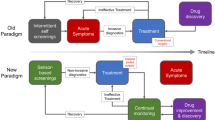
Abstract
The promise of point-of-care medical diagnostics — tests that can be carried out at the site of patient care — is enormous, bringing the benefits of fast and reliable testing and allowing rapid decisions on the course of treatment to be made. To this end, much innovation is occurring in technologies for use in biodiagnostic tests. Assays based on nanomaterials, for example, are now beginning to make the transition from the laboratory to the clinic. But the potential for such assays to become part of routine medical testing depends on many scientific factors, including sensitivity, selectivity and versatility, as well as technological, financial and policy factors.

Similar content being viewed by others
References
Heid, C. A., Stevens, J., Livak, K. J. & Williams, P. M. Real time quantitative PCR. Genome Res. 6, 986–994 (1996).
Engvall, E. & Perlmann, P. Enzyme-linked immunosorbent assay (ELISA). Quantitative assay of immunoglobulin G. Immunochemistry 8, 871–874 (1971).
Rosi, N. L. & Mirkin, C. A. Nanostructures in biodiagnostics. Chem. Rev. 105, 1547–1562 (2005). This review highlights how nanomaterials contribute to diagnostics.
Alivisatos, P. The use of nanocrystals in biological detection. Nature Biotechnol. 22, 47–52 (2004).
Taton, T. A., Mirkin, C. A. & Letsinger, R. L. Scanometric DNA array detection with nanoparticle probes. Science 289, 1757–1760 (2000).
Nam, J. M., Thaxton, C. S. & Mirkin, C. A. Nanoparticle-based bio-bar codes for the ultrasensitive detection of proteins. Science 301, 1884–1886 (2003).
Elghanian, R., Storhoff, J. J., Mucic, R. C., Letsinger, R. L. & Mirkin, C. A. Selective colorimetric detection of polynucleotides based on the distance-dependent optical properties of gold nanoparticles. Science 277, 1078–1081 (1997).
You, C.-C. et al. Detection and identification of proteins using nanoparticle–fluorescent polymer 'chemical nose' sensors. Nature Nanotechnol. 2, 318–323 (2007).
Dubertret, B., Calame, M. & Libchaber, A. J. Single-mismatch detection using gold-quenched fluorescent oligonucleotides. Nature Biotechnol. 19, 365–370 (2001).
Seferos, D. S., Giljohann, D. A., Hill, H. D., Prigodich, A. E. & Mirkin, C. A. Nano-flares: probes for transfection and mRNA detection in living cells. J. Am. Chem. Soc. 129, 15477–15479 (2007).
Wang, Z., Hu, J., Jin, Y., Yao, X. & Li, J. In situ amplified chemiluminescent detection of DNA and immunoassay of IgG using special-shaped gold nanoparticles as label. Clin. Chem. 52, 1958–1961 (2006).
He, L. et al. Colloidal Au-enhanced surface plasmon resonance for ultrasensitive detection of DNA hybridization. J. Am. Chem. Soc. 122, 9071–9077 (2000).
Cao, Y. C., Jin, R. & Mirkin, C. A. Nanoparticles with Raman spectroscopic fingerprints for DNA and RNA detection. Science 297, 1536–1540 (2002).
Wang, J., Liu, G. & Jan, M. R. Ultrasensitive electrical biosensing of proteins and DNA: carbon-nanotube derived amplification of the recognition and transduction events. J. Am. Chem. Soc. 126, 3010–3011 (2004).
Chen, R. J. et al. Noncovalent functionalization of carbon nanotubes for highly specific electronic biosensors. Proc. Natl Acad. Sci. USA 100, 4984–4989 (2003).
Cui, Y., Wei, Q., Park, H. & Lieber, C. M. Nanowire nanosensors for highly sensitive and selective detection of biological and chemical species. Science 293, 1289–1292 (2001).
Soman, C. P. & Giorgio, T. D. Quantum dot self-assembly for protein detection with sub-picomolar sensitivity. Langmuir 24, 4399–4404 (2008).
Catalano, S. M. et al. The role of amyloid-β derived diffusible ligands (ADDLs) in Alzheimer's disease. Curr. Top. Med. Chem. 6, 597–608 (2006).
Perrin, R., Fagan, A. & Holtzman, D. Multimodal techniques for diagnosis and prognosis of Alzheimer's disease. Nature 461, 916–922 (2009).
Georganopoulou, D. G. et al. Nanoparticle-based detection in cerebral spinal fluid of a soluble pathogenic biomarker for Alzheimer's disease. Proc. Natl Acad. Sci. USA 102, 2273–2276 (2005).
Thaxton, C. S. et al. Nanoparticle-based bio-barcode assay redefines 'undetectable' PSA and biochemical recurrence after radical prostatectomy. Proc. Natl Acad. Sci. USA 106, 18437–18442 (2009).
Sano, T., Smith, C. L. & Cantor, C. R. Immuno-PCR: very sensitive antigen detection by means of specific antibody–DNA conjugates. Science 258, 120–122 (1992).
McFarland, A. D. & Van Duyne, R. P. Single silver nanoparticles as real-time optical sensors with zeptomole sensitivity. Nano Lett. 3, 1057–1062 (2003).
Liu, J. & Lu, Y. A colorimetric lead biosensor using DNAzyme-directed assembly of gold nanoparticles. J. Am. Chem. Soc. 125, 6642–6643 (2003).
Lee, J.-S., Han, M. S. & Mirkin, C. A. Colorimetric detection of mercuric ion in aqueous media using DNA-functionalized gold nanoparticles. Angew. Chem. Int. Edn Engl. 46, 4093–4096 (2007).
Hirsch, L. R., Jackson, J. B., Lee, A., Halas, N. J. & West, J. L. A whole blood immunoassay using gold nanoshells. Anal. Chem. 75, 2377–2381 (2003).
Jayasena, S. D. Aptamers: an emerging class of molecules that rival antibodies in diagnostics. Clin. Chem. 45, 1628–1650 (1999).
Ellington, A. D. & Szostak, J. W. In vitro selection of RNA molecules that bind specific ligands. Nature 346, 818–822 (1990). This paper describes the identification of RNA aptamers.
Schena, M., Shalon, D., Davis, R. W. & Brown, P. O. Quantitative monitoring of gene expression patterns with a complementary DNA microarray. Science 270, 467–470 (1995). This study demonstrated the use of microarray analysis for profiling gene expression patterns.
Chan, W. C. & Nie, S. Quantum dot bioconjugates for ultrasensitive nonisotopic detection. Science 281, 2016–2018 (1998).
Bruchez, M. Jr, Moronne, M., Gin, P., Weiss, S. & Alivisatos, A. P. Semiconductor nanocrystals as fluorescent biological labels. Science 281, 2013–2016 (1998).
Park, J. H. et al. Biodegradable luminescent porous silicon nanoparticles for in vivo applications. Nature Mater. 8, 331–336 (2009).
Zheng, G., Patolsky, F., Cui, Y., Wang, W. U. & Lieber, C. M. Multiplexed electrical detection of cancer markers with nanowire sensor arrays. Nature Biotechnol. 23, 1294–1301 (2005).
Acknowledgements
We thank P. Patel for assistance with preparing Table 1. We also thank the US National Science Foundation, the National Cancer Institute and the National Institutes of Health for research support.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
C.A.M. is one of the co-founders of Nanosphere (Northbrook, Illinois).
Additional information
Reprints and permission information is available at http://www.nature.com/reprints. The authors declare competing financial interests: details accompany the full-text HTML version of the paper at http://www.nature.com/nature. Correspondence should be addressed to C.A.M. (chadnano@northwestern.edu).
Rights and permissions
About this article
Cite this article
Giljohann, D., Mirkin, C. Drivers of biodiagnostic development. Nature 462, 461–464 (2009). https://doi.org/10.1038/nature08605
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature08605
- Springer Nature Limited
This article is cited by
-
Advances in nanobiosensors during the COVID-19 pandemic and future perspectives for the post-COVID era
Nano Convergence (2024)
-
Aptamer-based gold nanoparticle aggregates for ultrasensitive amplification-free detection of PSMA
Scientific Reports (2023)
-
Direct digital sensing of protein biomarkers in solution
Nature Communications (2023)
-
Nucleic Acid Based Testing (NABing): A Game Changer Technology for Public Health
Molecular Biotechnology (2023)
-
Stimuli-responsive DNA-based hydrogels for biosensing applications
Journal of Nanobiotechnology (2022)