Abstract
Background:
There is growing interest in end-of-life care in cancer patients. We aim to characterise health service use and costs in decedents with cancer history and examine factors associated with resource use and costs at life’s end.
Methods:
We used routinely collected claims data to quantify health service use and associated costs in two cohorts of elderly Australians diagnosed with cancer: one cohort died from cancer (n=4271) and the other from non-cancer causes (n=3072). We used negative binomial regression to examine the factors associated with these outcomes.
Results:
Those who died from cancer had significantly higher rates of hospitalisations and medicine use but lower rates of emergency department use than those who died from non-cancer causes. Overall health care costs were significantly higher in those who died from cancer than those dying from other causes; and 40% of costs were expended in the last month of life.
Conclusions:
We analysed health services use and costs from a payer perspective, and highlight important differences in patterns of care by cause of death in patients with a cancer history. In particular, there are growing numbers of highly complex patients approaching the end of life and the heterogeneity of these populations may present challenges for effective health service delivery.
Similar content being viewed by others
Main
Almost 29 million people are living with cancer worldwide according to the most recent 5-year prevalence estimates (Ferlay et al, 2010; Bray et al, 2013). In countries with highly developed health care systems, cancer treatments have improved significantly in recent years such that, relative to the increase in cancer prevalence, cancer mortality is declining (Australian Institute of Health and Welfare (AIHW), 2011; Australian Institute of Health and Welfare (AIHW), 2013; Siegel et al, 2014). As the number of people living with a cancer diagnosis increases, so does the burden on health systems (Sullivan et al, 2011). Many cancer survivors have complex health care needs including ongoing monitoring to prevent recurrence and the management of long-term treatment side-effects and psychological morbidity (Clarke and Currie, 2009; Phillips and Currow, 2010; Boyes et al, 2011; Lambert et al, 2013).
The most resource-intensive periods for cancer patients are the year following diagnosis and the last year of life (Yabroff et al, 2007). Innovation in end-of-life treatments has lagged behind cancer diagnostics and therapeutics. However, the improved tolerability of cancer therapies means that active treatments often continue later into the course of illness (DeVita and Rosenberg, 2012). In recognising the importance of improving the care delivered to patients at the end-of-life, there is a growing literature examining the nature and quality of care received by cancer patients as death approaches (Langton et al, 2014).
Our recent systematic review highlighted the growth in research using routinely collected health care data to examine patterns of health service use, quality of care, and associated costs in decedents with a cancer history (Langton et al, 2014). The review found significant variations in health services received by cancer patients at the end of life, with patterns of care dependant on a wide range of patient (e.g., age, sex, disease characteristics), socio-economic (e.g., location of residence), and health system (e.g., private vs public health care) factors (e.g., Lunney et al, 2002; Legler et al, 2011). Our review also identified a number of important knowledge gaps that we aim to address in the current study. Importantly, most studies in the review were limited to the North American setting and focused on a single aspect of care (such as acute care or palliative services). Moreover, despite research indicating that patterns of end-of-life care vary by cause of death (Moorin and Holman, 2006; Gielen et al, 2010; Setoguchi et al, 2010; Rosenwax et al, 2011), no studies specifically compared health service use in decedents with a cancer history by cause of death (i.e., compared those who died from cancer vs other causes).
Our overall aim is to characterise health service use and associated costs from a health care payer perspective in the last six months of life in a cohort of elderly decedents with a cancer history. We also compare and contrast health service use and costs by cause of death (cancer vs other causes). Finally, we examine the factors associated with health service use and costs in the last 6 months of life.
Methods
Study population
This study used a subset of the cancer cohort from the End-of-Life in Cancer Care (EOL-CC) study, the details of which have been published in a research protocol (Langton et al, 2015). The cohort includes Australian Government Department of Veterans’ Affairs (DVA) clients with a notifiable cancer diagnosis recorded in the New South Wales Central Cancer Registry (NSW CCR) between 1994 and 2009; were at least 65 years at death (Caughey et al, 2008; Pearson and Schaffer, 2014); resided in New South Wales for the 18 months before death; had full health care entitlements and received at least one health service in the last 12 months of life. We included only decedents for whom cause of death information was available (i.e., decedents who died between 2005 and 2008), and further stratified the cohort according to the underlying cause of death, ‘cancer’ or ‘other causes’, using the NSW CCR. The NSW CCR does not collect data on the specific cause of non-cancer deaths; as such we could not report characteristics or outcomes by cause of death beyond the label ‘non-cancer’. Note that in a similar cohort, we found that the most common causes of death were heart failure, cerebrovascular disease, heart disease, chronic lung disease, and dementia; for more information, see our research protocol (Langton et al, 2015).
Department of Veterans' Affairs clients have access to the universal health care arrangements provided to all Australian permanent residents and citizens plus additional DVA-approved services and pharmaceutical items not subsidised for the general population. The vast majority of services of interest in this particular study (e.g., physicians’ visits, hospitalisaitons, ED visits, and most medicines) are those covered by the universal health care arrangements available to all Australians. Examples of additional DVA subsidised services include a range of allied health services and medical items such as bandages. (For more details see: Pearson and Schaffer, 2014; Langton et al, 2015).
Department of Veterans' Affairs clients are a major subgroup of the elderly Australian population. In December 2010, they constituted ∼6% of those aged 65 years and older, 12% of those 75 years and older, and 27% of Australians aged at least 85 years (Australian Government Department of Veterans’ Affairs (DVA), 2010). DVA clients residing in NSW (Australia’s most populous state) account for 34% of the total Australian DVA population and have a similar age and gender profile to clients residing in other Australian states (Australian Government Department of Veterans’ Affairs (DVA), 2010). In addition, when compared with Australians of similar age, DVA clients have very similar rates of health service use (Australian Institute of Health and Welfare (AIHW), 2002; Tooth et al, 2012). Survey data show that DVA clients aged 65 years and older have similar health conditions to the general Australian population aged 65 years and over (Australian Institute of Health and Welfare (AIHW), 2010).
Data sources and linkage
Data were linked by a third party under the custodianship of the DVA, the NSW Ministry of Health and the Cancer Institute NSW, using best practice, probabilistic, and privacy-preserving protocols (Centre for Health Record Linkage (CHeReL), http://www.cherel.org.au/). The linked data sets capture information on DVA client characteristics, residence in an aged care facility, cancer notification history, and cause of death (NSW CCR and Registry of Births, Deaths and Marriages). In addition, they include information about all subsidised health services including hospital admissions (Admitted Patient Data Collection, APDC) and emergency department (ED) presentations (Emergency Department Data Collection, EDDC). The data sets also capture dispensed medicines (Repatriation Pharmaceutical Benefits Scheme, RPBS) and clinician visits and procedures (DVA health services file); procedures include imaging services (e.g., ultrasound, CT, MRI), pathology investigations, therapeutic services (e.g., radiotherapy) and surgery. Additional information about these data sets and the linkage methods used are described in detail in our research protocol (Langton et al, 2015).
Statistical analyses
Our outcomes were specific types of health service use (hospital admissions, ED visits, clinician visits and procedures, and prescribed medicines use) and associated costs in the last 6 months of life based on six constructed ‘months’ consisting of 30 days each; the last ‘month’ included the date of death. All analyses were reported separately to compare those who died from cancer with those who died from other causes (henceforth referred to as the comparison cohort). For descriptive data on health service use and costs, we present means (95% confidence intervals (CIs)) in the body of the manuscript. However, as health service use and costs are generally skewed, with higher means compared with medians for both cohorts, we present medians (inter-quartile ranges (IQRs)) in Supplementary Tables 1 and 2.
Health service use
We calculated person-level use of health services for the last 6 months of life, and for each month to death, by health service type. As our study observation time was constant (all individuals are observed for exactly 180 days up to the date of death), health services use per person is equivalent to rates of use per time period (30 days ‘month’ or 180 days ‘6 months’). We also compared the percentage increase in mean service use across both cohorts, by service type, across the last 6 months of life.
Costs
We allocated unit costs in 2009/10 Australian dollars (AUD) to each item of health service use. Total costs were calculated as the sum of hospital costs (excluding the pharmacy component for private hospital patients that is captured in dispensing data), ED costs, clinician visits and procedures, and prescription medicines costs. More details about the cost calculations by service type are reported elsewhere (Reeve and Haas, 2014; Langton et al, 2015).
Factors associated with health service use and costs in the last 6 months of life
We used negative binomial regression to examine factors associated with health service use (hospitalisations, ED visits, clinician visits and procedures, and prescription drugs), overall costs and specific health care costs. Factors were selected based on those commonly used in similar studies (Langton et al, 2014) and included cause of death (cancer vs other cause) and a range of other patient characteristics including age, sex, co-morbidity burden, time from cancer diagnosis to death, cancer spread at diagnosis, location of residence and place of death (Currow et al, 2012; McNamara et al, 2013; Langton et al, 2014, 2015). Co-morbidity burden was estimated in periods before the last 6 months of life using two methods: hospitalisation codes (Charlson index; Sundararajan et al, 2004) and prescription dispensing history (RxRisk; Sloan et al, 2003). Two methods were used as different estimates of co-morbidity burden are obtained depending on the index used, with the Charlson likely to under ascertain morbidity (Lu et al, 2011); this is discussed in more detail elsewhere (Langton et al, 2015).
We represented the strength of the estimated associations by adjusted incident rate ratios with 95% confidence intervals, and two-tailed P-values of <0.05 used as a criterion for statistical significance. We used SAS software version 9.3 (SAS Institute) for data manipulation and performed statistical analyses using STATA software version 12 (StataCorp).
Ethics
The NSW Population and Health Services Ethics Committee (approval number 2013/11/494) and the Department of Veterans’ Affairs Human Research Ethics Committee (E013/015) approved this project.
Results
Cohort characteristics
Of the 7343 decedents meeting our study eligibility criteria, 4271 (60%) died from cancer and the remainder died from other causes (Table 1). Both cohorts consisted predominantly of men (>65%) and the median age at death was 85 and 86 years, for the cohort who died from cancer and the comparison cohort, respectively.
The most commonly diagnosed cancers were similar to the general Australian population with the exception of breast cancer that was under-represented due to a predominantly male cohort (Australian Institute of Health and Welfare (AIHW), 2011) (Table 2). The cohort who died from cancer had a greater proportion of patients with lung cancer and patients with multiple cancers than the comparison cohort. Co-morbidity burden (as measured by hospitalisations) was similar in both cohorts. However, when measured by prescriptions dispensed, the proportion of decedents with six or more co-morbidities was higher in the comparison cohort (46.1%) than the cohort that died from cancer (39.4%).
Health service use and costs during the last 6 months of life
Overall health services use and costs
More than 90% of people dying from cancer and 80% of the comparison cohort were admitted to hospital at least once. Approximately two-thirds of people in both cohorts visited the ED and fewer than 10% spent time in intensive care. Hospitalisations were the greatest drivers of cost, accounting for more than 80% of total health care costs in the last 6 months of life. Health care costs were higher in the cohort dying from cancer; the average total cost of health care per patient was AUD$ 30 001 in the cohort who died from cancer and $26 131 in the comparison cohort (Table 3, see Supplementary Table 1 for median (IQR) service use and costs).
Hospitalisations
The mean number of hospitalisations per person was 3.1 (95% CI 3.1–3.2; with an average total cost of $24 531) in the cohort dying from cancer and 2.7 in the comparison cohort (95% CI 2.6–2.8; average total cost $21 444). Of those admitted to hospital at least once, the mean number of days accrued in hospital was 32.4 (95% CI 31.5–33.3) in the cohort who died from cancer and 30.4 (95% CI 29.1–31.6) in the comparison cohort (Table 3). Most hospitalisations were for reasons other than palliative and hospice care; only 21.5% of the cohort dying from cancer and 5.1% of the comparison cohort received a palliative care service while in hospital.
Emergency department visits
Emergency department visits were few across both cohorts with a mean of one visit per person at an average cost of $533 for the cohort dying from cancer and $613 for the comparison cohort (Table 3). Approximately 80% of all ED visits resulted in admission to hospital across both cohorts.
Clinician visits and procedures
Clinician visits/procedures and associated costs were greater in the cohort dying from cancer (90.7 services 95% CI 88.9–92.4, AUD$ 7337) than the comparison cohort (84.5 services 95% CI 82–87, AUD$ 6249). These differences were driven by higher use of medical consultations (GPs and specialists) and diagnostic and therapeutic procedures in the cohort who died from cancer (Table 3). Use of specialist pain and palliative care services was low in both cohorts; about 10% of those who died from cancer and 1% of the comparison cohort received a specific pain and palliative care management service. However, it is possible that issues related to palliative care may have been covered in chronic disease management services (more than 1/5 of decedents in both cohorts received chronic disease management services).
Prescription medicines
Both cohorts had similar rates of prescribed medicines use with an average of 41 prescriptions per decedent over the last 6 months of life (Table 3); the most commonly prescribed medicines were those used for symptom management and pain relief (e.g., paracetamol, temazepam, and oxycodone) (Supplementary Table 2). The mean cost of medicines per person in the cohort dying from cancer was AUD$ 1930 compared with AUD$ 1609 for the comparison cohort; the difference was driven by the costs of antineoplastic and immunomodulating agents (mean costs of $622 per person in those dying from cancer compared with $247 in the comparison cohort). However, it is important to note that the percentage of patients receiving any cancer therapeutic in the last 6 months of life was low across both cohorts; 10% in the cohort dying from cancer and 7% in the comparison cohort (classification of cancer therapeutics is detailed elsewhere (Langton et al, 2015).
Monthly changes in health service use and costs across the last 6 months of life
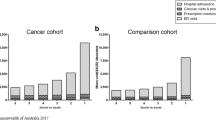
In both cohorts, rates of health service use increased as death approached such that around 40% of all costs incurred in the last 6 months of life were associated with the care received in the last month of life (Figures 1 and 2). Specifically, hospitalisations in both cohorts increased, with the greatest increase seen in the last 2 months of life (67% increase in the cohort who died from cancer and 80% increase in the comparison cohort). Similarly, monthly rates of ED use increased substantially during the last 3 months of life; two-fold in the cohort who died from cancer and three-fold in the comparison cohort. The overall rate of visits and procedures per month increased from about 10 services at 6 months before death to more than 25 services in the last month of life, representing more than a 150% increase in service use. Finally, the rate of dispensed medicines per month increased by 36% in the cohort who died from cancer and 19% in the comparison cohort.
Health services use in the last 6 months of life by cohort and by month. (A) Prescription medicines dispensed per decedent per month, (B) clinician visits and procedures per decedent per month, (C) hospital admissions per decedent per month, (D) emergency department visits per decedent per month. © Commonwealth of Australia 2016.
Factors associated with health service use and costs in the last 6 months of life
Multivariable analyses demonstrated that persons who died from cancer had significantly higher rates of hospitalisations (adjusted IRR: 1.07; 95% CI: 1.02–1.12, P=0.010) and prescribed medicine use (adjusted IRR: 1.07; 95% CI: 1.04–1.11, P<0.001) but lower rates of ED use (adjusted IRR: 0.85; 95% CI: 0.80–0.90, P<0.001) than the comparison cohort (Supplementary Table 3). A number of other factors were associated with increased use of all types of health services including a younger age at death, residence outside major cities, and higher co-morbidity burden. Finally, some factors were uniquely associated with an increased use of a specific type of health service. In particular, a longer time from cancer diagnosis to death was associated with increased prescription medicines use, but fewer hospitalisations, ED visits and clinician visits/procedures. In addition, males and those who died in residential aged care had significantly fewer hospitalisations, ED visits, and clinician visits/procedures. People living in areas of higher socio-economic advantage received significantly more prescription drugs, clinician visits/procedures, and hospitalisations but were less likely to visit the ED compared with persons living in disadvantaged areas (see Supplementary Table 3 for more detail).
Costs
The cohort that died from cancer had significantly higher average overall costs than the comparison cohort (adjusted IRR: 1.06; 95% CI: 1.02–1.11, P=0.006) (Table 4); they had 6% higher costs for clinician visits and procedures (adjusted IRR: 1.06; 95% CI: 1.02–1.10, P=0.003) and 29% higher costs for prescription medicines (adjusted IRR: 1.29; 95% CI: 1.23–1.36, P<0.001) (Supplementary Table 4). ED costs were 17% lower for those who died from cancer than the comparison cohort (adjusted IRR: 0.83; 95% CI: 0.73–0.95, P=0.005). A number of factors were associated with higher average overall costs including being male, living in a major city, and having a higher co-morbidity burden. Those who died at older ages and 12 months or more after cancer diagnosis had up to 20% lower overall costs. Persons who died outside of hospital had at least 42% lower overall costs compared with those who died in hospital.
Discussion
This study provides a comprehensive analysis from the health care payer perspective of end-of-life health service use and costs. It compares service use and costs by cause of death in a population of people over 65 with a history of cancer. Regardless of cause of death, health care use increased during the last 6 months of life, particularly in the last 2 months of life. Consistent with previous research, hospital-based services were heavily used at the end of life and accounted for the majority of health care costs (Levinsky et al, 2001; Emanuel et al, 2002; Polder et al, 2006). However, there were some important differences in health service use according to cause of death; prescription medicine use, and clinician visits and procedures were significantly higher in decedents who died from cancer than in persons who died from other causes. In contrast, patients who died from causes other than cancer were significantly more likely to visit the ED, particularly in the last 2 months of life. Total costs were 6% higher in the cohort who died from cancer vs the cohort who died from other causes.
Our study sheds light on the complexity of end-of-life cancer patient populations and the full range of health care services subsidised by a health care payer in patients who die from cancer or from other conditions; this is particularly important in the context of growing numbers of people dying with a cancer history (Australian Institute of Health and Welfare (AIHW), 2011, 2013; Siegel et al, 2014). Our results suggest that a cancer cause of death, rather than a cancer history, may predict patterns of health service use at the end-of-life. However, there are several other factors associated with health service uses such as co-morbidity burden; about 90% of decedents had at least one co-morbidity in addition to cancer and not surprisingly, multi-morbidity was associated with increased health service use and costs. Some have argued that ‘multi-morbidity’ is now the most common chronic condition; as such it will continue to be an important issue when evaluating the quality of end-of-life care (Tinetti et al, 2012; Banerjee, 2014). For instance, future end-of-life treatment guidelines may need to be tailored to multi-morbid patients rather than single diseases such as cancer. However, there is also evidence that specific diseases may influence the end-of-life trajectory; for example a recent study showed that frail patients (those with conditions such as dementia and Alzheimer’s disease prior to a cancer diagnosis) died sooner following a cancer diagnosis compared with non-frail patients (Urquhart et al, 2015). Although we did not examine frailty specifically, our results suggest that certain populations may have more complex end-of-life needs, for example, populations that survive for a shorter time following cancer diagnosis were more likely to be admitted to hospital or visit the ED in the last 6 months of life which may suggest that existing community or home care services are not meeting the needs of these patients.
Despite research showing that many patients would prefer to die outside the hospital setting (Barnato et al, 2007; Zhang et al, 2009; Fischer et al, 2013), the rates of hospital use and proportion of patients dying in hospital suggests that this is the dominant pathway for elderly Australian cancer patients. Regardless of cause of death, decedents received an average of more than 40 prescription medicines, close to 90 visits to clinicians and procedures, and were admitted to hospital around three times in the last 6 months of life. Although the overall number of services were similar, there were important differences in specific patterns of health care use by cohort. For example, palliative service use was higher in the cohort who died from cancer; this may reflect the fact that palliative services are traditionally geared towards patients dying from cancer and trajectories of decline in cancer patients tend to be more predicable than other causes of death despite the fact that many other conditions are amendable to palliative care (Rosenwax and McNamara, 2006).
It is well recognised that high volumes of expensive services do not equate with quality end-of-life care. For example, North American research suggests that receipt of certain services (e.g., hospice, primary care) and omission of others (e.g., chemotherapy, ICU) may reflect higher quality care, however in practice, defining and measuring the quality of care is challenging due to variation in patient perspectives on what constitutes high-quality care in the setting of a life-limiting illness (Earle et al, 2003; Grunfeld et al, 2008; Ho et al, 2011). These measures have not been developed or tested in the Australian setting; however, if we were to interpret our findings in the context of these indicators as described in the literature (Earle et al, 2005; Barbera et al, 2015b), the low rates of chemotherapy use can be interpreted as favourable. Our results are consistent with several studies showing that the likelihood of receiving ‘aggressive’ or life-sustaining treatments such as chemotherapy, ED visits, and admission to intensive care units and hospitals decreases with increasing age (Virnig et al, 2002; Gagnon et al, 2004; Shugarman et al, 2007, 2008; Burge et al, 2008; Setoguchi et al, 2008; Ho et al, 2011). The low uptake of pain and palliative specialist services is not desirable, however, rates of palliative care services delivered may have been underestimated due to hospital and community coding practices in Australia (Australian Institute of Health and Welfare (AIHW), 2014). Further research is required to benchmark rates of these quality indicators to the Australian setting, as has been done in other jurisdictions (Earle et al, 2005; Barbera et al, 2015a).
For the purposes of identifying targets for resource allocation to improve end-of-life care, research supports consideration of broader quality indicators (i.e., indicators outside the hospital setting and before the last month of life) (Langton et al, 2014). For example, the volume of health services use in the period before cancer diagnosis has been found to predict the time from cancer diagnosis to death (Urquhart et al, 2015). The nature of health service use is also important as there is increasing evidence suggesting that continuity of primary care and interventions such as multidisciplinary case conferences involving palliative care teams can reduce subsequent ED visits and hospitalisations and may result in more cost-effective care overall (Burge et al, 2003; Abernethy et al, 2013; Almaawiy et al, 2014; Seow et al, 2016). Indeed, some of the most common reasons for ED visits are for issues such as uncontrolled pain and failure to cope that may be more effectively managed outside of the hospital setting (Barbera et al, 2010). On a larger scale, investment in community-based services and integrated care across community and acute sectors could result in significant cost savings at a health system level.
There are a number of strengths of our study, including our patient-level analysis using multiple linked routine data collections. However, our findings relating to DVA clients may not be generalizable to the elderly Australian cancer population. There is evidence that DVA clients have similar rates of health service use when compared with Australians of a similar age (Australian Institute of Health and Welfare (AIHW), 2002; Tooth et al, 2012); although this has not been examined specifically in relation to end-of-life care. Comparison of our current results with the most recent study on end-of-life care in the general population in Australia (Rosenwax et al, 2011) suggests rates of hospital admissions, ED visits, and death in hospital are similar to DVA clients. However, direct comparisons are not possible due to the different end-of-life period and cohort inclusion criteria of each study. Moreover, our results do not provide information on patterns of end-of-life care in people younger than 65 years of age, a group that represents up to one-third of cancer deaths in Australia (Australian Institute of Health and Welfare (AIHW), 2014; AIHW, 2015). Our data reflect the most recent available at the time this research was undertaken, however, there may have been changes in practice in Australia since 2008. The significant time lag between the end of the study period and publication is a common issue for research using routinely collected health data to examine patterns of health care use (Langton et al, 2014).
The retrospective study design used here has been criticised by some as reducing the validity and representativeness of cohorts (Bach et al, 2004), yet there is also research showing similar patterns of health care use when prospective and retrospective methodologies are directly compared (Pyenson et al, 2004; Setoguchi et al, 2008). Certainly, in clinical practice, the prediction of survival time across patient populations is challenging (Aabom et al, 2005; Boyd and Murray, 2010). While using death as a starting point and looking back 6 or 12 months is a common method for studies such as this (Langton et al, 2014), there may be some merit in further developing methodologies that examine end-of-life care in the period between a clinical event (such as initiation of opioids or enrolment in hospice) and death (Guest et al, 2006). Cancer is one of the few diseases with a recognizable terminal phase, however, service and cost outcomes are likely to represent a mix of different clinical scenarios because of differing trajectories of decline at the end-of-life even in patients with similar disease profiles (Lunney et al, 2002; Murray et al, 2005). Related to this, our comparison cohort was a heterogeneous group of patients who died from a range of conditions including dementia, organ failure, and lung disease that each have their own distinct patterns of end-of-life care (Rosenwax and McNamara, 2006; Goodman et al, 2010; Setoguchi et al, 2010; Langton et al, 2013).
Our study provides a near-complete analysis of hospital- and community-based services use and costs in the last 6 months of life for people diagnosed with cancer. We found important differences in the patterns of end-of-life care by underlying cause of death in patients with a cancer history but a range of other factors (e.g., morbidity burden, age) also contribute to the care received by terminally ill cancer patients. Our findings are consistent with the global literature and highlight that the most elderly decedents use fewer health care services across the board (Setoguchi et al, 2008; Shugarman et al, 2008; Ho et al, 2011); however, we do not know if this reflects best quality care for elderly populations. Indeed, there are calls to examine better ways to translate models of quality care into measurable end-points that are required to establish interventions as evidence-based (Halpern, 2015). The complexity and heterogeneity of our cohorts and that of similar studies presents challenges for end-of-life planning. To this end a focus, on making best use of existing data to effectively identify patient complexity and health care needs before the final few days or weeks of life will be important.
Change history
24 May 2016
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Aabom B, Kragstrup J, Vondeling H, Bakketeig LS, Stovring H (2005) Defining cancer patients as being in the terminal phase: who receives a formal diagnosis, and what are the effects? J Clin Oncol 23: 7411–7416.
Abernethy AP, Currow DC, Shelby-James T, Rowett D, May F, Samsa GP, Hunt R, Williams H, Esterman A, Phillips PA (2013) Delivery strategies to optimize resource utilization and performance status for patients with advanced life-limiting illness: results from the ‘palliative care trial’ [ISRCTN 81117481]. J Pain Symptom Manage 45: 488–505.
Almaawiy U, Pond GR, Sussman J, Brazil K, Seow H (2014) Are family physician visits and continuity of care associated with acute care use at end-of-life? A population-based cohort study of homecare cancer patients. Palliat Med 28: 176–183.
Australian Government Department of Veterans’ Affairs (DVA) (2010) Treatment population statistics: quarterly report—December 2010.
Australian Institute of Health and Welfare (AIHW) (2002) Health care usage and costs. A comparison of veterans and war widows and widowers with the rest of the community (Canberra, Australia). Report number: cat. no. PHE 42.
Australian Institute of Health and Welfare (AIHW) (2010) Australia’s health 2010 (AIHW). Australia's health series no. 10, cat. no. AUS122.
Australian Institute of Health and Welfare (AIHW) (2011) Cancer in Australia: actual incidence and mortality data from 1982 to 2007 and projections to 2010. Asia Pac J Clin Oncol 7: 325–338.
Australian Institute of Health and Welfare (AIHW) (2013) Cancer survival and prevalence in Australia: period estimates from 1982 to 2010. Asia Pac J Clin Oncol 9: 29–39.
Australian Institute of Health and Welfare (AIHW) (2014) Palliative care services in Australia 2014 (Canberra, Australia) Cat. no. HWI 128.
Australian Institute of Health and Welfare (AIHW) (2015) Age at death Available from http://www.aihw.gov.au/deaths/age-at-death/ accessed July 2015.
Bach PB, Schrag D, Begg CB (2004) Resurrecting treatment histories of dead patients: a study design that should be laid to rest. JAMA 292: 2765–2770.
Banerjee S (2014) Multimorbidity-older adults need health care that can count past one. Lancet 385: 587–589.
Barbera L, Seow H, Sutradhar R, Chu A, Burge F, Fassbender K, McGrail K, Lawson B, Liu Y, Pataky R, Potapov A (2015a) Quality Indicators of End-of-Life Care in Patients With Cancer: What Rate Is Right? J Oncol Pract JOP 2015: 004416.
Barbera L, Seow H, Sutradhar R, Chu A, Burge F, Fassbender K, McGrail K, Lawson B, Liu Y, Pataky R, Potapov A (2015b) Quality of end-of-life cancer care in Canada: a retrospective four-province study using administrative health care data. Curr Oncol 22: 341–355.
Barbera L, Taylor C, Dudgeon D (2010) Why do patients with cancer visit the emergency department near the end of life? C Can Med Assoc J 182: 563–568.
Barnato AE, Herndon MB, Anthony DL, Gallagher PM, Skinner JS, Bynum JPW, Fisher ES (2007) Are regional variations in end-of-life care intensity explained by patient preferences?: A Study of the US Medicare Population. Med Care 45: 386–393.
Boyd K, Murray SA (2010) Recognising and managing key transitions in end of life care. BMJ 341: c4863.
Boyes AW, Girgis A, D’Este C, Zucca AC (2011) Flourishing or floundering? Prevalence and correlates of anxiety and depression among a population-based sample of adult cancer survivors 6 months after diagnosis. J Affect Disord 135: 184–192.
Bray F, Ren JS, Masuyer E, Ferlay J (2013) Global estimates of cancer prevalence for 27 sites in the adult population in 2008. Int J Cancer 132: 1133–1145.
Burge F, Lawson B, Johnston G (2003) Family physician continuity of care and emergency department use in end-of-life cancer care. Med Care 41: 992–1001.
Burge FI, Lawson BJ, Johnston GM, Grunfeld E (2008) A population-based study of age inequalities in access to palliative care among cancer patients. Med Care 46: 1203–1211.
Caughey GE, Vitry AI, Gilbert AL, Roughead EE (2008) Prevalence of comorbidity of chronic diseases in Australia. BMC Public Health 8: 221.
Clarke DM, Currie KC (2009) Depression, anxiety and their relationship with chronic diseases: a review of the epidemiology, risk and treatment evidence. Med J Aust 190: S54–S60.
Currow DC, Allingham S, Bird S, Yates P, Lewis J, Dawber J, Eagar K (2012) Referral patterns and proximity to palliative care inpatient services by level of socio-economic disadvantage. A national study using spatial analysis. BMC Health Serv Res 12: 424.
DeVita VT, Rosenberg SA (2012) Two hundred years of cancer research. N Engl J Med 366: 2207–2214.
Earle CC, Neville BA, Landrum MB, Souza JM, Weeks JC, Block SD, Grunfeld E, Ayanian JZ (2005) Evaluating claims-based indicators of the intensity of end-of-life cancer care. Int J Qual Health Care 17: 505–509.
Earle CC, Park ER, Lai B, Weeks JC, Ayanian JZ, Block S (2003) Identifying potential indicators of the quality of end-of-life cancer care from administrative data. J Clin Oncol 21: 1133–1138.
Emanuel EJ, Ash A, Yu W, Gazelle G, Levinsky NG, Saynina O, McClellan M, Moskowitz M (2002) Managed care, hospice use, site of death, and medical expenditures in the last year of life. Arch Intern Med 162: 1722–1728.
Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM (2010) Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer 127: 2893–2917.
Fischer S, Min S-J, Cervantes L, Kutner J (2013) Where do you want to spend your last days of life? Low concordance between preferred and actual site of death among hospitalized adults. J Hosp Med 8: 178–183.
Gagnon B, Mayo NE, Hanley J, MacDonald N (2004) Pattern of care at the end of life: does age make a difference in what happens to women with breast cancer? J Clin Oncol 22: 3458–3465.
Gielen B, Remacle A, Mertens R (2010) Patterns of health care use and expenditure during the last 6 months of life in Belgium: differences between age categories in cancer and non-cancer patients. Health Policy (New York) 97: 53–61.
Goodman C, Evans C, Wilcock J, Froggatt K, Drennan V, Sampson E, Blanchard M, Bissett M, Iliffe S (2010) End of life care for community dwelling older people with dementia: an integrated review. Int J Geriatr Psychiatry 25: 329–337.
Grunfeld E, Urquhart R, Mykhalovskiy E, Folkes A, Johnston G, Burge FI, Earle CC, Dent S (2008) Toward population-based indicators of quality end-of-life care: testing stakeholder agreement. Cancer 112: 2301–2308.
Guest JF, Ruiz FJ, Greener MJ, Trotman IF (2006) Palliative care treatment patterns and associated costs of healthcare resource use for specific advanced cancer patients in the UK. Eur J Cancer Care (Engl) 15: 65–73.
Halpern SD (2015) Toward Evidence-Based End-of-Life Care. N Engl J Med. 373: 2001–2003.
Ho TH, Barbera L, Saskin R, Lu H, Neville BA, Earle CC (2011) Trends in the aggressiveness of end-of-life cancer care in the universal health care system of Ontario, Canada. J Clin Oncol 29: 1587–1591.
Lambert SD, Girgis A, Lecathelinais C, Stacey F (2013) Walking a mile in their shoes: anxiety and depression among partners and caregivers of cancer survivors at 6 and 12 months post-diagnosis. Support Care Cancer 21: 75–85.
Langton JM, Blanch B, Drew AK, Haas M, Ingham JM, Pearson S-A (2014) Retrospective studies of end-of-life resource utilization and costs in cancer care using health administrative data: A systematic review. Palliat Med 28: 1167–1196.
Langton JM, Srasuebkul P, Buckley NA, Pearson S-A (2013) Prescription drug use in the last 12 months of life: Observations in a cohort of elderly patients. Proceedings of the Abstracts of the 29th International Conference on Pharmacoepidemiology & Therapeutic Risk Management 25–28 August 2013; Montréal, Canada. Pharmacoepidemiology and Drug Safety 22 (Suppl 1): p 352.
Langton JM, Srasuebkul P, Reeve R, Parkinson B, Gu Y, Buckley NA, Haas M, Viney R, Pearson S-A (2015) Resource use, costs and quality of end of life care: Observations in a cohort of elderly Australian cancer decedents. Implement Sci 10: 25.
Legler A, Bradley EH, Carlson MDA (2011) The effect of comorbidity burden on health care utilization for patients with cancer using hospice. J Palliat Med 14: 751–756.
Levinsky NG, Yu W, Ash A, Moskowitz M, Gazelle G, Saynina O, Emanuel EJ (2001) Influence of age on Medicare expenditures and medical care in the last year of life. JAMA 286: 1349–1355.
Lu CY, Barratt J, Vitry A, Roughead E (2011) Charlson and Rx-Risk comorbidity indices were predictive of mortality in the Australian health care setting. J Clin Epidemiol 64: 223–228.
Lunney JR, Lynn J, Hogan C (2002) Profiles of older medicare decedents. J Am Geriatr Soc 50: 1108–1112.
McNamara BA, Rosenwax LK, Murray K, Currow DC (2013) Early admission to community-based palliative care reduces use of emergency departments in the ninety days before death. J Palliat Med 16: 774–779.
Moorin RE, Holman CDJ (2006) The effects of socioeconomic status, accessibility to services and patient type on hospital use in Western Australia: a retrospective cohort study of patients with homogenous health status. BMC Health Serv Res 6: 74.
Murray SA, Kendall M, Boyd K, Sheikh A (2005) Illness trajectories and palliative care. BMJ 330: 1007–1011.
Pearson S-A, Schaffer A (2014) The use and impact of cancer medicines in routine clinical care: methods and observations in a cohort of elderly Australians. BMJ Open 4: e004099.
Phillips JL, Currow DC (2010) Cancer as a chronic disease. Collegian 17: 47–50.
Polder JJ, Barendregt JJ, van Oers H (2006) Health care costs in the last year of life—the Dutch experience. Soc Sci Med 63: 1720–1731.
Pyenson B, Connor S, Fitch K, Kinzbrunner B (2004) Medicare cost in matched hospice and non-hospice cohorts. J Pain Symptom Manag 28: 200–210.
Reeve R, Haas M (2014) Estimating the cost of Emergency Department presentations in NSW, CHERE Working Paper.
Rosenwax LK, McNamara BA (2006) Who receives specialist palliative care in Western Australia—and who misses out. Palliat Med 20: 439–445.
Rosenwax LK, McNamara BA, Murray K, McCabe RJ, Aoun SM, Currow DC (2011) Hospital and emergency department use in the last year of life: a baseline for future modifications to end-of-life care. Med J Aust 194: 570–573.
Seow H, Barbera L, Pataky R, Lawson B, O’Leary E, Fassbender K, McGrail K, Burge F, Brouwers M, Sutradhar R (2016) Does increasing homecare nursing reduce emergency department visits at the end of life? a population-based cohort study of cancer decedents. J Pain Symptom Manage 51: 204–212.
Setoguchi S, Earle CC, Glynn R, Stedman M, Polinski JM, Corcoran CP, Haas JS (2008) Comparison of prospective and retrospective indicators of the quality of end-of-life cancer care. J Clin Oncol 26: 5671–5678.
Setoguchi S, Glynn RJ, Stedman M, Flavell CM, Levin R, Stevenson LW (2010) Hospice, opiates, and acute care service use among the elderly before death from heart failure or cancer. Am Heart J 160: 139–144.
Shugarman LR, Bird CE, Schuster CR, Lynn J (2007) Age and gender differences in Medicare expenditures at the end of life for colorectal cancer decedents. J Women’s Heal 16: 214–227.
Shugarman LR, Bird CE, Schuster CR, Lynn J (2008) Age and gender differences in medicare expenditures and service utilization at the end of life for lung cancer decedents. Womens Heal Issues 18: 199–209.
Siegel R, Ma J, Zou Z, Jemal A (2014) Cancer statistics, 2014. CA Cancer J Clin 64: 9–29.
Sloan KL, Sales AE, Liu C-F, Fishman P, Nichol P, Suzuki NT, Sharp ND (2003) Construction and characteristics of the RxRisk-V: a VA-adapted pharmacy-based case-mix instrument. Med Care 41: 761–774.
Sullivan R, Peppercorn J, Sikora K, Zalcberg J, Meropol NJ, Amir E, Khayat D, Boyle P, Autier P, Tannock IF, Fojo T, Siderov J, Williamson S, Camporesi S, McVie JG, Purushotham AD, Naredi P, Eggermont A, Brennan MF, Steinberg ML, De Ridder M, McCloskey SA, Verellen D, Roberts T, Storme G, Hicks RJ, Ell PJ, Hirsch BR, Carbone DP, Schulman KA, Catchpole P, Taylor D, Geissler J, Brinker NG, Meltzer D, Kerr D, Aapro M (2011) Delivering affordable cancer care in high-income countries. Lancet Oncol 12: 933–980.
Sundararajan V, Henderson T, Perry C, Muggivan A, Quan H, Ghali WA (2004) New ICD-10 version of the Charlson comorbidity index predicted in-hospital mortality. J Clin Epidemiol 57: 1288–1294.
Tinetti ME, Fried TR, Boyd CM (2012) Designing health care for the most common chronic condition—multimorbidity. JAMA 307: 2493–2494.
Tooth LR, Hockey R, Treloar S, McClintock C, Dobson A (2012) Does government subsidy for costs of medical and pharmaceutical services result in higher service utilization by older widowed women in Australia? BMC Health Serv Res 12: 179.
Urquhart R, Johnston G, Abdolell M, Porter GA (2015) Patterns of health care utilization preceding a colorectal cancer diagnosis are strong predictors of dying quickly following diagnosis. BMC Palliat Care 14: 2.
Virnig BA, Marshall McBean A, Kind S, Dholakia R (2002) Hospice use before death: variability across cancer diagnoses. Med Care 40: 73–78.
Yabroff KR, Warren JL, Brown ML (2007) Costs of cancer care in the USA: a descriptive review. Nat Clin Pract Oncol 4: 643–656.
Zhang B, Wright AA, Huskamp HA, Nilsson ME, Maciejewski ML, Earle CC, Block SD, Maciejewski PK, Prigerson HG (2009) Health care costs in the last week of life associations with end-of-life conversations. Arch Intern Med 169: 480–488.
Acknowledgements
We thank the Centre for Health Record Linkage for undertaking the data linkage process. We also thank Ms Melisa Litchfield for her assistance in supporting the data analysis. Funding: This research is supported, in part, by a National Health and Medical Research Council Health (NHMRC) Services Research Capacity Building Grant (ID: 571926), NHMRC Centre of Research Excellence in Medicines and Ageing (ID: 1060407), Cancer Australia Grant (ID: 568773) and a University of Sydney, Faculty of Pharmacy Small Project Grant. SP is supported by a Cancer Institute New South Wales Career Development Fellowship (ID: 12/CDF/2-25).
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Competing interests
The Australian Government Department of Veterans’ Affairs is the copyright owner of the data presented in this manuscript (including all tables, figures and supplementary content) and the authors have obtained permission from the Australian Government Department of Veterans’ Affairs to publish this manuscript. The views expressed in this version of the work do not necessarily represent the views of the Minister for Veterans’ Affairs or the Department of Veterans’ Affairs. The Commonwealth does not give any warranty nor accept any liability in relation to the contents of this work.
Additional information
This work is published under the standard license to publish agreement. After 12 months the work will become freely available and the license terms will switch to a Creative Commons Attribution-NonCommercial-Share Alike 4.0 Unported License.
Supplementary Information accompanies this paper on British Journal of Cancer website
Supplementary information
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 4.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/4.0/
About this article
Cite this article
Langton, J., Reeve, R., Srasuebkul, P. et al. Health service use and costs in the last 6 months of life in elderly decedents with a history of cancer: a comprehensive analysis from a health payer perspective. Br J Cancer 114, 1293–1302 (2016). https://doi.org/10.1038/bjc.2016.75
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bjc.2016.75
- Springer Nature Limited
Keywords
This article is cited by
-
Healthcare Costs at the End of Life for Patients with Non-cancer Diseases and Cancer in Denmark
PharmacoEconomics - Open (2023)
-
Longitudinal patterns and predictors of healthcare utilization among cancer patients on home-based palliative care in Singapore: a group-based multi-trajectory analysis
BMC Medicine (2022)
-
Care pathways at end-of-life for cancer decedents: registry based analyses of the living situation, healthcare utilization and costs for all cancer decedents in Norway in 2009-2013 during their last 6 months of life
BMC Health Services Research (2022)
-
The decisional balance, attitudes, and practice behaviors, its predicting factors, and related experiences of advance care planning in Taiwanese patients with advanced cancer
BMC Palliative Care (2022)
-
Hospitalizations and emergency department visits trends among elderly individuals in proximity to death: a retrospective population-based study
Scientific Reports (2021)