 385–389 (2000).
Ting, L.M., Kim, A.C., Cattamanchi, A. & Ernst, J.D. Mycobacterium tuberculosis inhibits IFN-γ transcriptional responses without inhibiting activation of STAT1. J. Immunol. 163, 3898–3906 (1999).
Rich, A.R. The Pathogenesis of Tuberculosis (Blackwell Scientific Publications, Oxford, 2002).
Opie, E.L. & Aronson, J.D. Tubercle bacilli in latent tuberculosis lesions and in lung tissue without tuberculosis lesions. Arch. Pathol. 4, 1–21 (1927).
Hernandez-Pando, R. et al. Persistence of DNA from Mycobacterium tuberculosis in superficially normal lung tissue during latent infection. Lancet 356, 2133–2138 (2000).
Vandiviere, H.M., Loring, W.E., Melvin, I. & Willis, S. The treated pulmonary lesion and its tubercule bacillus. II The death and the resurrection. Am. J. Med. Sci. 232, 30–37 (1956).
Rees, R.J. & Hart, P.D. Analysis of the host-parasite equilibrium in chronic murine tuberculosis by total and viable bacillary counts. Br. J. Exp. Pathol. 42, 83–88 (1961).
Wallace, J.G. The heat resistance of tubercule in the lungs of infected mice. Am. Rev. Respir. Dis. 83, 866–871 (1961).
McKinney, J.D. et al. Persistence of Mycobacterium tuberculosis in macrophages and mice requires the glyoxylate shunt enzyme isocitrate lyase. Nature 406, 735–738 (2000).
Stewart, G.R. et al. Overexpression of heat-shock proteins reduces survival of Mycobacterium tuberculosis in the chronic phase of infection. Nature Med. 7, 732–737 (2001).
Perez, E. et al. An essential role for phoP in Mycobacterium tuberculosis virulence. Mol. Microbiol. 41, 179–187 (2001).
McCune, R.M., Feldmann, F.M., Lambert, H.P. & McDermott, W. Microbial persistence. I. The capacity of tubercle bacilli to survive sterilization in mouse tissues. J. Exp. Med. 123, 445–468 (1966).
Wayne, L.G. & Sohaskey, C.D. Nonreplicating persistence of Mycobacterium tuberculosis. Annu. Rev. Microbiol. 55, 139–163 (1902).
Sherman, D.R. et al. Regulation of the Mycobacterium tuberculosis hypoxic response gene encoding α-crystallin. Proc. Natl. Acad. Sci. USA 98, 7534–7539 (2001).
Keane, J. et al. Tuberculosis associated with infliximab, a tumor necrosis factor α-neutralizing agent. N. Engl. J. Med. 345, 1098–1104 (2001).
Moreira, A.L. et al. Mycobacterial antigens exacerbate disease manifestations in Mycobacterium tuberculosis-infected mice. Infect. Immun. 70, 2100–2107 (2002).
O'Callaghan, D. et al. A homologue of the Agrobacterium tumefaciens VirB and Bordetella pertussis Ptl type IV secretion systems is essential for intracellular survival of Brucella suis. Mol. Microbiol. 33, 1210–1220 (1999).
Boschiroli, M.L. et al. The Brucella suis virB operon is induced intracellularly in macrophages. Proc. Natl. Acad. Sci. USA 99, 1544–1549 (2002).
Boulton, I.C. & Gray-Owen, S.D. Neisserial binding to CEACAM1 arrests the activation and proliferation of CD4+ T lymphocytes. Nature Immunol. 3, 229–236 (2002).
Byrne, G.I. et al. Chlamydia pneumoniae expresses genes required for DNA replication but not cytokinesis during persistent infection of HEp-2 cells. Infect. Immun. 69, 5423–5429 (2001).
Fischer, S.F., Schwarz, C., Vier, J. & Hacker, G. Characterization of antiapoptotic activities of Chlamydia pneumoniae in human cells. Infect. Immun. 69, 7121–7129 (2001).
Fraser, C.M. et al. Genomic sequence of a Lyme disease spirochaete, Borrelia burgdorferi. Nature 390, 580–586 (1997).
Merz, A.J. & So, M. Interactions of pathogenic neisseriae with epithelial cell membranes. Annu. Rev. Cell Dev. Biol. 16, 423–457 (2000).
Brennan, M.J. & Delogu, G. The PE multigene family: a 'molecular mantra' for mycobacteria. Trends. Microbiol. 10, 246–249 (2002).
Porcella, S.F. & Schwan, T.G. Borrelia burgdorferi and Treponema pallidum: a comparison of functional genomics, environmental adaptations, and pathogenic mechanisms. J. Clin. Invest. 107, 651–656 (2001).
Sansonetti, P. Phagocytosis of bacterial pathogens: implications in the host response. Semin. Immunol. 13, 381–390 (2001).
Galyov, E.E. et al. A secreted effector protein of Salmonella dublin is translocated into eukaryotic cells and mediates inflammation and fluid secretion in infected ileal mucosa. Mol. Microbiol. 25, 903–912 (1997).
Acknowledgements
Supported by The Wellcome Trust (G. D. and D. Y.) and the Medical Research Council (T. H.).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Young, D., Hussell, T. & Dougan, G. Chronic bacterial infections: living with unwanted guests. Nat Immunol 3, 1026–1032 (2002). https://doi.org/10.1038/ni1102-1026
Issue Date:
DOI: https://doi.org/10.1038/ni1102-1026
- Springer Nature America, Inc.
This article is cited by
-
Association of quorum sensing and biofilm formation with Salmonella virulence: story beyond gathering and cross-talk
Archives of Microbiology (2021)
-
Interaction studies on bacterial stringent response protein RelA with uncharged tRNA provide evidence for its prerequisite complex for ribosome binding
Current Genetics (2019)
-
Mechanisms and consequences of diversity-generating immune strategies
Nature Reviews Immunology (2017)
-
High-throughput quantitation of inorganic nanoparticle biodistribution at the single-cell level using mass cytometry
Nature Communications (2017)
-
How type 1 fimbriae help Escherichia coli to evade extracellular antibiotics
Scientific Reports (2016)