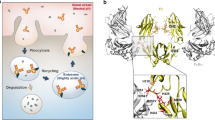
Abstract
The serum protein complement factor H (FH) ensures downregulation of the complement alternative pathway, a branch of innate immunity, upon interaction with specific glycans on host cell surfaces. Using ligand-based NMR, we screened a comprehensive set of sialylated glycans for binding to FH and solved the crystal structure of a ternary complex formed by the two C-terminal domains of FH, a sialylated trisaccharide and the complement C3b thioester–containing domain. Key residues in the sialic acid binding site are conserved from mice to men, and residues linked to atypical hemolytic uremic syndrome cluster within this binding site, suggesting a possible role for sialic acid as a host marker also in other mammals and a critical role in human renal complement homeostasis. Unexpectedly, the FH sialic acid binding site is structurally homologous to the binding sites of two evolutionarily unrelated proteins. The crystal structure also advances our understanding of bacterial immune evasion strategies.




Similar content being viewed by others
References
Ricklin, D., Hajishengallis, G., Yang, K. & Lambris, J.D. Complement: a key system for immune surveillance and homeostasis. Nat. Immunol. 11, 785–797 (2010).
Zipfel, P.F. Complement and immune defense: from innate immunity to human diseases. Immunol. Lett. 126, 1–7 (2009).
Ram, S., Lewis, L.A. & Rice, P.A. Infections of people with complement deficiencies and patients who have undergone splenectomy. Clin. Microbiol. Rev. 23, 740–780 (2010).
Blue, C.E., Spiller, O.B. & Blackbourn, D.J. The relevance of complement to virus biology. Virology 319, 176–184 (2004).
Kim, D.D. & Song, W.-C. Membrane complement regulatory proteins. Clin. Immunol. 118, 127–136 (2006).
Lambris, J.D., Ricklin, D. & Geisbrecht, B.V. Complement evasion by human pathogens. Nat. Rev. Microbiol. 6, 132–142 (2008).
Schmidt, C.Q. et al. The central portion of factor H (modules 10–15) is compact and contains a structurally deviant CCP module. J. Mol. Biol. 395, 105–122 (2010).
Janssen, B.J.C., Christodoulidou, A., McCarthy, A., Lambris, J.D. & Gros, P. Structure of C3b reveals conformational changes that underlie complement activity. Nature 444, 213–216 (2006).
Ferreira, V.P. & Pangburn, M.K. Factor H mediated cell surface protection from complement is critical for the survival of PNH erythrocytes. Blood 110, 2190–2192 (2007).
Renner, B. et al. The complement inhibitors Crry and factor H are critical for preventing autologous complement activation on renal tubular epithelial cells. J. Immunol. 185, 3086–3094 (2010).
Fearon, D.T. Regulation by membrane sialic acid of β1H-dependent decay-dissociation of amplification C3 convertase of the alternative complement pathway. Proc. Natl. Acad. Sci. USA 75, 1971–1975 (1978).
Pangburn, M.K. & Müller-Eberhard, H.J. Complement C3 convertase: cell surface restriction of β1H control and generation of restriction on neuraminidase-treated cells. Proc. Natl. Acad. Sci. USA 75, 2416–2420 (1978).
Ram, S. et al. A novel sialic acid binding site on factor H mediates serum resistance of sialylated Neisseria gonorrhoeae. J. Exp. Med. 187, 743–752 (1998).
Pangburn, M.K., Ferreira, V.P. & Cortes, C. Discrimination between host and pathogens by the complement system. Vaccine 26, I15–I21 (2008).
Clark, S.J. et al. Impaired binding of the age-related macular degeneration-associated complement factor H 402H allotype to Bruch's membrane in human retina. J. Biol. Chem. 285, 30192–30202 (2010).
Prosser, B.E. et al. Structural basis for complement factor H linked age-related macular degeneration. J. Exp. Med. 204, 2277–2283 (2007).
Clark, S.J. et al. Tissue-specific host recognition by complement factor H is mediated by differential activities of its glycosaminoglycan-binding regions. J. Immunol. 190, 2049–2057 (2013).
Paulson, J.C., Macauley, M.S. & Kawasaki, N. Siglecs as sensors of self in innate and adaptive immune responses. Ann. NY Acad. Sci. 1253, 37–48 (2012).
Crespo, H.J., Lau, J.T.Y. & Videira, P.A. Dendritic cells: a spot on sialic acid. Front. Immunol. 4, 491 (2013).
Meri, S. & Pangburn, M.K. Discrimination between activators and nonactivators of the alternative pathway of complement: regulation via a sialic acid/polyanion binding site on factor H. Proc. Natl. Acad. Sci. USA 87, 3982–3986 (1990).
Meri, S. & Pangburn, M.K. Regulation of alternative pathway complement activation by glycosaminoglycans: specificity of the polyanion binding site on factor H. Biochem. Biophys. Res. Commun. 198, 52–59 (1994).
Morgan, H.P. et al. Structural basis for engagement by complement factor H of C3b on a self surface. Nat. Struct. Mol. Biol. 18, 463–470 (2011).
Kajander, T. et al. Dual interaction of factor H with C3d and glycosaminoglycans in host-nonhost discrimination by complement. Proc. Natl. Acad. Sci. USA 108, 2897–2902 (2011).
Varki, A. et al. Essentials of Glycobiology (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, 2009).
Mayer, M. & Meyer, B. Group epitope mapping by saturation transfer difference NMR to identify segments of a ligand in direct contact with a protein receptor. J. Am. Chem. Soc. 123, 6108–6117 (2001).
Williamson, M.P. Applications of the NOE in molecular biology. Annu. Rep. NMR. Spectrosc. 65, 77–109 (2009).
Claasen, B., Axmann, M., Meinecke, R. & Meyer, B. Direct observation of ligand binding to membrane proteins in living cells by a saturation transfer double difference (STDD) NMR spectroscopy method shows a significantly higher affinity of integrin α(IIb)β3 in native platelets than in liposomes. J. Am. Chem. Soc. 127, 916–919 (2005).
Ferreira, V.P., Herbert, A.P., Hocking, H.G., Barlow, P.N. & Pangburn, M.K. Critical role of the C-terminal domains of factor H in regulating complement activation at cell surfaces. J. Immunol. 177, 6308–6316 (2006).
Kavanagh, D., Goodship, T.H. & Richards, A. Atypical hemolytic uremic syndrome. Semin. Nephrol. 33, 508–530 (2013).
Herbert, A.P., Uhrín, D., Lyon, M., Pangburn, M.K. & Barlow, P.N. Disease-associated sequence variations congregate in a polyanion recognition patch on human factor H revealed in three-dimensional structure. J. Biol. Chem. 281, 16512–16520 (2006).
Spitzer, D., Mitchell, L.M., Atkinson, J.P. & Hourcade, D.E. Properdin can initiate complement activation by binding specific target surfaces and providing a platform for de novo convertase assembly. J. Immunol. 179, 2600–2608 (2007).
Kemper, C., Atkinson, J.P. & Hourcade, D.E. Properdin: emerging roles of a pattern-recognition molecule. Annu. Rev. Immunol. 28, 131–155 (2010).
Varki, A. & Kornfeld, S. An autosomal dominant gene regulates the extent of 9-O-acetylation of murine erythrocyte sialic acids. A probable explanation for the variation in capacity to activate the human alternate complement pathway. J. Exp. Med. 152, 532–544 (1980).
Michalek, M.T., Mold, C. & Bremer, E.G. Inhibition of the alternative pathway of human complement by structural analogues of sialic acid. J. Immunol. 140, 1588–1594 (1988).
Okada, N., Yasuda, T. & Okada, H. Restriction of alternative complement pathway activation by sialosylglycolipids. Nature 299, 261–263 (1982).
Ferreira, V.P. et al. The binding of factor H to a complex of physiological polyanions and C3b on cells is impaired in atypical hemolytic uremic syndrome. J. Immunol. 182, 7009–7018 (2009).
Herbert, A.P. et al. Structural and functional characterization of the product of disease-related factor H gene conversion. Biochemistry 51, 1874–1884 (2012).
Maga, T.K., Nishimura, C.J., Weaver, A.E., Frees, K.L. & Smith, R.J.H. Mutations in alternative pathway complement proteins in American patients with atypical hemolytic uremic syndrome. Hum. Mutat. 31, E1445–E1460 (2010).
Schmidt, C.Q. et al. Rational engineering of a minimized immune inhibitor with unique triple-targeting properties. J. Immunol. 190, 5712–5721 (2013).
Galeano, B. et al. Mutation in the key enzyme of sialic acid biosynthesis causes severe glomerular proteinuria and is rescued by N-acetylmannosamine. J. Clin. Invest. 117, 1585–1594 (2007).
Mulloy, B., Forster, M.J., Jones, C. & Davies, D.B. N.m.r. and molecular-modelling studies of the solution conformation of heparin. Biochem. J. 293, 849–858 (1993).
Klein, P.J. et al. Thomsen Friedenreich antigen in hæmolytic-uræmic syndrome. Lancet 2, 1024–1025 (1977).
Copelovitch, L. & Kaplan, B.S. Streptococcus pneumoniae–associated hemolytic uremic syndrome. Pediatr. Nephrol. 23, 1951–1956 (2008).
Bhattacharjee, A. et al. Structural basis for complement evasion by Lyme disease pathogen Borrelia burgdorferi. J. Biol. Chem. 288, 18685–18695 (2013).
Pickering, M.C. et al. Spontaneous hemolytic uremic syndrome triggered by complement factor H lacking surface recognition domains. J. Exp. Med. 204, 1249–1256 (2007).
Skerka, C., Chen, Q., Fremeaux-Bacchi, V. & Roumenina, L.T. Complement factor H related proteins (CFHRs). Mol. Immunol. 56, 170–180 (2013).
Zhuravleva, M.A., Trandem, K. & Sun, P.D. Structural implications of Siglec-5–mediated sialoglycan recognition. J. Mol. Biol. 375, 437–447 (2008).
Rademacher, C. et al. A Siglec-like sialic-acid–binding motif revealed in an adenovirus capsid protein. Glycobiology 22, 1086–1091 (2012).
Seiradake, E. et al. The cell adhesion molecule 'CAR' and sialic acid on human erythrocytes influence adenovirus in vivo biodistribution. PLoS Pathog. 5, e1000277 (2009).
Nicholson, J.K., Foxall, P.J., Spraul, M., Farrant, R.D. & Lindon, J.C. 750 MHz 1H and 1H–13C NMR spectroscopy of human blood plasma. Anal. Chem. 67, 793–811 (1995).
Kabsch, W. Integration, scaling, space-group assignment and post-refinement. Acta Crystallogr. D Biol. Crystallogr. 66, 133–144 (2010).
Murshudov, G.N., Vagin, A.A. & Dodson, E.J. Refinement of macromolecular structures by the maximum-likelihood method. Acta Crystallogr. D Biol. Crystallogr. 53, 240–255 (1997).
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D Biol. Crystallogr. 60, 2126–2132 (2004).
Adams, P.D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D Biol. Crystallogr. 66, 213–221 (2010).
Davis, I.W. et al. MolProbity: all-atom contacts and structure validation for proteins and nucleic acids. Nucleic Acids Res. 35, W375–W383 (2007).
Frank, M., Lütteke, T. & von der Lieth, C.-W. GlycoMapsDB: a database of the accessible conformational space of glycosidic linkages. Nucleic Acids Res. 35, 287–290 (2007).
Papadopoulos, J.S. & Agarwala, R. COBALT: constraint-based alignment tool for multiple protein sequences. Bioinformatics 23, 1073–1079 (2007).
Acknowledgements
The authors thank R. Sprangers (Max Planck Institute for Development Biology, Tübingen, Germany), for assistance with NMR spectra collection, the SLS beamline staff (Paul Scherrer Institute, Villigen, Switzerland) for assistance with crystallographic data collection and H. Hengel (University Hospital Tübingen) for performing venipuncture and assistance with serum preparation. Funding was provided by the University of Tübingen Program for the Promotion of Junior Researchers and the University Hospital Tübingen Fortüne program (2186-0-0 to B.S.B.). D.K. is a Wellcome Trust intermediate clinical fellow.
Author information
Authors and Affiliations
Contributions
B.S.B. designed, conducted and analyzed experiments. J.P.H. advised crystallization, and D.U. advised NMR experiments. J.P.H., A.P.H., D.K. and D.U. provided proteins. D.U. provided glycans. T.S. supervised the project and provided vital reagents. All authors discussed findings. B.S.B. and T.S. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Results, Supplementary Figures 1–6 and Supplementary Tables 1 and 2. (PDF 2385 kb)
Rights and permissions
About this article
Cite this article
Blaum, B., Hannan, J., Herbert, A. et al. Structural basis for sialic acid–mediated self-recognition by complement factor H. Nat Chem Biol 11, 77–82 (2015). https://doi.org/10.1038/nchembio.1696
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.1696
- Springer Nature America, Inc.
This article is cited by
-
The monosialoganglioside GM1a protects against complement attack
Cell Death Discovery (2023)
-
Helicobacter pylori initiates successful gastric colonization by utilizing L-lactate to promote complement resistance
Nature Communications (2023)
-
Low molecular weight polysialic acid binds to properdin and reduces the activity of the alternative complement pathway
Scientific Reports (2022)
-
Binding-driven reactivity attenuation enables NMR identification of selective drug candidates for nucleic acid targets
Communications Chemistry (2022)
-
Dendrimer end-terminal motif-dependent evasion of human complement and complement activation through IgM hitchhiking
Nature Communications (2021)