Abstract
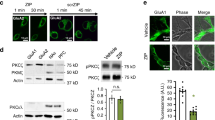
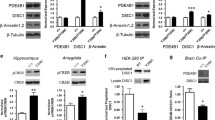
Protein kinase M-ζ (PKM-ζ) is a constitutively active form of atypical protein kinase C that is exclusively expressed in the brain and implicated in the maintenance of long-term memory1,2,3,4,5,6,7,8,9. Most studies that support a role for PKM-ζ in memory maintenance have used pharmacological PKM-ζ inhibitors such as the myristoylated zeta inhibitory peptide (ZIP) or chelerythrine. Here we use a genetic approach and target exon 9 of the Prkcz gene to generate mice that lack both protein kinase C-ζ (PKC-ζ) and PKM-ζ (Prkcz−/− mice). Prkcz−/− mice showed normal behaviour in a cage environment and in baseline tests of motor function and sensory perception, but displayed reduced anxiety-like behaviour. Notably, Prkcz−/− mice did not show deficits in learning or memory in tests of cued fear conditioning, novel object recognition, object location recognition, conditioned place preference for cocaine, or motor learning, when compared with wild-type littermates. ZIP injection into the nucleus accumbens reduced expression of cocaine-conditioned place preference in Prkcz−/− mice. In vitro, ZIP and scrambled ZIP inhibited PKM-ζ, PKC-ι and PKC-ζ with similar inhibition constant (Ki) values. Chelerythrine was a weak inhibitor of PKM-ζ (Ki = 76 μM). Our findings show that absence of PKM-ζ does not impair learning and memory in mice, and that ZIP can erase reward memory even when PKM-ζ is not present.



Similar content being viewed by others
References
Hardt, O., Migues, P. V., Hastings, M., Wong, J. & Nader, K. PKMζ maintains 1-day- and 6-day-old long-term object location but not object identity memory in dorsal hippocampus. Hippocampus 20, 691–695 (2010)
He, Y. Y. et al. PKMζ maintains drug reward and aversion memory in the basolateral amygdala and extinction memory in the infralimbic cortex. Neuropsychopharmacology 36, 1972–1981 (2011)
Li, Y. Q. et al. Inhibition of PKMζ in nucleus accumbens core abolishes long-term drug reward memory. J. Neurosci. 31, 5436–5446 (2011)
Migues, P. V. et al. PKMζ maintains memories by regulating GluR2-dependent AMPA receptor trafficking. Nature Neurosci. 13, 630–634 (2010)
Parsons, R. G. & Davis, M. Temporary disruption of fear-potentiated startle following PKMz inhibition in the amygdala. Nature Neurosci. 14, 295–296 (2011)
Pastalkova, E. et al. Storage of spatial information by the maintenance mechanism of LTP. Science 313, 1141–1144 (2006)
Serrano, P. et al. PKMζ maintains spatial, instrumental, and classically conditioned long-term memories. PLoS Biol. 6, e318 (2008)
Shabashov, D., Shohami, E. & Yaka, R. Inactivation of PKMζ in the NAc shell abolished cocaine-conditioned reward. J. Mol. Neurosci. 47, 546–553 (2011)
Shema, R., Sacktor, T. C. & Dudai, Y. Rapid erasure of long-term memory associations in the cortex by an inhibitor of PKM zeta. Science 317, 951–953 (2007)
Hernandez, A. I. et al. Protein kinase M ζ synthesis from a brain mRNA encoding an independent protein kinase C ζ catalytic domain. Implications for the molecular mechanism of memory. J. Biol. Chem. 278, 40305–40316 (2003)
Chou, M. M. et al. Regulation of protein kinase C ζ by PI 3-kinase and PDK-1. Curr. Biol. 8, 1069–1078 (1998)
Le Good, J. A. et al. Protein kinase C isotypes controlled by phosphoinositide 3-kinase through the protein kinase PDK1. Science 281, 2042–2045 (1998)
Crawley, J. N. Behavioral phenotyping strategies for mutant mice. Neuron 57, 809–818 (2008)
Clarke, J. R., Cammarota, M., Gruart, A., Izquierdo, I. & Delgado-Garcia, J. M. Plastic modifications induced by object recognition memory processing. Proc. Natl Acad. Sci. USA 107, 2652–2657 (2010)
Brabant, C., Quertemont, E. & Tirelli, E. Influence of the dose and the number of drug-context pairings on the magnitude and the long-lasting retention of cocaine-induced conditioned place preference in C57BL/6J mice. Psychopharmacology (Berl.) 180, 33–40 (2005)
Lisman, J. Memory erasure by very high concentrations of ZIP may not be due to PKM-ζ. Hippocampus 22, 648–649 (2011)
Davies, S. P., Reddy, H., Caivano, M. & Cohen, P. Specificity and mechanism of action of some commonly used protein kinase inhibitors. Biochem. J. 351, 95–105 (2000)
Wu-Zhang, A. X., Schramm, C. L., Nabavi, S., Malinow, R. & Newton, A. C. Cellular pharmacology of protein kinase Mζ (PKMζ) contrasts with its in vitro profile: implications for PKMζ as a mediator of memory. J. Biol. Chem. 287, 12879–12885 (2012)
Nishikawa, K., Toker, A., Johannes, F. J., Songyang, Z. & Cantley, L. C. Determination of the specific substrate sequence motifs of protein kinase C isozymes. J. Biol. Chem. 272, 952–960 (1997)
Naik, M. U. et al. Distribution of protein kinase Mζ and the complete protein kinase C isoform family in rat brain. J. Comp. Neurol. 426, 243–258 (2000)
Yao, Y. et al. Matching biochemical and functional efficacies confirm ZIP as a potent competitive inhibitor of PKMζ in neurons. Neuropharmacology 64, 37–44 (2013)
Choi, D. S., Wang, D., Dadgar, J., Chang, W. S. & Messing, R. O. Conditional rescue of protein kinase C ε regulates ethanol preference and hypnotic sensitivity in adult mice. J. Neurosci. 22, 9905–9911 (2002)
Chen, J. et al. The type 1 equilibrative nucleoside transporter regulates anxiety-like behavior in mice. Genes Brain Behav. 6, 776–783 (2007)
Hodge, C. W. et al. Decreased anxiety-like behavior, reduced stress hormones, and neurosteroid supersensitivity in mice lacking protein kinase Cε. J. Clin. Invest. 110, 1003–1010 (2002)
Acknowledgements
We thank V. N. Kharazia and A. J. Lean for assistance. This work was supported by National Institutes of Health grant AA017072 (R.O.M.), a Canadian Institute of Health Research Post-doctoral Fellowship (A.M.L.), and funds provided by the State of California for medical research on alcohol and drug abuse to UCSF.
Author information
Authors and Affiliations
Contributions
A.M.L. designed experiments, collected and analysed data, and wrote the manuscript. B.R.K., J.P.L., M.E.Z., C.Q., T.M. and S.C.F.-W. collected and analysed data. D.W. collected and analysed the data from the in vitro kinase assays. J.D. produced the constructs for the kinase assays and for generation of the mutant mice, and genotyped the mice. R.O.M. designed experiments, analysed data and co-authored the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Figures
This file contains Supplementary Figures 1-3. (PDF 357 kb)
Rights and permissions
About this article
Cite this article
Lee, A., Kanter, B., Wang, D. et al. Prkcz null mice show normal learning and memory. Nature 493, 416–419 (2013). https://doi.org/10.1038/nature11803
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature11803
- Springer Nature Limited
This article is cited by
-
Sex differences in pre- and post-synaptic glutamate signaling in the nucleus accumbens core
Biology of Sex Differences (2023)
-
Ectoderm-derived frontal bone mesenchymal stem cells promote traumatic brain injury recovery by alleviating neuroinflammation and glutamate excitotoxicity partially via FGF1
Stem Cell Research & Therapy (2022)
-
Modeling suggests combined-drug treatments for disorders impairing synaptic plasticity via shared signaling pathways
Journal of Computational Neuroscience (2021)
-
Zeta Inhibitory Peptide attenuates learning and memory by inducing NO-mediated downregulation of AMPA receptors
Nature Communications (2020)
-
GSK-3β activation is required for ZIP-induced disruption of learned fear
Scientific Reports (2020)