Abstract
The nuclear factor-κB (NF-κB) signalling pathway participates in a multitude of biological processes, which imply the requirement of a complex and precise regulation. IκB (for Inhibitor of kappaB) proteins, which bind and retain NF-κB dimers in the cytoplasm, are the main contributors to negative regulation of NF-κB under non-stimulation conditions. Nevertheless, increasing evidences indicate that IκB proteins exert specific nuclear roles that directly contribute to the control of gene transcription. In particular, hypophosphorylated IκBβ can bind the promoter region of TNFα leading to persistent gene transcription in macrophages and contributing to the regulation of the inflammatory response. Recently, we demonstrated that phosphorylated and SUMOylated IκBα reside in the nucleus of the cells where it binds to chromatin leading to specific transcriptional repression. Mechanistically, IκBα associates and regulates Polycomb Repressor Complex activity, a function that is evolutionary conserved from flies to mammals, as indicate the homeotic phenotype of Drosophila mutants. Here we discuss the implications of chromatin-bound IκBα function in the context of tumorigenesis.
Similar content being viewed by others
Main
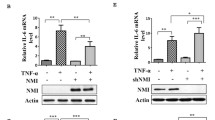
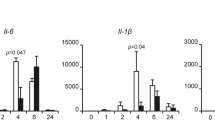
The nuclear factor-κB (NF-κB) family of transcription factors has an important role in the regulation of biological processes such as immunity and inflammation, apoptosis, stress response or ageing (Hayden and Ghosh, 2004). This family is composed of five members: RelA (p65), RelB and c-Rel, and the precursor proteins NF-κB1 (p105) and NF-κB2 (p100) that can be processed into p50 and p52, respectively. Nuclear factor-κB transcription factors function as homo- and hetero-dimers that activate or repress gene expression in a context-dependent manner. Under resting conditions, most NF-κB resides in the cytoplasm bound to particular inhibitory proteins of the inhibitor of kappaB (IκB) family, IκBα, IkBβ or IκBɛ, which generate an additional degree of complexity. Phosphorylation of IκB at specific serine residues by the IκB kinase (IKK) complex induces its ubiquitynation and subsequent proteasomal degradation, thus releasing the NF-κB factor leading to its nuclear translocation and gene transcription (Beg et al, 1992). However, recent studies have provided evidence that IκB proteins are more than NF-κB inhibitors. In 2010, Rao and colleagues demonstrated that following LPS stimulation, hypophosphorylated newly synthesised IκBβ binds p65 and c-Rel at the promoter region of specific genes such as TNFα and protects the NF-κB factor from IκBα association and chromatin release leading to persistent transcriptional activity (Rao et al, 2010). The same mechanism operates at the IL1-β promoter (Scheibel et al, 2010).
Some years ago, we found that in fibroblasts nuclear IκBα associates with NF-κB-independent gene promoters, such as hes1 and herp2, which are general regulators of cell differentiation, correlating with their transcriptional repression. TNFα treatment induced IKK recruitment to these particular genes leading to IκBα dissociation and transcriptional activation (Aguilera et al, 2004). Importantly, IKK was found constitutively bound to hes1 and herp2 promoters in colorectal cancer cells associated with increased gene expression (Fernandez-Majada et al, 2007a). Now, we have demonstrated that in keratinocytes there is a fraction of phosphorylated and SUMOylated IκBα (PS-IκBα) that binds to chromatin through direct association with histones leading to NF-κB-independent gene regulation (Mulero et al, 2013). Interestingly, nuclear PS-IκBα seems to be crucial for proper skin homeostasis, which might explain the severe skin phenotype of IκBα-deficient mice that die 5 days after birth because of a massive skin inflammation likely associated with a defective barrier function (Beg et al, 1995; Klement et al, 1996; Rebholz et al, 2007; Table 1). In addition, our results strongly suggest that aberrant accumulation of cytoplasmic IκBα in keratinocytes promotes squamous cell carcinoma (SCC; Mulero et al, 2013), which is in agreement with the oncogenic phenotype of ectopic IκBα-SR expression in the skin (van Hogerlinden et al, 1999, 2004).
I κ B α , an old cytoplasmic protein with a novel nuclear function, or vice-versa
IκBα regulates epidermal homeostasis
Mammalian epidermis consists of four different layers: basal, spinous, granular and cornified. The proliferating basal layer of the epidermis contains most of the stem and progenitor cells of this tissue, which then undergoes a differentiation process that culminates in their fully maturation and enucleation as they reach the surface layer, thus generating the cornified layer of keratinocytes. However, this is not an easy task for a tissue that is permanently exposed to a plethora of external insults such as UV radiation, extreme temperature variations and chemical exposure among others. The success of the skin differentiation process depends on the integration of intrinsic cellular programmes with external signals including the response of the immune system. Recently, we found that PS-IκBα is predominantly distributed in the nucleus of keratinocytes bound to the promoter of genes such as HOX and IRX (Mulero et al, 2013), which control skin differentiation and proliferation but are also involved in the developmental regulation. Once in the chromatin, PS-IκBα facilitates the recruitment of polycomb repressive complex 2 to gene promoters and dictates their competence to be induced following TNFα stimulation, thus establishing a mechanistic link between inflammatory signals and skin homeostasis. Remarkably, this role for IκBα as a regulator of polycomb function is operating during Drosophila development since mutations in Cactus (the IκBα orthologue in flies) enhanced the homeotic phenotype of Pc (Polycomb) mutations. These results together with the identification of IκBα orthologues in the worm Caenorhabditis elegans that does not have a real NF-κB pathway suggest that this newly identified function of PS-IκBα is an ancestral function previous to the specialisation of the immune system.
Nuclear IκBα as a tumour suppressor in the skin
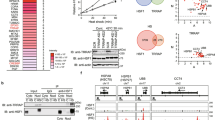
Indicative of a tumour-suppressor function for PS-IκBα, immunohistochemical analysis of a panel of human skin samples demonstrated that IκBα remains nuclear in the keratinocytes of benign skin lesions such as elastosis, psoriasis, actinic keratosis and Bowen disease, but is specifically lost in the more malignant lesions such as SCC. Further analysis of a cohort of 112 patients with urogenital SCC at different stages of tumour progression showed that in samples corresponding to invasive and metastatic SCC, IκBα was totally excluded from the nucleus but accumulated in the cytoplasm (Mulero et al, 2013), suggesting that beyond loss of nuclear IκBα, cytoplasmic IκBα (likely SUMOylated-IκBα) might have additional pro-tumorigenic functions, which will be investigated. This possibility is in agreement with the skin phenotype of different mice strains ectopically expressing the IκBα super-repressor mutant (IκBα-SR), which develop SCC-resembling tumours in the skin (Dajee et al, 2003; van Hogerlinden et al, 1999, 2004; Table 1), what contrast with the dogma that constitutive NF-κB activation results in pro-tumorigenic phenotypes that are prevented following NF-κB inhibition (Ben-Neriah and Karin, 2011). Similarly, Dajee et al (2003) demonstrated that mutant RASV12 failed to generate tumours when expressed in primary human keratinocytes (that were growth arrested), but it efficiently induced tumours resembling human SCC when co-expressed with IκBα-SR. Based on others and our results, we speculate that cytoplasmic accumulation of IκBα in skin tumours might sequester nuclear co-represors and HDACs (Aguilera et al, 2004; Fernandez-Majada et al, 2007a, 2007b; Hoberg et al, 2004), thus promoting a global miss-regualtion of gene transcription. In this sense, specific HDAC proteins contain SUMO-binding elements that could favour their binding to PS-IκBα either in the nucleus or the cytoplasm (model in Figure 1). Whether the cytoplasmic IκBα that is found in SCC samples is SUMOylated and/or phosphorylated remains to be addressed.
Schematic representation of PS-I κ B α structure depicting the six ankyrin repeats and the C-terminal PEST domain. SUMOylable lysines K21 and K22 are shown in green, whereas serines 32 and 36 (that are phosphorylated in PS-IκBα) are indicated in red. Nuclear IκBα is aberrantly localised in the cytoplasm of skin cancer cells. Thus, whereas in healthy skin, PS-IκBα binds chromatin at the promoter of HOX and IRX family of genes repressing their transcription, in SCC, IκBα is excluded from the nucleus and accumulates in the cytoplasm of the cells likely sequestering specific transcriptional repressors.
More recently, a knock-in mouse containing an IκBα protein with a mutated nuclear export sequence that is mostly localised in the nucleus of most cells has been characterised. These mutant mice show impaired canonical and alternative NF-κB pathways in the mature B cells with the absence of secondary lymphoid organs, but they display functional skin with no evidence of critical barrier defects (Wuerzberger-Davis et al, 2011). However, a detailed analysis of mutant skins from 7- to 8-week-old animals revealed significant differences with wild-type mice including increased proliferation index, expansion of the K14-positive basal layer, and reduction on the thickness of the intermediate K10-positive skin layer (Mulero et al, 2013). Future experiments should address the question of whether constitutively nuclear IκBα results in altered Hox and Irx expression in the basal or supra-basal layer of keratinocytes, and whether it protects these cells from undergoing tumorigenesis under specific transformation procedures or even with ageing.
IκB kinase alpha (IKKα) modulates IκBα localisation in keratinocytes
It is worth mentioning, that not only IκBα but also the IKKα displays a predominantly nuclear distribution in the skin (Marinari et al, 2008; Zhu et al, 2007). Loss of IKKα associates with altered proliferation and differentiation of epidermal keratinocytes (Hu et al, 1999; Sil et al, 2004), whereas mutations that resulted in IKKα loss or in the generation of truncated proteins that failed to interact with chromatin led to the development of skin papillomas and SCC (Liu et al, 2006; Park et al, 2007). Conversely, chemically induced skin carcinogenesis is reverted by overexpression of wild-type IKKα (Liu et al, 2006). Our recent results indicated that active IKKα induced a partial accumulation of SUMOylated-IκBα in the cytoplasmic compartment of keratinocytes, and IKK activation correlates with cytoplasmic accumulation of IκBα in human SCC samples (Mulero et al, 2013). These results open the possibility that IκBα miss-regulation might contribute to the phenotypes observed downstream of IKKα alterations, although it remains to be studied how IKKα regulates IκBα distribution. Because chromatin-bound IκBα is already phosphorylated in the residues known to be targets of canonical IKK activity, and we found that this phosphorylation is IKK independent, we speculate that nuclear IKKα might induce PS-IκBα chromatin release by regulating specific editing enzymes (i.e., phosphatase and desumoylase), scaffold proteins or proteins associated with the repressive PS-IκBα complex such as histones or members of the polycomb repressive complex 2, resulting in a reduced affinity of IκBα with chromatin.
Different IκBα populations include different post-translational modifications
Several publications previously established the link between SUMOylation and transcriptional repression by specific transcription factors such as CtBP, KAP1 or Sp3 (reviewed in Garcia-Dominguez and Reyes (2009)). SUMOylated-IκBα had previously been identified in different types of cells, however, no specific functions for this modified version of the molecule had been ascribed. In 1998, Desterro and colleagues detected the existence of endogenous SUMO-1-modified IκBα in a panel of cancer cell lines, being SUMO-1 linked to lysine 21 of IκBα. Because lysine 21 is involved in the signal-mediated degradation of IκBα (following poly-ubiquitination), SUMO1-IκBα was resistant to degradation and led to reduced NF-κB activity in these cells. Interestingly, SUMOylation was reduced in IκBα mutant proteins mimicking serine 32 and 36 phosphorylation that is a mark for degradation (Desterro et al, 1998). Accumulation of SUMO-1-bound IκBα is also detected downstream of adenosine signalling during hypoxia and reoxigenation (Liu et al, 2009), although its functional contribution in this system is unknown. Recently, it was shown that IκBα was modified by SUMO-2/3 but also by a combination of SUMO and ubiquitin chains (Culver et al, 2010) resulting in an IκBα molecule that can still be regulated by degradation following NF-κB activation (Aillet et al, 2012). The use of particular cell types, and the variations in the technical approaches (including the reagents used for stabilising SUMO chains and for detection) can explain the different results obtained by each group, however, it is still possible that several modifications coexist in particular IκBα subpopulations leading to functional specificities. Interestingly, SUMOylable proteins are characterised by the presence of a specific ΨKxE/D motif, which is the case of IκBα but not of other IκB orthologues such as IκBβ (Kracklauer and Schmidt, 2003). An extended consensus has also been reported, in which SUMOylation is imposed by negatively charged residues (including phosphorylated aminoacids) downstream of the ΨKxE/D motif (Hietakangas et al, 2003). Thus, one could speculate that IκBα SUMOylation is subsequent to its phosphorylation at serines 32 and/or 36 by specific kinase/s other than IKKβ (Mulero et al, 2013). Thus, it is of crucial importance to identify such kinases, investigate how they are regulated under specific conditions, different tissues or particular processes, and evaluate their contribution to IκBα SUMOylation and function. Finally, studying the contribution of SUMO modifications to IκBα function is not an easy job as mutants that fail to be SUMOylated (i.e., the K21-22 mutant) are also unable to be ubiquitinated and degraded, thus leading to strong NF-κB phenotypes. To circumvent this problem, we are currently generating SUMO-IκBα fusion proteins, which will be tested for their chromatin- or NF-κB-related activities.
The discovery that PS-IκBα exerts a nuclear function that affects NF-κB-independent transcription in response to NF-κB-related stimuli such as TNFα is also a temptation to re-examine previous results that might have misinterpreted because of the most prominent role of cytoplasmic IκBα. A putative involvement of PS-IκBα in other types of cancer should also be studied; mainly in those that are associated with aberrant or chronic IKK activity such is the case of inflammation-related ones.
On the other hand, it is possible that specific elements of the chromatin-bound IκBα complex are directly involved in IκBα SUMOylation. For example, Pc2, one of the mammalian orthologues of the Drosophila Polycomb (PRC1) that we found as functionally associated with IκBα/Cactus during Drosophila development, is a SUMO E3 ligase (Kagey et al, 2003). Moreover, it is not just IκBα but other chromatin components such as the histones and elements of the PRC complex including EZH2 and SUZ12 are capable to be SUMOylated, which might impact on their capacity to recruit transcriptional repressors at specific genomic regions, which will be addressed in a near future.
Future therapeutic perspectives based on IκBα
The importance of investigating the mechanisms involved in IκBα and PS-IκBα regulation and their functional relevance in tumour progression is evidenced by the fact that over 250 000 patients are diagnosed with SCC in the world each year. Squamous cell carcinoma is the second most common skin cancer that arises in any region of the body but more frequently in areas exposed to the UV light. Although infrequent (about 10% of the cases), the appearance of metastasis in SCC patients has a very bad prognosis because the absence of treatments, especially in aged patients. Moreover, to date there are no good biomarkers that predict metastatic SCC potential beyond the previous appearance of local relapse, tumour staging or the presence of MYC amplifications (Toll et al, 2009). We are currently investigating whether detection of cytoplasmic IκBα or PS-IκBα, or/and nuclear-active IKKα identifies the small fraction of SCC patients that will develop tumour relapse or metastasis. Those patients could be then selected for more aggressive therapies, but also as candidates for future personalised treatments based on IκBα/IKKα-based therapies.
Conclusions
Initially, IκB proteins were described as cytoplasmic regulators of the NF-κB pathway; however, several studies focused on IκBα and IκBβ demonstrated the existence of specific nuclear roles for both proteins. First, it was found that IκBα contains a functional nuclear localisation signal that is essential for the efficient post-activation termination of NF-κB signalling by facilitating chromatin release of p50-p65 dimers (Arenzana-Seisdedos et al, 1997). However, IκBα can be retained bound to the chromatin at specific promoter regions through its interactions with histones H2A and H4, thus playing an essential contribution to skin differentiation. We propose that PS-IκBα can exert both pro-oncogenic and tumour-suppressor functions, which need to be investigated in detail. In contrast, nuclear IκBβ is mainly associated with the regulation of the inflammatory response, through the maintenance of persistent cytokine expression in macrophages. The finding that this polycomb-associated IκBα function is conserved in the Drosophila development, together with and the identification of IκB homologues in worms lacking a truly NF-κB pathway suggests that this is an ancestral function that might exert a more general contribution to the cellular physiology. Finally, the identification of specific post-translational modifications that are restricted to chromatin-bound IκBα converts its modifying enzymes and PS-IκBα in attractive targets for novel therapeutic strategies specific for particular IκBα-associated pathologies and patients.
References
Aguilera C, Hoya-Arias R, Haegeman G, Espinosa L, Bigas A (2004) Recruitment of IkappaBalpha to the hes1 promoter is associated with transcriptional repression. Proc Natl Acad Sci USA 101 (47): 16537–16542.
Aillet F, Lopitz-Otsoa F, Egana I, Hjerpe R, Fraser P, Hay RT, Rodriguez MS, Lang V (2012) Heterologous SUMO-2/3-ubiquitin chains optimize IkappaBalpha degradation and NF-kappaB activity. PLoS One 7 (12): e51672.
Arenzana-Seisdedos F, Turpin P, Rodriguez M, Thomas D, Hay RT, Virelizier JL, Dargemont C (1997) Nuclear localization of I kappa B alpha promotes active transport of NF-kappa B from the nucleus to the cytoplasm. J Cell Sci 110 (Pt 3): 369–378.
Beg AA, Ruben SM, Scheinman RI, Haskill S, Rosen CA, Baldwin AS Jr (1992) I kappa B interacts with the nuclear localization sequences of the subunits of NF-kappa B: a mechanism for cytoplasmic retention. Genes Dev 6 (10): 1899–1913.
Beg AA, Sha WC, Bronson RT, Baltimore D (1995) Constitutive NF-kappa B activation, enhanced granulopoiesis, and neonatal lethality in I kappa B alpha-deficient mice. Genes Dev 9 (22): 2736–2746.
Ben-Neriah Y, Karin M (2011) Inflammation meets cancer, with NF-kappaB as the matchmaker. Nat Immunol 12 (8): 715–723.
Culver C, Sundqvist A, Mudie S, Melvin A, Xirodimas D, Rocha S (2010) Mechanism of hypoxia-induced NF-kappaB. Mol Cell Biol 30 (20): 4901–4921.
Dajee M, Lazarov M, Zhang JY, Cai T, Green CL, Russell AJ, Marinkovich MP, Tao S, Lin Q, Kubo Y, Khavari PA (2003) NF-kappaB blockade and oncogenic Ras trigger invasive human epidermal neoplasia. Nature 421 (6923): 639–643.
Desterro JM, Rodriguez MS, Hay RT (1998) SUMO-1 modification of IkappaBalpha inhibits NF-kappaB activation. Mol Cell 2 (2): 233–239.
Fernandez-Majada V, Aguilera C, Villanueva A, Vilardell F, Robert-Moreno A, Aytes A, Real FX, Capella G, Mayo MW, Espinosa L, Bigas A (2007a) Nuclear IKK activity leads to dysregulated notch-dependent gene expression in colorectal cancer. Proc Natl Acad Sci USA 104 (1): 276–281.
Fernandez-Majada V, Pujadas J, Vilardell F, Capella G, Mayo MW, Bigas A, Espinosa L (2007b) Aberrant cytoplasmic localization of N-CoR in colorectal tumors. Cell Cycle 6 (14): 1748–1752.
Garcia-Dominguez M, Reyes JC (2009) SUMO association with repressor complexes, emerging routes for transcriptional control. Biochim Biophys Acta 1789 (6-8): 451–459.
Hayden MS, Ghosh S (2004) Signaling to NF-kappaB. Genes Dev 18 (18): 2195–2224.
Hietakangas V, Ahlskog JK, Jakobsson AM, Hellesuo M, Sahlberg NM, Holmberg CI, Mikhailov A, Palvimo JJ, Pirkkala L, Sistonen L (2003) Phosphorylation of serine 303 is a prerequisite for the stress-inducible SUMO modification of heat shock factor 1. Mol Cell Biol 23 (8): 2953–2968.
Hoberg JE, Yeung F, Mayo MW (2004) SMRT derepression by the IkappaB kinase alpha: a prerequisite to NF-kappaB transcription and survival. Mol Cell 16 (2): 245–255.
Hu Y, Baud V, Delhase M, Zhang P, Deerinck T, Ellisman M, Johnson R, Karin M (1999) Abnormal morphogenesis but intact IKK activation in mice lacking the IKKalpha subunit of IkappaB kinase. Science 284 (5412): 316–320.
Kagey MH, Melhuish TA, Wotton D (2003) The polycomb protein Pc2 is a SUMO E3. Cell 113 (1): 127–137.
Klement JF, Rice NR, Car BD, Abbondanzo SJ, Powers GD, Bhatt PH, Chen CH, Rosen CA, Stewart CL (1996) IkappaBalpha deficiency results in a sustained NF-kappaB response and severe widespread dermatitis in mice. Mol Cell Biol 16 (5): 2341–2349.
Kracklauer MP, Schmidt C (2003) At the crossroads of SUMO and NF-kappaB. Mol Cancer 2: 39.
Liu B, Park E, Zhu F, Bustos T, Liu J, Shen J, Fischer SM, Hu Y (2006) A critical role for I kappaB kinase alpha in the development of human and mouse squamous cell carcinomas. Proc Natl Acad Sci USA 103 (46): 17202–17207.
Liu Q, Li J, Khoury J, Colgan SP, Ibla JC (2009) Adenosine signaling mediates SUMO-1 modification of IkappaBalpha during hypoxia and reoxygenation. J Biol Chem 284 (20): 13686–13695.
Marinari B, Moretti F, Botti E, Giustizieri ML, Descargues P, Giunta A, Stolfi C, Ballaro C, Papoutsaki M, Alema S, Monteleone G, Chimenti S, Karin M, Costanzo A (2008) The tumor suppressor activity of IKKalpha in stratified epithelia is exerted in part via the TGF-beta antiproliferative pathway. Proc Natl Acad Sci USA 105 (44): 17091–17096.
Mulero MC, Ferres-Marco D, Islam A, Margalef P, Pecoraro M, Toll A, Drechsel N, Charneco C, Davis S, Bellora N, Gallardo F, Lopez-Arribillaga E, Asensio-Juan E, Rodilla V, Gonzalez J, Iglesias M, Shih V, Mar Alba M, Di Croce L, Hoffmann A, Miyamoto S, Villa-Freixa J, Lopez-Bigas N, Keyes WM, Dominguez M, Bigas A, Espinosa L (2013) Chromatin-bound IkappaBalpha regulates a subset of polycomb target genes in differentiation and cancer. Cancer Cell 24 (2): 151–166.
Park E, Zhu F, Liu B, Xia X, Shen J, Bustos T, Fischer SM, Hu Y (2007) Reduction in IkappaB kinase alpha expression promotes the development of skin papillomas and carcinomas. Cancer Res 67 (19): 9158–9168.
Rao P, Hayden MS, Long M, Scott ML, West AP, Zhang D, Oeckinghaus A, Lynch C, Hoffmann A, Baltimore D, Ghosh S (2010) IkappaBbeta acts to inhibit and activate gene expression during the inflammatory response. Nature 466 (7310): 1115–1119.
Rebholz B, Haase I, Eckelt B, Paxian S, Flaig MJ, Ghoreschi K, Nedospasov SA, Mailhammer R, Debey-Pascher S, Schultze JL, Weindl G, Forster I, Huss R, Stratis A, Ruzicka T, Rocken M, Pfeffer K, Schmid RM, Rupec RA (2007) Crosstalk between keratinocytes and adaptive immune cells in an IkappaBalpha protein-mediated inflammatory disease of the skin. Immunity 27 (2): 296–307.
Scheibel M, Klein B, Merkle H, Schulz M, Fritsch R, Greten FR, Arkan MC, Schneider G, Schmid RM (2010) IkappaBbeta is an essential co-activator for LPS-induced IL-1beta transcription in vivo. J Exp Med 207 (12): 2621–2630.
Seitz CS, Lin Q, Deng H, Khavari PA (1998) Alterations in NF-kappaB function in transgenic epithelial tissue demonstrate a growth inhibitory role for NF-kappaB. Proc Natl Acad Sci USA 95 (5): 2307–2312.
Shih VF, Kearns JD, Basak S, Savinova OV, Ghosh G, Hoffmann A (2009) Kinetic control of negative feedback regulators of NF-kappaB/RelA determines their pathogen- and cytokine-receptor signaling specificity. Proc Natl Acad Sci USA 106 (24): 9619–9624.
Sil AK, Maeda S, Sano Y, Roop DR, Karin M (2004) IkappaB kinase-alpha acts in the epidermis to control skeletal and craniofacial morphogenesis. Nature 428 (6983): 660–664.
Toll A, Salgado R, Yebenes M, Martin-Ezquerra G, Gilaberte M, Baro T, Sole F, Alameda F, Espinet B, Pujol RM (2009) MYC gene numerical aberrations in actinic keratosis and cutaneous squamous cell carcinoma. Br J Dermatol 161 (5): 1112–1118.
van Hogerlinden M, Rozell BL, Ahrlund-Richter L, Toftgard R (1999) Squamous cell carcinomas and increased apoptosis in skin with inhibited Rel/nuclear factor-kappaB signaling. Cancer Res 59 (14): 3299–3303.
van Hogerlinden M, Rozell BL, Toftgard R, Sundberg JP (2004) Characterization of the progressive skin disease and inflammatory cell infiltrate in mice with inhibited NF-kappaB signaling. J Invest Dermatol 123 (1): 101–108.
Wuerzberger-Davis SM, Chen Y, Yang DT, Kearns JD, Bates PW, Lynch C, Ladell NC, Yu M, Podd A, Zeng H, Huang TT, Wen R, Hoffmann A, Wang D, Miyamoto S (2011) Nuclear export of the NF-kappaB inhibitor IkappaBalpha is required for proper B cell and secondary lymphoid tissue formation. Immunity 34 (2): 188–200.
Zhu F, Xia X, Liu B, Shen J, Hu Y, Person M (2007) IKKalpha shields 14-3-3sigma, a G(2)/M cell cycle checkpoint gene, from hypermethylation, preventing its silencing. Mol Cell 27 (2): 214–227.
Acknowledgements
LE is an investigator at the Carlos III program. MM is an investigator of the Sara Borrell program (CD09/00421). This work was supported by Instituto de Salud Carlos III Grant PI13/00448 to LE, Red Temática de Investigación Cooperativa en Cáncer (RD12/0036/0054) and Agència de Gestió d’Ajuts Universitaris i de Recerca (2009SGR-23).
Author information
Authors and Affiliations
Corresponding author
PowerPoint slides
Rights and permissions
This work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Espinosa, L., Bigas, A. & Mulero, M. Novel functions of chromatin-bound IκBα in oncogenic transformation. Br J Cancer 111, 1688–1692 (2014). https://doi.org/10.1038/bjc.2014.84
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bjc.2014.84
- Springer Nature Limited