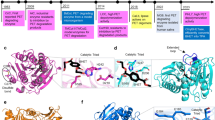
Abstract
Engineering enzymes to degrade anthropogenic compounds efficiently is challenging. We obtained Rhodococcus rhodochrous haloalkane dehalogenase mutants with up to 32-fold higher activity than wild type toward the toxic, recalcitrant anthropogenic compound 1,2,3-trichloropropane (TCP) using a new strategy. We identified key residues in access tunnels connecting the buried active site with bulk solvent by rational design and randomized them by directed evolution. The most active mutant has large aromatic residues at two out of three randomized positions and two positions modified by site-directed mutagenesis. These changes apparently enhance activity with TCP by decreasing accessibility of the active site for water molecules, thereby promoting activated complex formation. Kinetic analyses confirmed that the mutations improved carbon-halogen bond cleavage and shifted the rate-limiting step to the release of products. Engineering access tunnels by combining computer-assisted protein design with directed evolution may be a valuable strategy for refining catalytic properties of enzymes with buried active sites.



Similar content being viewed by others
References
Doyle, S.A., Fung, S.Y. & Koshland, D.E. Jr. Redesigning the substrate specificity of an enzyme: isocitrate dehydrogenase. Biochemistry 39, 14348–14355 (2000).
Whittle, E. & Shanklin, J. Engineering delta 9–16:0-acyl carrier protein (ACP) desaturase specificity based on combinatorial saturation mutagenesis and logical redesign of the castor delta 9–18:0-ACP desaturase. J. Biol. Chem. 276, 21500–21505 (2001).
Wymer, N. et al. Directed evolution of a new catalytic site in 2-keto-3-deoxy-6-phosphogluconate aldolase from Escherichia coli. Structure 9, 1–9 (2001).
Reetz, M.T., Wang, L.W. & Bocola, M. Directed evolution of enantioselective enzymes: iterative cycles of CASTing for probing protein-sequence space. Angew. Chem. Int. Edn Engl. 45, 1236–1241 (2006).
Fox, R.J. et al. Improving catalytic function by ProSAR-driven enzyme evolution. Nat. Biotechnol. 25, 338–344 (2007).
Bartsch, S., Kourist, R. & Bornscheuer, U.T. Complete inversion of enantioselectivity towards acetylated tertiary alcohols by a double mutant of a Bacillus subtilis esterase. Angew. Chem. Int. Edn Engl. 47, 1508–1511 (2008).
Rothlisberger, D. et al. Kemp elimination catalysts by computational enzyme design. Nature 453, 190–195 (2008).
Woycechowsky, K.J., Vamvaca, K. & Hilvert, D. Novel enzymes through design and evolution. Adv. Enzymol. 75, 241–294 (2007).
Morley, K.L. & Kazlauskas, R.J. Improving enzyme properties: when are closer mutations better? Trends Biotechnol. 23, 231–237 (2005).
van der Meer, J.R., Devos, W.M., Harayama, S. & Zehnder, A.J.B. Molecular mechanisms of genetic adaptation to xenobiotic compounds. Microbiol. Rev. 56, 677–694 (1992).
Janssen, D.B., Dinkla, I.J., Poelarends, G.J. & Terpstra, P. Bacterial degradation of xenobiotic compounds: evolution and distribution of novel enzyme activities. Environ. Microbiol. 7, 1868–1882 (2005).
Swanson, P.E. Dehalogenases applied to industrial-scale biocatalysis. Curr. Opin. Biotechnol. 10, 365–369 (1999).
Yujing, M. & Mellouki, A. Rate constants for the reactions of OH with chlorinated propanes. Phys. Chem. Chem. Phys. 3, 2614–2617 (2001).
Bosma, T., Damborsky, J., Stucki, G. & Janssen, D.B. Biodegradation of 1,2,3-trichloropropane through directed evolution and heterologous expression of a haloalkane dehalogenase gene. Appl. Environ. Microbiol. 68, 3582–3587 (2002).
Bosma, T., Kruizinga, E., de Bruin, E.J., Poelarends, G.J. & Janssen, D.B. Utilization of trihalogenated propanes by Agrobacterium radiobacter AD1 through heterologous expression of the haloalkane dehalogenase from Rhodococcus sp. strain m15–3. Appl. Environ. Microbiol. 65, 4575–4581 (1999).
Yokota, T., Omori, T. & Kodama, T. Purification and properties of haloalkane dehalogenase from Corynebacterium sp. strain m15–3. J. Bacteriol. 169, 4049–4054 (1987).
Janssen, D.B. et al. Purification and characterization of a bacterial dehalogenase with activity toward halogenated alkanes, alcohols and ethers. Eur. J. Biochem. 171, 67–72 (1988).
Sallis, P.J., Armfield, S.J., Bull, A.T. & Hardman, D.J. Isolation and characterization of a haloalkane halidohydrolase from Rhodococcus erythropolis Y2. J. Gen. Microbiol. 136, 115–120 (1990).
Kulakova, A.N., Larkin, M.J. & Kulakov, L.A. The plasmid-located haloalkane dehalogenase gene from Rhodococcus rhodochrous NCIMB 13064. Microbiology 143, 109–115 (1997).
Poelarends, G.J., Wilkens, M., Larkin, M.J., van Elsas, J.D. & Janssen, D.B. Degradation of 1,3-dichloropropene by Pseudomonas cichorii 170. Appl. Environ. Microbiol. 64, 2931–2936 (1998).
Newman, J. et al. Haloalkane dehalogenase: structure of a Rhodococcus enzyme. Biochemistry 38, 16105–16114 (1999).
Otyepka, M. & Damborsky, J. Functionally relevant motions of haloalkane dehalogenases occur in the specificity-modulating cap domains. Protein Sci. 11, 1206–1217 (2002).
Verschueren, K.H.G., Seljee, F., Rozeboom, H.J., Kalk, K.H. & Dijkstra, B.W. Crystallographic analysis of the catalytic mechanism of haloalkane dehalogenase. Nature 363, 693–698 (1993).
Gray, K.A. et al. Rapid evolution of reversible denaturation and elevated melting temperature in a microbial haloalkane dehalogenase. Adv. Synth. Catal. 343, 607–617 (2001).
Banas, P., Otyepka, M., Jerabek, P., Petrek, M. & Damborsky, J. Mechanism of enhanced conversion of 1,2,3-trichloropropane by mutant haloalkane dehalogenase revealed by molecular modeling. J. Comput. Aided Mol. Des. 20, 375–383 (2006).
Petrek, M. et al. CAVER: A new tool to explore routes from protein clefts, pockets and cavities. BMC Bioinformatics 7, 316 (2006).
Winn, P.J., Ludemann, S.K., Gauges, R., Lounnas, V. & Wade, R.C. Comparison of the dynamics of substrate access channels in three cytochrome P450s reveals different opening mechanisms and a novel functional role for a buried arginine. Proc. Natl. Acad. Sci. USA 99, 5361–5366 (2002).
Pavelka, A., Chovancova, E. & Damborsky, J. HotSpot Wizard: a web server for identification of hot spots in protein engineering. Nucleic Acids Res. 37 (web server issue): W376–W383 (2009).
Chovancova, E., Kosinski, J., Bujnicki, J.M. & Damborsky, J. Phylogenetic analysis of haloalkane dehalogenases. Proteins Struct. Funct. Bioinf. 67, 305–316 (2007).
Neylon, C. Chemical and biochemical strategies for the randomization of protein encoding DNA sequences: library construction methods for directed evolution. Nucleic Acids Res. 32, 1448–1459 (2004).
Bosma, T., Pikkemaat, M.G., Kingma, J., Dijk, J. & Janssen, D.B. Steady-state and pre-steady-state kinetic analysis of halopropane conversion by a Rhodococcus haloalkane dehalogenase. Biochemistry 42, 8047–8053 (2003).
Schanstra, J.P. & Janssen, D.B. Kinetics of halide release of haloalkane dehalogenase: evidence for a slow conformational change. Biochemistry 35, 5624–5632 (1996).
Prokop, Z. et al. Catalytic mechamism of the haloalkane dehalogenase LinB from Sphingomonas paucimobilis UT26. J. Biol. Chem. 278, 45094–45100 (2003).
Aharoni, A. et al. The “evolvability” of promiscuous protein functions. Nat. Genet. 37, 73–76 (2005).
Stucki, G. & Thuer, M. Experiences of a large-scale application of 1,2-dichloroethane degrading microorganisms for groundwater treatment. Environ. Sci. Technol. 29, 2339–2345 (1995).
Hur, S., Kahn, K. & Bruice, T.C. Comparison of formation of reactive conformers for the SN2 displacements by CH3CO2- in water and by Asp124–CO2- in a haloalkane dehalogenase. Proc. Natl. Acad. Sci. USA 100, 2215–2219 (2003).
Devi-Kesavan, L.S. & Gao, J. Combined QM/MM study of the mechanism and kinetic isotope effect of the nucleophilic substitution reaction in haloalkane dehalogenase. J. Am. Chem. Soc. 125, 1532–1540 (2003).
Olsson, M.H. & Warshel, A. Solute solvent dynamics and energetics in enzyme catalysis: the S(N)2 reaction of dehalogenase as a general benchmark. J. Am. Chem. Soc. 126, 15167–15179 (2004).
Marek, J. et al. Crystal structure of the haloalkane dehalogenase from Sphingomonas paucimobilis UT26. Biochemistry 39, 14082–14086 (2000).
Chaloupkova, R. et al. Modification of activity and specificity of haloalkane dehalogenase from Sphingomonas paucimobilis UT26 by engineering of its entrance tunnel. J. Biol. Chem. 278, 52622–52628 (2003).
Schleinkofer, K., Sudarko, Winn, P.J., Ludemann, S.K. & Wade, R.C. Do mammalian cytochrome P450s show multiple ligand access pathways and ligand channelling? EMBO Rep. 6, 584–589 (2005).
Cojocaru, V., Winn, P.J. & Wade, R.C. The ins and outs of cytochrome P450s. Biochim. Biophys. Acta 1770, 390–401 (2007).
Carmichael, A.B. & Wong, L.L. Protein engineering of Bacillus megaterium CYP102. The oxidation of polycyclic aromatic hydrocarbons. Eur. J. Biochem. 268, 3117–3125 (2001).
Schmitt, J., Brocca, S., Schmid, R.D. & Pleiss, J. Blocking the tunnel: engineering of Candida rugosa lipase mutants with short chain length specificity. Prot. Eng. 15, 595–601 (2002).
Lauble, H. et al. Structure determinants of substrate specificity of hydroxynitrile lyase from Manihot esculenta. Protein Sci. 11, 65–71 (2002).
Fishman, A., Tao, Y., Bentley, W.E. & Wood, T.K. Protein engineering of toluene 4-monooxygenase of Pseudomonas mendocina KR1 for synthesizing 4-nitrocatechol from nitrobenzene. Biotechnol. Bioeng. 87, 779–790 (2004).
Fedorov, R., Vasan, R., Ghosh, D.K. & Schlichting, I. Structures of nitric oxide synthase isoforms complexed with the inhibitor AR-R17477 suggest a rational basis for specificity and inhibitor design. Proc. Natl. Acad. Sci. USA 101, 5892–5897 (2004).
Kotik, M., Stepanek, V., Kyslik, P. & Maresova, H. Cloning of an epoxide hydrolase-encoding gene from Aspergillus niger M200, overexpression in E. coli, and modification of activity and enantioselectivity of the enzyme by protein engineering. J. Biotechnol. 132, 8–15 (2007).
Feingersch, R., Shainsky, J., Wood, T.K. & Fishman, A. Protein engineering of toluene monooxygenases for synthesis of chiral sulfoxides. Appl. Environ. Microbiol. 74, 1555–1566 (2008).
Iwasaki, I., Utsumi, S. & Ozawa, T. New colorimetric determination of chloride using mercuric thiocyanate and ferric ion. Bull. Chem. Soc. Jpn. 25, 226 (1952).
Acknowledgements
We thank V. de Lorenzo (Centro Nacional de Biotecnologia, Spain) and U. Bornscheuer (University Greifswald, Germany) for critical reading of this manuscript. R.C.W. gratefully acknowledges the support of the Klaus Tschira Foundation and M.P. acknowledges a scholarship from the Japan Society for Promotion of Science. We acknowledge financial support from the Ministry of Education of the Czech Republic (LC06010 and MSM0021622412), the Grant Agency of the Czech Republic (201/07/0927 and 203/08/0114), the Grant Agency of the Czech Academy of Sciences (IAA401630901), the North Atlantic Treaty Organization (EST.CLG.980504), and Grants-in-Aid from the Ministry of Education, Culture, Sports, Science, and Technology, Japan and the Ministry of Agriculture, Forestry, and Fisheries, Japan. We acknowledge the Supercomputing Centre Brno for computational resources.
Author information
Authors and Affiliations
Contributions
M.P. performed mutagenesis and activity measurements; M.K. performed molecular modeling and designed mutants; Z.P. performed pre-steady state kinetics measurements; R.C. performed CD spectroscopy measurements and solvent kinetic isotopic effect measurements; P.B. and M.O. designed mutants; R.C.W. contributed the RAMD modeling tool; M.T. and Y.N. contributed molecular biology tools; J.D. interpreted data and designed mutants. All authors contributed to the writing of the paper.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–4, Supplementary Tables 1–3 and Supplementary Methods (PDF 826 kb)
Rights and permissions
About this article
Cite this article
Pavlova, M., Klvana, M., Prokop, Z. et al. Redesigning dehalogenase access tunnels as a strategy for degrading an anthropogenic substrate. Nat Chem Biol 5, 727–733 (2009). https://doi.org/10.1038/nchembio.205
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.205
- Springer Nature America, Inc.
This article is cited by
-
Compact storage of additively weighted Voronoi diagrams
The Visual Computer (2023)
-
Structure-guided engineering of a flavin-containing monooxygenase for the efficient production of indirubin
Bioresources and Bioprocessing (2022)
-
Structure-guided steric hindrance engineering of Bacillus badius phenylalanine dehydrogenase for efficient l-homophenylalanine synthesis
Biotechnology for Biofuels (2021)
-
Tunnel engineering for modulating the substrate preference in cytochrome P450BsβHI
Bioresources and Bioprocessing (2021)
-
Current advances in design and engineering strategies of industrial enzymes
Systems Microbiology and Biomanufacturing (2021)