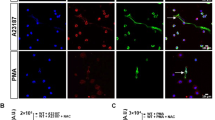
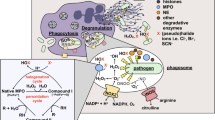
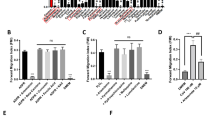
Abstract
Neutrophil leukocytes have a pivotal function in innate immunity. Dogma dictates that the lethal blow is delivered to microbes by reactive oxygen species (ROS) and halogens1,2, products of the NADPH oxidase, whose impairment causes immunodeficiency. However, recent evidence indicates that the microbes might be killed by proteases, activated by the oxidase through the generation of a hypertonic, K+-rich and alkaline environment in the phagocytic vacuole3. Here we show that K+ crosses the membrane through large-conductance Ca2+-activated K+ (BKCa) channels. Specific inhibitors of these channels, iberiotoxin and paxilline, blocked oxidase-induced 86Rb+ fluxes and alkalinization of the phagocytic vacuole, whereas NS1619, a BKCa channel opener, enhanced both. Characteristic outwardly rectifying K+ currents, reversibly inhibited by iberiotoxin, were demonstrated in neutrophils and eosinophils and the expression of the α-subunit of the BK channel was confirmed by western blotting. The channels were opened by the combination of membrane depolarization and elevated Ca2+ concentration, both consequences of oxidase activity. Remarkably, microbial killing and digestion were abolished when the BKCa channel was blocked, revealing an essential and unexpected function for this K+ channel in the microbicidal process.




Similar content being viewed by others
References
Klebanoff, S. J. Antimicrobial mechanisms in neutrophilic polymorphonuclear leukocytes. Semin. Hematol. 12, 117–142 (1975)
Babior, B. M. Phagocytes and oxidative stress. Am. J. Med. 109, 33–44 (2000)
Reeves, E. P. et al. Killing activity of neutrophils is mediated through activation of proteases by K+ flux. Nature 416, 291–297 (2002)
Thrasher, A. J., Keep, N. H., Wientjes, F. & Segal, A. W. Chronic granulomatous disease. Biochim. Biophys. Acta 1227, 1–24 (1994)
Henderson, L. M., Chappell, J. B. & Jones, O. T. The superoxide-generating NADPH oxidase of human neutrophils is electrogenic and associated with an H+ channel. Biochem. J. 246, 325–329 (1987)
Henderson, L. M., Chappell, J. B. & Jones, O. T. Superoxide generation by the electrogenic NADPH oxidase of human neutrophils is limited by the movement of a compensating charge. Biochem. J. 255, 285–290 (1988)
Segal, A. W., Geisow, M., Garcia, R., Harper, A. & Miller, R. The respiratory burst of phagocytic cells is associated with a rise in vacuolar pH. Nature 290, 406–409 (1981)
Galvez, A. et al. Purification and characterization of a unique, potent, peptidyl probe for the high conductance calcium-activated potassium channel from venom of the scorpion Buthus tamulus. J. Biol. Chem. 265, 11083–11090 (1990)
Sanchez, M. & McManus, O. B. Paxilline inhibition of the alpha-subunit of the high-conductance calcium-activated potassium channel. Neuropharmacology 35, 963–968 (1996)
Clapp, L. H. & Tinker, A. Potassium channels in the vasculature. Curr. Opin. Nephrol. Hypertens. 7, 91–98 (1998)
Garcia, M. L. et al. Use of toxins to study potassium channels. J. Bioenerg. Biomembr. 23, 615–646 (1991)
Santos-Silva, J. E., Santos-Silva, M. C., Cunha, F. D. Q. & Assreuy, J. The role of ATP-sensitive potassium channels in neutrophil migration and plasma exudation. J. Pharmac. Exp. Ther. 300, 946–951 (2002)
Maingret, F., Patel, A. J., Lazdunski, M. & Honoré, E. The endocannabinoid anandamide is a direct and selective blocker of the background K+ channel TASK-1. EMBO J. 20, 47–54 (2001)
Lawson, K. Potassium channel openers as potential therapeutic weapons in ion channel disease. Kidney Int. 57, 838–845 (2000)
Fernandez-Fernandez, J. M., Nobles, M., Currid, A., Vazquez, E. & Valverde, M. A. Maxi K+ channel mediates regulatory volume decrease response in a human bronchial epithelial cell line. Am. J. Physiol. Cell Physiol. 283, C1705–C1714 (2002)
DeCoursey, T. E., Morgan, D. & Cherny, V. V. The voltage dependence of NADPH oxidase reveals why phagocytes need proton channels. Nature 422, 531–534 (2003)
Schrenzel, J. et al. Electron currents generated by the human phagocyte NADPH oxidase. Nature 392, 734–737 (1998)
Petreccia, D. C., Nauseef, W. M. & Clark, R. A. Respiratory burst of normal human eosinophils. J. Leukoc. Biol. 41, 283–288 (1987)
Henderson, L. M., Chappell, J. B. & Jones, O. T. Internal pH changes associated with the activity of NADPH oxidase of human neutrophils. Further evidence for the presence of an H+ conducting channel. Biochem. J. 251, 563–567 (1988)
Cherny, V. V., Henderson, L. M., Xu, W., Thomas, L. L. & DeCoursey, T. E. Activation of NADPH oxidase-related proton and electron currents in human eosinophils by arachidonic acid. J. Physiol. (Lond.) 535, 783–794 (2001)
Kaczorowski, G. J. et al. High-conductance calcium activated potassium channel; structure, pharmacology, and function. J. Bioenerg. Biomembr. 28, 255–267 (1996)
Mahomed, G. A. & Anderson, R. Activation of human neutrophils with chemotactic peptide, opsonised zymosan and the calcium ionophore A23187, but not with a phorbol ester, is accompanied by efflux and store-operated influx of calcium. Inflammation 24, 559–569 (2000)
Jankowski, A. & Grinstein, S. A noninvasive fluorimetric procedure for measurement of membrane potential. Quantification of the NADPH oxidase-induced depolarization in activated neutrophils. J. Biol. Chem. 274, 26098–26104 (1999)
Tsien, R. Y. Fluorescent indicators of ion concentrations. Methods Cell Biol. 30, 127–156 (1989)
Vergun, O. et al. Exploration of the role of reactive oxygen species in glutamate neurotoxicity in rat hippocampal neurones in culture. J. Physiol. (Lond.) 531, 147–163 (2001)
Demaurex, N., Monod, A., Lew, D. P. & Krause, K. H. Characterization of receptor-mediated and store-regulated Ca2+ influx in human neutrophils. Biochem. J. 297, 595–601 (1994)
Grinstein, S. & Furuya, W. Cytoplasmic pH regulation in phorbol ester-activated human neutrophils. Am. J. Physiol. 251, C55–C65 (1986)
Willoughby, D., Thomas, R. & Schwiening, C. The effects of intracellular pH changes on resting cytosolic calcium in voltage-clamped snail neurones. J. Physiol. (Lond.) 530, 405–416 (2001)
Salvemini, D., Riley, D. P. & Cuzzocrea, S. SOD mimetics are coming of age. Nature Rev. Drug Discov. 1, 367–374 (2002)
Pick, E. & Keisari, Y. A simple colorimetric method for the measurement of hydrogen peroxide produced by cells in culture. J. Immunol. Methods 38, 161–170 (1980)
Acknowledgements
We thank P. Rich for helpful discussions, S. Ranasinghe for technical help with the FLIPR, and A. Scott for the illustrations. Financial support was provided by the Wellcome Trust and Medical Research Council.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Rights and permissions
About this article
Cite this article
Ahluwalia, J., Tinker, A., Clapp, L. et al. The large-conductance Ca2+-activated K+ channel is essential for innate immunity. Nature 427, 853–858 (2004). https://doi.org/10.1038/nature02356
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature02356
- Springer Nature Limited
This article is cited by
-
A lysosomal K+ channel regulates large particle phagocytosis by facilitating lysosome Ca2+ release
Scientific Reports (2020)
-
α-Hemolysin of uropathogenic E. coli regulates NLRP3 inflammasome activation and mitochondrial dysfunction in THP-1 macrophages
Scientific Reports (2020)
-
Regulation of Reactive Oxygen Species Generation in Cell Signaling
Molecules and Cells (2011)
-
The many roles of NOX2 NADPH oxidase-derived ROS in immunity
Seminars in Immunopathology (2010)
-
Zinc activates neutrophils’ oxidative burst
BioMetals (2010)