Abstract
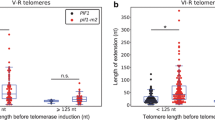
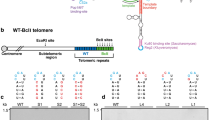
Telomeres, the nucleoprotein complexes at the end of eukaryotic chromosomes, protect them from degradation and ensure the replicative capacity of cells. In most human tumors and in budding yeast, telomere length is maintained by the activity of telomerase, an enzyme that adds dNTPs according to an internal RNA template. The dNTPs are generated with the help of the ribonucleotide reductase (RNR) complex. We have recently generated strains lacking the large subunit of RNR, Rnr1, which were kept viable by the expression of RNR complexes containing the Rnr1 homolog, Rnr3. Interestingly, we found that these Rnr1-deficient strains have short telomeres that are stably maintained, but cannot become efficiently elongated by telomerase. Thus, a basic maintenance of short telomeres is possible under conditions, where Rnr1 activity is absent, but a sustained elongation of short telomeres fully depends on Rnr1 activity. We show that Rnr3 cannot compensate for this telomeric function of Rnr1 even when overall cellular dNTP values are restored. This suggests that Rnr1 plays a role in telomere elongation beyond increasing cellular dNTP levels. Furthermore, our data indicate that telomerase may act in two different modes, one that is capable of coping with the “end-replication problem” and is functional even in the absence of Rnr1 and another required for the sustained elongation of short telomeres, which fully depends on the presence of Rnr1. Supply of dNTPs for telomere elongation is provided by the Mec1ATR checkpoint, both during regular DNA replication and upon replication fork stalling. We discuss the implications of these results on telomere maintenance in yeast and cancer cells.

Similar content being viewed by others
References
Adamczyk J, Deregowska A, Panek A, Golec E, Lewinska A, Wnuk M (2016) Affected chromosome homeostasis and genomic instability of clonal yeast cultures. Curr Genet 62:405–418. https://doi.org/10.1007/s00294-015-0537-3
Blackburn EH, Chan S, Chang J, Fulton TB, Krauskopf A, McEachern M, Prescott J, Roy J, Smith C, Wang H (2000) Molecular manifestations and molecular determinants of telomere capping. Cold Spring Harb Symp Quant Biol 65:253–263 doi
Boule JB, Vega LR, Zakian VA (2005) The yeast Pif1p helicase removes telomerase from telomeric DNA. Nature 438:57–61. https://doi.org/10.1038/nature04091
Evans SK, Lundblad V (1999) Est1 and Cdc13 as comediators of telomerase access. Science 286:117–120 doi
Forsyth NR, Wright WE, Shay JW (2002) Telomerase and differentiation in multicellular organisms: turn it off, turn it on, and turn it off again. Differentiation 69:188–197. https://doi.org/10.1046/j.1432-0436.2002.690412.x
Gadaleta MC, Gonzalez-Medina A, Noguchi E (2016) Timeless protection of telomeres. Curr Genet 62:725–730. https://doi.org/10.1007/s00294-016-0599-x
Grandin N, Bailly A, Charbonneau M (2005) Activation of Mrc1, a mediator of the replication checkpoint, by telomere erosion. Biology of the cell / under the auspices of the European Cell Biology Organization 97:799–814. https://doi.org/10.1042/BC20040526
Greider CW, Blackburn EH (1989) A telomeric sequence in the RNA of Tetrahymena telomerase required for telomere repeat synthesis. Nature 337:331–337 doi
Gupta A, Sharma S, Reichenbach P, Marjavaara L, Nilsson AK, Lingner J, Chabes A, Rothstein R, Chang M (2013) Telomere length homeostasis responds to changes in intracellular dNTP pools. Genetics 193:1095–1105. https://doi.org/10.1534/genetics.112.149120
Harley CB, Kim NW, Prowse KR, Weinrich SL, Hirsch KS, West MD, Bacchetti S, Hirte HW, Counter CM, Greider CW et al (1994) Telomerase, cell immortality, and cancer. Cold Spring Harb Symp Quant Biol 59:307–315 doi
Ho YH, Gasch AP (2015) Exploiting the yeast stress-activated signaling network to inform on stress biology and disease signaling. Curr Genet 61:503–511. https://doi.org/10.1007/s00294-015-0491-0
Huang M, Zhou Z, Elledge SJ (1998) The DNA replication and damage checkpoint pathways induce transcription by inhibition of the Crt1 repressor. Cell 94:595–605 doi
Kupiec M (2014) Biology of telomeres: lessons from budding yeast. FEMS Microbiol Rev 38:144–171 doi
Maicher A, Gazy I, Sharma S, Marjavaara L, Grinberg G, Shemesh K, Chabes A, Kupiec M (2017) Rnr1, but not Rnr3, facilitates the sustained telomerase-dependent elongation of telomeres. PLoS Genet 13:e1007082. https://doi.org/10.1371/journal.pgen.1007082
Makovets S, Blackburn EH (2009) DNA damage signalling prevents deleterious telomere addition at DNA breaks. Nat Cell Biol 11:1383–1386. https://doi.org/10.1038/ncb1985
Millet C, Makovets S (2016) Aneuploidy as a mechanism of adaptation to telomerase insufficiency. Curr Genet 62:557–564. https://doi.org/10.1007/s00294-015-0559-x
Niida H, Katsuno Y, Sengoku M, Shimada M, Yukawa M, Ikura M, Ikura T, Kohno K, Shima H, Suzuki H, Tashiro S, Nakanishi M (2010) Essential role of Tip60-dependent recruitment of ribonucleotide reductase at DNA damage sites in DNA repair during G1 phase. Genes Dev 24:333–338. https://doi.org/10.1101/gad.1863810
Nordlund P, Reichard P (2006) Ribonucleotide reductases. Annu Rev Biochem 75:681–706. https://doi.org/10.1146/annurev.biochem.75.103004.142443
Oldenburg DJ, Bendich AJ (2016) The linear plastid chromosomes of maize: terminal sequences, structures, and implications for DNA replication. Curr Genet 62:431–442. https://doi.org/10.1007/s00294-015-0548-0
Rubinstein L, Ungar L, Harari Y, Babin V, Ben-Aroya S, Merenyi G, Marjavaara L, Chabes A, Kupiec M (2014) Telomere length kinetics assay (TELKA) sorts the telomere length maintenance (tlm) mutants into functional groups. Nucleic Acids Res 42:6314–6325. https://doi.org/10.1093/nar/gku267
Smolikov S, Mazor Y, Krauskopf A (2004) ELG1, a regulator of genome stability, has a role in telomere length regulation and in silencing. In: Proceedings of the National Academy of Sciences of the United States of America 101: 1656–1661 https://doi.org/10.1073/pnas.0307796100
Stinus S, Paeschke K, Chang M (2017) Telomerase regulation by the Pif1 helicase: a length-dependent effect? Curr Genet. https://doi.org/10.1007/s00294-017-0768-6
Teixeira MT (2013) Saccharomyces cerevisiae as a Model to Study Replicative Senescence Triggered by Telomere Shortening. Front Oncol 3:101. https://doi.org/10.3389/fonc.2013.00101
Tkach JM, Yimit A, Lee AY, Riffle M, Costanzo M, Jaschob D, Hendry JA, Ou J, Moffat J, Boone C, Davis TN, Nislow C, Brown GW (2012) Dissecting DNA damage response pathways by analysing protein localization and abundance changes during DNA replication stress. Nat Cell Biol 14:966–976. https://doi.org/10.1038/ncb2549
Tong AS, Stern JL, Sfeir A, Kartawinata M, de Lange T, Zhu XD, Bryan TM (2015) ATM and ATR signaling regulate the recruitment of human telomerase to telomeres. Cell reports 13:1633–1646. https://doi.org/10.1016/j.celrep.2015.10.041
Yao R, Zhang Z, An X, Bucci B, Perlstein DL, Stubbe J, Huang M (2003) Subcellular localization of yeast ribonucleotide reductase regulated by the DNA replication and damage checkpoint pathways. In: Proceedings of the National Academy of Sciences of the United States of America 100: 6628–6633 https://doi.org/10.1073/pnas.1131932100
Zhao X, Muller EG, Rothstein R (1998) A suppressor of two essential checkpoint genes identifies a novel protein that negatively affects dNTP pools. Mol Cell 2:329–340 doi
Zhao X, Chabes A, Domkin V, Thelander L, Rothstein R (2001) The ribonucleotide reductase inhibitor Sml1 is a new target of the Mec1/Rad53 kinase cascade during growth and in response to DNA damage. EMBO J 20:3544–3553. https://doi.org/10.1093/emboj/20.13.3544
Acknowledgements
We thank all the Kupiec lab members, present and past, for support, ideas and encouragement. This work was supported by grants to MK from the Israel Science Foundation, the Israel Cancer Research Fund and the Minerva Center for in-lab evolution.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by M. Kupiec.
Rights and permissions
About this article
Cite this article
Maicher, A., Kupiec, M. Rnr1’s role in telomere elongation cannot be replaced by Rnr3: a role beyond dNTPs?. Curr Genet 64, 547–550 (2018). https://doi.org/10.1007/s00294-017-0779-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00294-017-0779-3