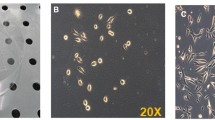
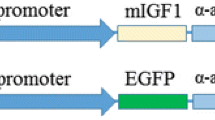
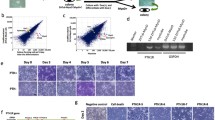
Abstract
Ectopic expression of a truncated growth hormone–releasing hormone (GHRH) from muscle tissues by a myogenic plasmid DNA vector directed by the skeletal α-actin promoter, pSK-GHRH, results in growth hormone (GH) secretion. Skeletal muscle-secreted GHRH is biologically active. The application of conditioned media harvested from pSK-GHRH transfected muscle cells to cultured pig primary anterior pituitary cells elicits GH release. A single intramuscular injection of 100 μg pSK-GHRH DNA elevates serum GH levels threefold to fourfold for up to 2 weeks, enhancing liver IGF-1 gene expression and increasing body weight approximately 10%.
Similar content being viewed by others
References
Scanlon, M.F., Issa, B.G., and Dieguez, C. 1996. Regulation of growth hormone secretion. Horm. Res. 46: 149–154.
Enright, W.J., Prendiville, D.J., Spicer, L.J., Strieker, P.R., Moloney, A.P., Mowles, T.F., and Campbell, R.M. 1993. Effects of growth hormone-releasing factor and(or) thyrotropin-releasing hormone on growth, feed efficiency, carcass characteristics, and blood hormones and metabolites in beef heifers. J. Anim. Sci. 71: 2395–2405.
Enright, W.J., Chapin, L.T., Moseley, W.M., Zinn, S.A., and Tucker, H.A. 1986. Growth hormone-releasing factor stimulates milk production and sustains growth hormone release in Holstein cows. J. Dairy Sci. 69: 344–351.
LeRoith, D., Yanowski, J., Kaldjian, E.P., Jaffe, E.S., LeRoith, T., Purdue, K., et al. 1996. The effects of growth hormone and insulin-like growth factor I on the immune system of aged female monkeys. Endocrinology 137: 1071–1079.
Kotzmann, H., Koller, M., Czernin, S., Clodi, M., Svoboda, T., Riedl, M., et al. 1994. Effect of elevated growth hormone concentrations on the phenotype and functions of human lymphocytes and natural killer cells. Neuroendocrinology 60: 618–625.
Iranmanesh, A., Lizarralde, G., and Veldhuis, J.D. 1991. Age and relative adiposity are specific negative determinants of the frequency and amplitude of growth hormone (GH) secretory bursts and the half-life of endogenous GH in healthy men. J. clin. Endocrinol. Metab. 73: 1081–1038.
D'Costa, A.P., Ingram, R.L., Lenham, J.E., and Sonntag, W.E. 1993. The regulation and mechanisms of action of growth hormone and insulin-like growth factor 1 during normal ageing. J. Reprod. Fertil. Suppl. 46: 87–98.
Corpas, E., Harman, S.M., and Blackman, M.R. 1993. Human growth hormone and human aging. Endocr. Rev. 14: 20–39.
Marcus, R., Butterfield, G., Holloway, L., Gilliland, L., Baylink, D.J., Hintz, R.L., and Sherman, B.M. 1990. Effects of short term administration of recombinant human growth hormone to elderly people. J. Clin. Endocrinol. Metab. 70: 519–527.
Salomon, F., Cuneo, R.C., Hesp, R., and Sonksen, P.H. 1989. The effects of treatment with recombinant human growth hormone on body composition and metabolism in adults with growth hormone deficiency. N. Engl. J. Med. 321: 1797–803.
Corpas, E., Harman, S.M., Pineyro, M.A., Roberson, R., and Blackman, M.R. 1993. Continuous subcutaneous infusions of growth hormone (GH) releasing hormone 1-44 for 14 days increase GH and insulin-like growth factor-l levels in old men. J. Clin. Endocrinol. Metab. 76: 134–138.
Mayo, K.E., Cerelli, G.M., Lebo, R.V., Bruce, B.D., Rosenfeld, M.G., and Evans, R.M. 1985. Gene encoding human growth hormone-releasing factor precursor: structure, sequence, and chromosomal assignment. Proc. Natl. Acad. Sci. USA 82: 63–67.
Mayo, K.E., Vale, W., Rivier, J., Rosenfeld, M.G., and Evans, R.M. 1983. Expression-cloning and sequence of a cDNA encoding human growth hormone-releasing factor. Nature 306: 86–88.
Riddell, D.C., Mallonee, R., Phillips, J.A., Parks, J.S., Sexton, L.A., and Hamerton, J.L. 1985. Chromosomal assignment of human sequences encoding arginine vasopressin-neurophysin II and growth hormone releasing factor. Som. Cell Mol. Genet. 11: 189–195.
Esch, F.S., Bohlen, P., Ling, N.C., Brazeau, P.E., Wehrenberg, W.B., Thorner, M.O., et al. 1982. Characterization of a 40 residue peptide from a human pancreatic tumor with growth hormone releasing activity. Biochem. Biophys. Res. Comm. 109: 152–158.
Thorner, M.O., Frohman, L.A., Leong, D.A., Thominet, J., Downs, T., Hellmann, P., et al. 1984. Extrahypothalamic growth-hormone-releasing factor (GRF) secretion is a rare cause of acromegaly: plasma GRF levels in 177 acromegalic patients. J. Clin. Endocrinol. Metab. 59: 846–849.
Wolff, J.A., Ludtke, J.J., Acsadi, G., Williams, P., and Jani, A. 1992. Long-term persistence of plasmid DNA and foreign gene expression in mouse muscle. Hum. Mol. Genet. 1: 363–369.
Wolff, J.A., Malone, R.W., Williams, P., Chong, W., Acsadi, G., Jani, A., and Feigner, P.L. 1990. Direct gene transfer into mouse muscle in vivo. Science 247: 1465–1468.
Davis, H.L., Whalen, R.G., and Demeneix, B.A. 1993. Direct gene transfer into skeletal muscle in vivo: factors affecting efficiency of transfer and stability of expression. Hum. Gene Ther. 4: 151–159.
Wells, D.J. 1993. Improved gene transfer by direct plasmid injection associated with regeneration in mouse skeletal muscle. FEBS Lett. 332: 179–182.
Bergsma, D.J., Grichnik, J.M., Gossett, L.M., and Schwartz, R.J. 1986. Delimitation and characterization of cis-acting DNA sequences required for the regulated expression and transcriptional control of the chicken skeletal alpha-act in gene. Mol. Cell. Biol. 6: 2462–2475.
Chow, K.L., and Schwartz, R.J. 1990. A combination of closely associated positive and negative cis-acting promoter elements regulates transcription of the skeletal alpha-actin gene. Mol. Ceil. Biol. 10: 528–538.
Lee, T.C., Zhang, Y., and Schwartz, R.J. 1994. Bifunctional transcriptional properties of YY1 in regulating muscle actin and c-myc gene expression during myogenesis. Oncogene 9: 1047–1052.
Ling, N., Baird, A., Wehrenberg, W.B., Ueno, N., Munegumi, T., and Brazeau, P. 1984. Synthesis and in vitro bioactivity of C-terminal deleted analogs of human growth hormone-releasing factor. Biochem. Biophys. Res. Comm. 123: 854–861.
Bertherat, J., Bluet-Pajot, M.T., and Epelbaum, J. 1995. Neuroendocrine regulation of growth hormone. Eur. J. Endocrinol. 132: 12–24.
Low, L.C. 1993. The therapeutic use of growth-hormone-releasing hormone. J. Pediatr. Endocrinol. 6: 15–20.
Yao, S.N., Smith, K.J., and Kurachi, K. 1994. Primary myoblast-mediated gene transfer: persistent expression of human factor IX in mice. Gene Therapy 1: 99–107.
Tripathy, S.K., Svensson, E.C., Black, H.B., Goldwasser, E., Margalith, M., Hobart, P.M., and Leiden, J.M. 1996. Long-term expression of erythropoietin in the systemic circulation of mice after intramuscular injection of a plasmid DNA vector. Proc. Natl. Acad. Sci. USA 93: 10876–10880.
Tripathy, S.K., Black, H.B., Goldwasser, E., and Leiden, J.M. 1996. Immune responses to transgene-encoded proteins limit the stability of gene expression after injection of replication-defective adenovirus vectors. Nature Medicine 2: 545–550.
Levy, M.Y., Barron, L.G., Meyer, K.B., and Szoka, F.C. Jr. 1996. Characterization of plasmid DNA transfer into mouse skeletal muscle: evaluation of uptake mechanism, expression and secretion of gene products into blood. Gene Therapy 3: 201–211.
Cao, Y., Wagner, J.K., Hindmarsh, P.C., Eble, A., and Mullis, P.E. 1995. Isolated growth hormone deficiency: testing the little mouse hypothesis in man and exclusion of mutations within the extracellular domain of the growth hormone-releasing hormone receptor. Pediatr. Res. 38: 962–926.
Cogan, J.D., Phillips, J.A. 3d, Sakati, N., Frisch, H., Schober, E., and Milner, R.D. 1993. Heterogeneous growth hormone (GH) gene mutations in familial GH deficiency. J. Clin. Endocrinol. Metab. 76: 1224–1228.
Vnencak-Jones, C.L., Phillips, J.A. 3d, Chen, E.Y., and Seeburg, P.H. 1988. Molecular basis of human growth hormone gene deletions. Proc. Natl. Acad. Sci. USA 85: 5615–5619.
Amselem, S., Duquesnoy, P., Duriez, B., Dastot, F., Sobrier, M.L., Valleix, S., and Goossens, M. 1993. Spectrum of growth hormone receptor mutations and associated haplotypes in Laron syndrome. Hum. Mol. Genet. 2: 355–359.
Amselem, S., Duquesnoy, P., Sobrier, M.L., and Goossens, M. 1991. Molecular defects in the growth hormone receptor. A. Paediatr. Scandinavica Suppl. 377: 81–86.
Meacham, L.R., Brown, M.R., Murphy, T.L., Keret, R., Silbergeld, A., Laron, Z., and Parks, J.S. 1993. Characterization of a noncontiguous gene deletion of the growth hormone receptor in Laron's syndrome. J. Clin. Endocrinol. Metab. 77: 1379–1383.
Parks, J.S., Abdul-Latif, H., Kinoshita, E., Meacham, L.R., Pfaffle, R.W., and Brown, M.R. 1993. Genetics of growth hormone gene expression. Hormone Res. 40: 54–61.
Parks, J.S., Pfaffle, R.W., Brown, M.R., Abdul-Latif, H., and Meacham, L.R. 1995. Growth hormone deficiency, pp. 473–90 in Molecular endocrinology: basic concepts and clinical correlations. Weintraub, B.D. (ed.) Raven Press, New York.
Wang, Y., DeMayo, F.J., Tsai, S.Y., and O'Malley, B.W. 1997. Ligand-inducible and liver-specific target gene expression in transgenic mice. Nature Biotechnology 15: 239–243.
Chow, K.L., Hogan, M.E., and Schwartz, R.J. 1991. Phased cis-acting promoter elements interact at short distances to direct avian skeletal alpha-actin gene transcription. Proc. Natl. Acad. Sci. USA 88: 1301–1305.
French, B.A., Bergsma, D.J., and Schwartz, R.J. 1990. Analysis of a CR1 (chicken repeat) sequence flanking the 5′ end of the gene encoding alpha-skeletal actin. Gene 88: 173–80.
Doumit, M.E. and Merkel, R.A. 1992. Conditions for isolation and culture of porcine myogenic satellite cells. Tissue & Cell 24: 253–262.
Tanner, J.W., Davis, S.K., McArthur, N.H., French, J.T., and Welsh, T.H., Jr. 1990. Modulation of growth hormone (GH) secretion and GH mRNA levels by GH-releasing factor, somatostatin and secretagogues in cultured bovine adenohy-pophysial cells. J. Endocrinol. 125: 109–115.
Barb, C.R., Estienne, M.J., Kraeling, R.R., Marple, D.N., Rampacek, G.B., Rahe, C.H., and Sartin, J.L. 1991. Endocrine changes in sows exposed to elevated ambient temperature during lactation. Domest. Anim. Endocrinol. 8: 117–127.
Danko, I., Fritz, J.D., Jiao, S., Hogan, K., Latendresse, J.S., and Wolff, J.A. 1994. Pharmacological enhancement of in vivo foreign gene expression in muscle. Gene Therapy 1: 114–121.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Draghia-Akli, R., Li, X. & Schwartz, R. Enhanced growth by ectopic expression of growth hormone releasing hormone using an injectable myogenic vector. Nat Biotechnol 15, 1285–1289 (1997). https://doi.org/10.1038/nbt1197-1285
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nbt1197-1285
- Springer Nature America, Inc.
This article is cited by
-
Double-blinded, Placebo-controlled Plasmid GHRH Trial for Cancer-associated Anemia in Dogs
Molecular Therapy (2008)
-
Synthetic muscle promoters: activities exceeding naturally occurring regulatory sequences
Nature Biotechnology (1999)
-
Electropermeabilization of skeletal muscle enhances gene transfer in vivo
Gene Therapy (1999)
-
Myogenic expression of an injectable protease-resistant growth hormone–releasing hormone augments long-term growth in pigs
Nature Biotechnology (1999)
-
Growth hormone technology develops new twist
Nature Biotechnology (1997)