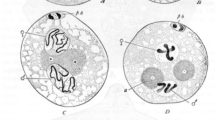
Abstract
Drosophila melanogaster heterochromatin protein 1 (HP1a or HP1)1 is believed to be involved in active transcription, transcriptional gene silencing and the formation of heterochromatin2,3,4,5,6,7. But little is known about the function of HP1 during development. Using a Gal4-induced RNA interference system, we showed that conditional depletion of HP1 in transgenic flies resulted in preferential lethality in male flies. Cytological analysis of mitotic chromosomes showed that HP1 depletion caused sex-biased chromosomal defects, including telomere fusions. The global levels of specific histone modifications, particularly the hallmarks of active chromatin, were preferentially increased in males as well. Expression analysis showed that approximately twice as many genes were specifically regulated by HP1 in males than in females. Furthermore, HP1-regulated genes showed greater enrichment for HP1 binding in males. Taken together, these results indicate that HP1 modulates chromosomal integrity, histone modifications and transcription in a sex-specific manner.




Similar content being viewed by others
References
James, T.C. et al. Distribution patterns of HP1, a heterochromatin-associated nonhistonechromosomal protein of Drosophila. Eur. J. Cell Biol. 50, 170–180 (1989).
Eissenberg, J.C. et al. Mutation in a heterochromatin-specific chromosomal protein is associated with suppression of position-effect variegation in Drosophila melanogaster. Proc. Natl. Acad. Sci. USA 87, 9923–9927 (1990).
Nielsen, S.J. et al. Rb targets histone H3 methylation and HP1 to promoters. Nature 412, 561–565 (2001).
Lachner, M., O'Carroll, D., Rea, S., Mechtler, K. & Jenuwein, T. Methylation of histone H3 lysine 9 creates a binding site for HP1 proteins. Nature 410, 116–120 (2001).
Grewal, S.I. & Elgin, S.C. Heterochromatin: new possibilities for the inheritance of structure. Curr. Opin. Genet. Dev. 12, 178–187 (2002).
Piacentini, L., Fanti, L., Berloco, M., Perrini, B. & Pimpinelli, S. Heterochromatin protein 1 (HP1) is associated with induced gene expression in Drosophila euchromatin. J. Cell Biol. 161, 707–714 (2003).
Cryderman, D.E. et al. Role of Drosophila HP1 in euchromatic gene expression. Dev. Dyn. 232, 767–774 (2005).
Fanti, L., Giovinazzo, G., Berloco, M. & Pimpinelli, S. The heterochromatin protein 1 prevents telomere fusions in Drosophila. Mol. Cell 2, 527–538 (1998).
Giordano, E., Rendina, R., Peluso, I. & Furia, M. RNAi triggered by symmetrically transcribed transgenes in Drosophila melanogaster. Genetics 160, 637–648 (2002).
Raftery, L.A., Sanicola, M., Blackman, R.K. & Gelbart, W.M. The relationship of decapentaplegic and engrailed expression in Drosophila imaginal disks: do these genes mark the anterior-posterior compartment boundary? Development 113, 27–33 (1991).
Hauck, B., Gehring, W.J. & Walldorf, U. Functional analysis of an eye specific enhancer of the eyeless gene in Drosophila. Proc. Natl. Acad. Sci. USA 96, 564–569 (1999).
Neufeld, T.P., de la Cruz, A.F., Johnston, L.A. & Edgar, B.A. Coordination of growth and cell division in the Drosophila wing. Cell 93, 1183–1193 (1998).
Stennicke, H.R., Ryan, C.A. & Salvesen, G.S. Reprieval from execution: the molecular basis of caspase inhibition. Trends Biochem. Sci. 27, 94–101 (2002).
Th'ng, J.P. Histone modifications and apoptosis: cause or consequence? Biochem. Cell Biol. 79, 305–311 (2001).
Jenuwein, T. & Allis, C.D. Translating the histone code. Science 293, 1074–1080 (2001).
Schubeler, D. et al. The histone modification pattern of active genes revealed through genome-wide chromatin analysis of a higher eukaryote. Genes Dev. 18, 1263–1271 (2004).
Schotta, G. et al. Central role of Drosophila SU(VAR)3–9 in histone H3–K9 methylation and heterochromatic gene silencing. EMBO J. 21, 1121–1131 (2002).
Ebert, A. et al. Su(var) genes regulate the balance between euchromatin and heterochromatin in Drosophila. Genes Dev. 18, 2973–2983 (2004).
Akhtar, A. & Becker, P.B. Activation of transcription through histone H4 acetylation by MOF, an acetyltransferase essential for dosage compensation in Drosophila. Mol. Cell 5, 367–375 (2000).
Christensen, T.W. & Tye, B.K. Drosophila MCM10 interacts with members of the prereplication complex and is required for proper chromosome condensation. Mol. Biol. Cell 14, 2206–2215 (2003).
Bergmann, A., Yang, A.Y. & Srivastava, M. Regulators of IAP function: coming to grips with the grim reaper. Curr. Opin. Cell Biol. 15, 717–724 (2003).
Kelley, R.L., Wang, J., Bell, L. & Kuroda, M.I. Sex lethal controls dosage compensation in Drosophila by a non-splicing mechanism. Nature 387, 195–199 (1997).
Orlando, V., Strutt, H. & Paro, R. Analysis of chromatin structure by in vivo formaldehyde cross-linking. Methods 11, 205–214 (1997).
Perrini, B. et al. HP1 controls telomere capping, telomere elongation, and telomere silencing by two different mechanisms in Drosophila. Mol. Cell 15, 467–476 (2004).
De Lucia, F., Ni, J.Q., Vaillant, C. & Sun, F.L. HP1 modulates the transcription of cell-cycle regulators in Drosophila melanogaster. Nucleic Acids Res. 33, 2852–2858 (2005).
Delattre, M., Spierer, A., Jaquet, Y. & Spierer, P. Increased expression of Drosophila Su(var)3–7 triggers Su(var)3–9-dependent heterochromatin formation. J. Cell Sci. 117, 6239–6247 (2004).
Peters, A.H. et al. Loss of the Suv39h histone methyltransferases impairs mammalian heterochromatin and genome stability. Cell 107, 323–337 (2001).
Bongiorni, S., Mazzuoli, M., Masci, S. & Prantera, G. Facultative heterochromatization in parahaploid male mealybugs: involvement of a heterochromatin-associated protein. Development 128, 3809–3817 (2001).
Shaffer, C.D., Wuller, J.M. & Elgin, S.C. Raising large quantities of Drosophila for biochemical experiments. Methods Cell Biol. 44, 99–108 (1994).
Sun, F.L. et al. The fourth chromosome of Drosophila melanogaster: interspersed euchromatic and heterochromatic domains. Proc. Natl. Acad. Sci. USA 97, 5340–5345 (2000).
Acknowledgements
We thank S.C.R. Elgin for providing us with monoclonal (C1A9) and polyclonal (rabbit #192) antibodies against D. melanogaster HP1; G. Reuter for providing antibody against D. melanogaster Su(var)3-9; E. Giordano for sharing unpublished information; G. Rueter for providing antibody against D. melanogaster Su(var)3-9; D. Schübeler and A. Peters for their critical reading of the manuscript; and S. Oakeley and H. Rothnie for their help on nomenclature. This work was supported by the Novartis Research Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Western and immunofluorescent detection of histone modifications in wild type and HP1-depleted larval cells. (PDF 287 kb)
Supplementary Fig. 2
The specificity of HP1 antibodies is determined by western and immunofluorescence. (PDF 178 kb)
Supplementary Table 1
Genes affected both in males and females. (PDF 232 kb)
Supplementary Table 2
Genes specifically affected in males. (PDF 361 kb)
Supplementary Table 3
Genes specifically affected in females. (PDF 231 kb)
Supplementary Table 4
Primers used to perform ChIP and RT-PCR. (PDF 27 kb)
Rights and permissions
About this article
Cite this article
Liu, LP., Ni, JQ., Shi, YD. et al. Sex-specific role of Drosophila melanogaster HP1 in regulating chromatin structure and gene transcription. Nat Genet 37, 1361–1366 (2005). https://doi.org/10.1038/ng1662
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng1662
- Springer Nature America, Inc.
This article is cited by
-
Transcriptional memory of dFOXO activation in youth curtails later-life mortality through chromatin remodeling and Xbp1
Nature Aging (2022)
-
Unravelling HP1 functions: post-transcriptional regulation of stem cell fate
Chromosoma (2021)
-
The large fraction of heterochromatin in Drosophila neurons is bound by both B-type lamin and HP1a
Epigenetics & Chromatin (2018)
-
Histone H1-mediated epigenetic regulation controls germline stem cell self-renewal by modulating H4K16 acetylation
Nature Communications (2015)
-
Sex-biased chromatin and regulatory cross-talk between sex chromosomes, autosomes, and mitochondria
Biology of Sex Differences (2014)