Abstract
Telomeres are DNA-protein structures that cap linear chromosomes and are essential for maintaining genomic stability and cell phenotype. We identified a novel human telomere-associated protein, TIN2, by interaction cloning using the telomeric DNA-binding-protein TRF1 as a bait. TIN2 interacted with TRF1 in vitro and in cells, and co-localized with TRF1 in nuclei and metaphase chromosomes. A mutant TIN2 that lacks amino-terminal sequences effects elongated human telomeres in a telomerase-dependent manner. Our findings suggest that TRF1 is insufficient for control of telomere length in human cells, and that TIN2 is an essential mediator of TRF1 function.







Similar content being viewed by others
Accession codes
References
Blackburn, E.H. Structure and function of telomeres. Nature 350, 569–573 (1991).
Aparicio, O.M., Billington, B.L. & Gottschling, D.E. Modifiers of position effect are shared between telomeric and silent mating-type loci in S. cerevisiae. Cell 66, 1279–1287 (1995).
Brachmann, C.B. et al. The SIR2 gene family, conserved from bacteria to humans, function in silencing, cell cycle progression and chromosome stability. Genes Dev. 9, 2888–2902 (1995).
Marchand, S., Buck, S.W., Moretti, P., Gilson, E. & Shore, D. Silencing of genes at nontelomeric sites in yeast is controlled by sequestration of silencing factors at telomeres by RAP1 protein. Genes Dev. 10, 1297–1309 (1996).
Campisi, J. The biology of replicative senescence. Eur. J. Cancer 33, 703–709 (1997).
Greider, C.W. Telomere length regulation. Annu. Rev. Biochem. 65, 337–365 (1996).
Lingner, J. & Cech, T.R. Telomerase and chromosome end maintenance. Curr. Opin. Genet. Dev. 8, 226– 232 (1998).
Nugent, C.I. & Lundblad, V. The telomerase reverse transcriptase: components and regulation. Genes Dev. 12, 1073–1085 (1998).
Harley, C.B., Futcher, A.B. & Greider, C.W. Telomeres shorten during aging of human fibroblasts. Nature 345, 458–460 (1990).
Shay, J.W. & Wright, W.E. Defining the molecular mechanisms of human cell immortalization. Biochim. Biophys. Acta 1071, 1–7 (1991).
Campisi, J., Dimri, G.P. & Hara, E. Control of replicative senescence. in Handbook of the Biology of Aging (eds Schneider, E. & Rowe, J.) 121 –149 (Academic, New York, 1996).
Bodnar, A.G. et al. Extension of life span by introduction of telomerase into normal human cells. Science 279, 349– 352 (1998).
Vaziri, H. & Benchimol, S. Reconstitution of telomerase activity in normal human cells leads to elongation of telomeres and extended replicative life span. Curr. Biol. 8, 279– 282 (1998).
Kiyono, T. et al. Both Rb/p16INK4a inactivation and telomerase activity are required to immortalize human epithelial cells. Nature 396, 84–88 (1998).
Bodnar, A.G., Kim, N.W., Effros, R.B. & Chiu, C.P. Mechanism of telomerase induction during T cell activation. Exp. Cell Res. 228, 58–64 (1996).
Buchkovich, K.J. & Greider, C.W. Telomerase regulation during entry into the cell cycle in normal human T cells. Mol. Biol. Cell 7, 1443–1454 (1996).
Kim, N.W. et al. Specific association of human telomerase activity with immortal cells and cancer. Science 266, 2011– 2015 (1994).
Bryan, T.M., Englezou, A., Dalla-Pozza, L., Dunham, M.A. & Reddel, R. Evidence for an alternative mechanism for maintaining telomere length in human tumors and tumor-derived cell lines. Nature Med. 3, 1271–1274 (1997).
Shore, D. Telomerase and telomere-binding proteins: controlling the end game. Trends Biochem. Sci. 22, 233–235 (1997).
Conrad, M.N., Wright, J.H., Wolf, A.J. & Zakian, V.A. RAP1 protein interacts with yeast telomeres in vivo: overproduction alters telomere structure and decreases chromosome stability. Cell 63, 739–750 (1990).
Kyrion, G., Boakye, K.A. & Lustig, A.J. C-terminal truncation of RAP1 results in the deregulation of telomere size, stability, and function in Saccharomyces cerevisiae. Mol. Cell. Biol. 12, 5159– 5173 (1992).
Wotton, D. & Shore, D. A novel RAP1p-interacting factor, RIF2p, cooperates with RIF1p to regulate telomere length in Saccharomyces cerevisiae. Genes Dev. 11, 748– 760 (1997).
Cockell, M. et al. The carboxy termini of SIR4 and RAP1 affect SIR3 localization: evidence for a multicomponent complex required for yeast telomeric silencing. J. Cell Biol. 129, 909– 924 (1995).
Grandin, N., Reed, S.I. & Charbonneau, M. STN1, a new Saccharomyces cerevisiae protein, is implicated in telomere size regulation in association with CDC13. Genes Dev. 11, 512–527 (1997).
Nugent, C.I., Hughes, T.R., Lue, N.F. & Lundblad, V. CDC13p: a single-strand telomeric DNA binding protein with a dual role in yeast telomere maintenance. Science 274, 249–252 (1996).
Chong, L. et al. A human telomeric protein. Science 270 , 1663–1667 (1995).
Shen, M., Haggblom, C., Vogt, M., Hunter, T. & Lu, K.P. Characterization and cell cycle regulation of the related telomeric proteins PIN2 and TRF1 suggest a role in mitosis. Proc. Natl Acad. Sci. USA 94, 13618–13623 (1997).
van Steensel, B. & de Lange, T. Control of telomere length by the human telomeric protein TRF1. Nature 385, 740–743 (1997).
Griffith, J., Bianchi, A. & de Lange, T. TRF1 promotes parallel pairing of telomeric tracts in vitro. J. Mol. Biol. 278, 79– 88 (1998).
van Steensel, B., Smogorzewska, A. & de Lange, T. TRF2 protects human telomeres from end to end fusions. Cell 92, 401–413 (1998).
Griffith, J.D. et al. Mammalian telomeres end in a large duplex loop. Cell 97, 503–514 (1999).
Smith, S., Giriat, I., Schmitt, A. & de Lange, T. Tankyrase, a poly (ADP-ribose) polymerase at human telomeres. Science 282, 1484–1487 (1998).
Chien, C.T., Bartel, P.L., Sternglanz, R. & Fields, S. The two-hybrid system: A method to identify and clone genes for proteins that interact with a protein of interest. Proc. Natl Acad. Sci USA 88, 9578–9582 (1991).
Weinrich, S.L. et al. Reconstitution of human telomerase with the template RNA component hTR and the catalytic protein subunit hTRT. Nature Genet. 17, 498–502 (1997).
Counter C.M. et al. Dissociation among in vitro telomerase activity, telomere maintenance, and cellular immortalization. Proc. Natl Acad. Sci. USA 95, 14723–14728 (1998).
Meyerson, M. et al. hEST2, the putative human telomerase catalytic subunit gene, is upregulated in tumor cells and during immortalization. Cell 90, 785–795 (1997).
Dimri, G.P., Testori, A., Acosta, M. & Campisi, J. Replicative senescence, aging and growth regulatory transcription factors. Biol. Signals 5, 154–162 (1996).
James, P., Halladay, J. & Craig, E.A. Genomic libraries and a host strain designed for highly efficient two-hybrid selection in yeast. Genetics 144 , 1425–1436 (1996).
Miller, A.D. & Rosman, G.J. Improved retroviral vectors for gene transfer and expression. Biotechniques 7, 980–988 (1989).
Morgenstern, J.P. & Land, H. Advanced mammalian gene transfer: high titre retroviral vectors with multiple drug selection markers and a complementary helper-free packaging cell line. Nucleic Acids Res. 18, 3587–3596 (1990).
Dimri, G.P., Hara, E. & Campisi, J. Regulation of two E2F-related genes in presenescent and senescent human fibroblasts. J. Biol. Chem. 269, 16180–16186 (1994).
Dimri G.P. et al. (1995). A novel biomarker identifies senescent human cells in culture and aging skin in vivo. Proc. Natl Acad. Sci. USA 92, 9363–9367 (1995).
Briand, P., Petersen, O.W. & van Deurs, B. A new diploid nontumorigenic human breast epithelial cell line isolated and propagated in a chemically defined medium. In Vitro Cell Dev. Biol. 23, 181–188 (1987).
Finer, M.H., Dull, T.J., Qin, L., Farson, D. & Roberts, M.R. kat: a high-efficiency retroviral transduction system for primary human T lymphocytes. Blood 83, 43–50 (1994).
Sambrook, J., Fritch, E.F. & Maniatis, T. Molecular Cloning (Cold Spring Harbor Press, New York, 1989).
Compton, D.A., Yen, T.J. & Cleveland, D.W. Identification of a novel centromere/kinetochore-associated protein using monoclonal antibodies generated against human mitotic chromosome scaffolds. J. Cell Biol. 112, 1083– 1097 (1991).
Zhong, Z., Shiue, L., Kaplan, S. & de Lange, T. A mammalian factor that binds telomeric TTAGGG repeats in vitro. Mol. Cell. Biol. 12, 4834–4843 (1992).
Acknowledgements
We thank P. James for yeast PJ69-4A and sharing strains before publication; R. Weinberg for hTERT cDNA; and M. Bissell and R. Lupu for HMT-3522 and MDA-453 cells. Supported by research (AG09909) and training (AG00266) grants from the National Institute on Aging, a fellowship (2F1B-0026) from the University of California Breast Cancer Research Program and research grant from the Ellison Medical Foundation, under contract DE-AC03-76SF00098 from the U.S. Department of Energy.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, Sh., Kaminker, P. & Campisi, J. TIN2, a new regulator of telomere length in human cells. Nat Genet 23, 405–412 (1999). https://doi.org/10.1038/70508
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/70508
- Springer Nature America, Inc.
This article is cited by
-
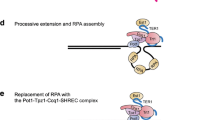
Shaping human telomeres: from shelterin and CST complexes to telomeric chromatin organization
Nature Reviews Molecular Cell Biology (2021)
-
Non-canonical roles of canonical telomere binding proteins in cancers
Cellular and Molecular Life Sciences (2021)
-
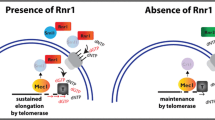
Regulation of human telomerase in homeostasis and disease
Nature Reviews Molecular Cell Biology (2020)
-
The role of telomere-binding modulators in pluripotent stem cells
Protein & Cell (2020)
-
Initiation of Pulmonary Fibrosis after Silica Inhalation in Rats is linked with Dysfunctional Shelterin Complex and DNA Damage Response
Scientific Reports (2019)