Abstract
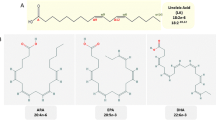
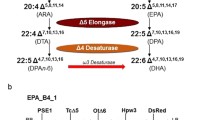
Very long chain polyunsaturated fatty acids (VLCPUFAs) such as arachidonic acid (AA), eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) are valuable commodities that provide important human health benefits1,2,3,4,5. We report the transgenic production of significant amounts of AA and EPA in Brassica juncea seeds via a stepwise metabolic engineering strategy. Using a series of transformations with increasing numbers of transgenes, we demonstrate the incremental production of VLCPUFAs, achieving AA levels of up to 25% and EPA levels of up to 15% of total seed fatty acids. Both fatty acids were almost exclusively found in triacylglycerols, with AA located preferentially at sn-2 and sn-3 positions and EPA distributed almost equally at all three positions. Moreover, we reconstituted the DHA biosynthetic pathway in plant seeds, demonstrating the practical feasibility of large-scale production of this important ω-3 fatty acid in oilseed crops.



Similar content being viewed by others
References
Funk, C.D. Prostaglandins and leukotrienes: advances in eicosanoid biology. Science 294, 1871–1875 (2001).
Hong, S. et al. Docosatrienes and 17S-resolvins generated from docosahexaenoic acid in murine brain,human blood, and glial cells. Autacoids in anti-inflammation. J. Biol. Chem. 278, 14677–14687 (2003).
Crawford, M.A. et al. Are deficits of arachidonic and docosahexaenoic acids responsible for the neural and vascular complications of preterm babies? Am. J. Clin. Nutr. 66, 1032S–1041S (1997).
Benatti, P., Peluso, G., Nicolai, R. & Calvani, M. Polyunsaturated fatty acids: biochemical, nutritional and epigenetic properties. J. Am. Coll. Nutr. 23, 281–302 (2004).
Spector, A. Essentiality of fatty acids. Lipids 34, S1–S3 (1999).
Slater, S. et al. Metabolic engineering of Arabidopsis and Brassica for poly(3-hydroxybutyrate-co-3-hydroxyvalerate) copolymer production. Nat. Biotechnol. 17, 1011–1016 (1999).
Ye, X. et al. Engineering the provitamin A (beta-carotene) biosynthetic pathway into (carotenoid-free) rice endosperm. Science 287, 303–305 (2000).
Thelen, J.J. & Ohlrogge, J.B. Metabolic engineering of fatty acid biosynthesis in plants. Metab. Eng. 4, 12–21 (2002).
Qi, B. et al. Production of very long chain polyunsaturated omega-3 and omega-6 fatty acids in plants. Nat. Biotechnol. 22, 739–745 (2004).
Abbadi, A. et al. Biosynthesis of very-long-chain polyunsaturated fatty acids in transgenic oilseeds: constraints on their accumulation. Plant Cell 16, 2734–2748 (2004).
Qiu, X. Biosynthesis of docosahexaenoic acid (DHA, 22:6–4,7,10,13,16,19): two distinct pathways. Prostaglandins Leukot. Essent. Fatty Acids 68, 181–186 (2003).
Hong, H. et al. High-level production of gamma-linolenic acid in Brassica juncea using a delta 6 desaturase from Pythium irregulare . Plant Physiol. 129, 354–362 (2002).
Qiu, X., Hong, H. & MacKenzie, S.L. Identification of a delta 4 fatty acid desaturase from Thraustochytrium sp. involved in the biosynthesis of docosahexaenoic acid by heterologous expression in Saccharomyces cerevisiae and Brassica juncea . J. Biol. Chem. 276, 31561–31566 (2001).
Zank, T.K. et al. Cloning and functional characterisation of an enzyme involved in the elongation of delta 6 polyunsaturated fatty acids from the moss Physcomitrella patens . Plant J. 31, 255–268 (2002).
Qiu, X., Reed, D.W., Hong, H., MacKenzie, S.L. & Covello, P.S. Identification and analysis of a gene from Calendula officinalis encoding a fatty acid conjugase. Plant Physiol. 125, 847–855 (2001).
Meyer, A. et al. Novel fatty acid elongases and their use for the reconstitution of docosahexaenoic acid biosynthesis. J. Lipid Res. 45, 1899–1909 (2004).
Leonard, A.E., Pereira, S.L., Sprecher, H. & Huang, Y.S. Elongation of long-chain fatty acids. Prog. Lipid Res. 43, 36–54 (2004).
Han, J. et al. Functional characterization of beta-ketoacyl-CoA synthase genes from Brassica napus L. Plant Mol. Biol. 46, 229–239 (2001).
Domergue, F. et al. Acyl carriers used as substrates by the desaturases and elongases involved in very long-chain polyunsaturated fatty acids biosynthesis reconstituted in yeast. J. Biol. Chem. 278, 35115–35126 (2003).
Wiberg, E., Banas, A. & Stymne, S. Fatty acid distribution and lipid metabolism in developing seeds of laurate-producing rape (Brassica napus L.). Planta 203, 341–348 (1997).
Wiberg, E., Edwards, P., Byrne, J., Stymne, S. & Dehesh, K. The distribution of caprylate, caprate and laurate in lipids from developing and mature seeds of transgenic Brassica napus L. Planta 212, 33–40 (2000).
Graham, I.A., Cirpus, P., Rein, D. & Napier, J.A. The use of very long chain polyunsaturated fatty acids to ameliorate metabolic syndrome: transgenic plants as an alternative sustainable source to fish oils. Nutr. Bul. 29, 228–233 (2004).
Napier, J.A., Beaudoin, F., Michaelson, L.V. & Sayanova, O. The production of long chain polyunsaturated fatty acids in transgenic plants by reverse-engineering. Biochimie 86, 785–792 (2004).
Ericson, M.L. et al. Structure of the rapeseed 1.7 S storage protein, napin, and its precursor. J. Biol. Chem. 261, 14576–14581 (1986).
Radke, S., Turner, J. & Faccioti, D. Transformation and regeneration of Brassica rapa using Agrobacterium tumefaciens . Plant Cell Rep. 11, 499–505 (1992).
Lynch, D. & Steponkus, P. Plasma membrane lipid alterations associated with cold acclimation of winter rye seedlings (Secale cereale L. cv. Puma). Plant Physiol. 83, 761–767 (1987).
Myher, J.J. & Kuksis, A. Stereospecific analysis of triacylglycerols via racemic phosphatidylcholines and phospholipase C. Can. J. Biochem. 57, 117–124 (1979).
Acknowledgements
The authors thank Derek Potts for providing B. juncea germplasm, and Darwin Reed and Mike Giblin for assistance with GC and GC/MS analysis. We also thank Jonathan Page for comments on an earlier version of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
This research was funded in part by BASF BmbH, Ludwigshafen, Germany.
Rights and permissions
About this article
Cite this article
Wu, G., Truksa, M., Datla, N. et al. Stepwise engineering to produce high yields of very long-chain polyunsaturated fatty acids in plants. Nat Biotechnol 23, 1013–1017 (2005). https://doi.org/10.1038/nbt1107
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt1107
- Springer Nature America, Inc.
This article is cited by
-
Identification of genes associated with fatty acid biosynthesis based on 214 safflower core germplasm
BMC Genomics (2023)
-
Zinc oxide nanoparticles alleviate cadmium stress by modulating plant metabolism and decreasing cadmium accumulation in Perilla frutescents
Plant Growth Regulation (2023)
-
Evaluation of long-chain omega-3 canola oil on Atlantic salmon growth, performance, and essential fatty acid tissue accretion across the life cycle: a review
Aquaculture International (2023)
-
Fatty acid composition of oil crops: genetics and genetic engineering
Genetic Resources and Crop Evolution (2022)
-
PUFA synthase-independent DHA synthesis pathway in Parietichytrium sp. and its modification to produce EPA and n-3DPA
Communications Biology (2021)