Abstract
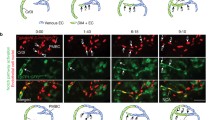
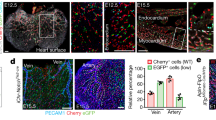
Arteries and veins are morphologically, functionally and molecularly very different, but how this distinction is established during vasculogenesis is unknown1,2. Here we show, by lineage tracking in zebrafish embryos, that angioblast precursors for the trunk artery and vein are spatially mixed in the lateral posterior mesoderm. Progeny of each angioblast, however, are restricted to one of the vessels. This arterial–venous decision is guided by gridlock (grl), an artery-restricted gene that is expressed in the lateral posterior mesoderm3. Graded reduction of grl expression, by mutation or morpholino antisense, progressively ablates regions of the artery, and expands contiguous regions of the vein, preceded by an increase in expression of the venous marker EphB4 receptor (ephb4)2 and diminution of expression of the arterial marker ephrin-B2 (efnb2)2. grl is downstream of notch4, and interference with notch signalling, by blocking Su(H)4, similarly reduces the artery and increases the vein. Thus, a notch–grl pathway controls assembly of the first embryonic artery, apparently by adjudicating an arterial versus venous cell fate decision.





Similar content being viewed by others
References
Carmeliet, P. Mechanisms of angiogenesis and arteriogenesis. Nature Med. 6, 389–395 (2000).
Yancopoulos, G. D., Klagsbrun, M. & Folkman, J. Vasculogenesis, angiogenesis, and growth factors: ephrins enter the fray at the border. Cell 93, 661–664 (1998).
Zhong, T. P., Rosenberg, M., Mohideen, M. A., Weinstein, B. & Fishman, M. C. Gridlock, an HLH gene required for assembly of the aorta in zebrafish. Science 287, 1820–1824 (2000).
Artavanis-Tsakonas, S., Rand, M. D. & Lake, R. J. Notch signaling: cell fate control and signal integration in development. Science 284, 770–776 (1999).
Thompson, M. A. et al. The cloche and spadetail genes differentially affect hematopoiesis and vasculogenesis. Dev. Biol. 197, 248–269 (1998).
Fouquet, B., Weinstein, B. M., Serluca, F. C. & Fishman, M. C. Vessel patterning in the embryo of the zebrafish: guidance by notochord. Dev. Biol. 183, 37–48 (1997).
Cleaver, O. & Krieg, P. A. VEGF mediates angioblast migration during development of the dorsal aorta in Xenopus. Development 125, 3905–3914 (1998).
Pardanaud, L. & Dieterlen-Lievre, F. Manipulation of the angiopoietic/hemangiopoietic commitment in the avian embryo. Development 126, 617–627 (1999).
Serbedzija, G. N., Chen, J. N. & Fishman, M. C. Regulation in the heart field of zebrafish. Development 125, 1095–1101 (1998).
Noden, D. M. Embryonic origins and assembly of blood vessels. Am. Rev. Respir. Dis. 140, 1097–1103 (1989).
Weinstein, B. M., Stemple, D. L., Driever, W. & Fishman, M. C. Gridlock, a localized heritable vascular patterning defect in the zebrafish. Nature Med. 1, 1143–1147 (1995).
Nasevicius, A. & Ekker, S. C. Effective targeted gene ‘knockdown’ in zebrafish. Nature Genet. 26, 216–220 (2000).
Nakagawa, O., Nakagawa, M., Richardson, J. A., Olson, E. N. & Srivastava, D. HRT1, HRT2, and HRT3: a new subclass of bHLH transcription factors marking specific cardiac, somitic, and pharyngeal arch segments. Dev. Biol. 216, 72–84 (1999).
Leimeister, C., Externbrink, A., Klamt, B. & Gessler, M. Hey genes: a novel subfamily of hairy- and Enhancer of split related genes specifically expressed during mouse embryogenesis. Mech. Dev. 85, 173–177 (1999).
Kokubo, H., Lun, Y. & Johnson, R. L. Identification and expression of a novel family of bHLH cDNAs related to Drosophila hairy and enhancer of split. Biochem. Biophys. Res. Commun. 260, 459–465 (1999).
Chin, M. T. et al. Cardiovascular basic helix loop helix factor 1, a novel transcriptional repressor expressed preferentially in the developing and adult cardiovascular system. J. Biol. Chem. 275, 6381–6387 (2000).
Fisher, A. L., Ohsako, S. & Caudy, M. The WRPW motif of the hairy-related basic helix-loop-helix repressor proteins acts as a 4-amino-acid transcription repression and protein–protein interaction domain. Mol. Cell Biol. 16, 2670–2677 (1996).
Fisher, A. & Caudy, M. The function of hairy-related bHLH repressor proteins in cell fate decisions. Bioessays 20, 298–306 (1998).
Nakagawa, O. et al. Members of the HRT family of basic helix-loop-helix proteins act as transcriptional repressors downstream of Notch signaling. Proc. Natl Acad. Sci. USA 97, 13655–13660 (2000).
Bierkamp, C. & Campos-Ortega, J. A. A zebrafish homologue of the Drosophila neurogenic gene Notch and its pattern of transcription during early embryogenesis. Mech. Dev. 43, 87–100 (1993).
Shirayoshi, Y. et al. Proto-oncogene of int-3, a mouse Notch homologue, is expressed in endothelial cells during early embryogenesis. Genes Cells 2, 213–224 (1997).
Bailey, A. M. & Posakony, J. W. Suppressor of hairless directly activates transcription of enhancer of split complex genes in response to Notch receptor activity. Genes Dev. 9, 2609–2622 (1995).
Wettstein, D. A., Turner, D. L. & Kintner, C. The Xenopus homolog of Drosophila Suppressor of Hairless mediates Notch signaling during primary neurogenesis. Development 124, 693–702 (1997).
Holder, N. & Klein, R. Eph receptors and epherins: effectors of morphogenesis. Development 126, 2033–2044 (1999).
Wang, H. U., Chen, Z. F. & Anderson, D. J. Molecular distinction and angiogenic interaction between embryonic arteries and veins revealed by ephrin-B2 and its receptor Eph-B4. Cell 93, 741–753 (1998).
Chan, J. et al. Morphogenesis of prechordal plate and notochord requires intact Eph/ephrin B signaling. Dev. Biol. 234, 470–482 (2001).
Krebs, L. T. et al. Notch signaling is essential for vascular morphogenesis in mice. Genes Dev. 14, 1343–1352 (2000).
Jensen, J. et al. Control of endodermal endocrine development by Hes-1. Nature Genet. 24, 36–44 (2000).
Sestan, N., Artavanis-Tsakonas, S. & Rakic, P. Contact-dependent inhibition of cortical neurite growth mediated by notch signaling. Science 286, 741–746 (1999).
Furukawa, T. et al. Rax, Hes1, and Notch1 promote the formation of Muller glia by postnatal retinal progenitor cells. Neuron 26, 383–394 (2000).
Acknowledgements
We thank members of the Fishman laboratory for advice during these experiments, especially J.-N. Chen for help with the preliminary work on lineage analysis and C. Simpson for histological analyses. We thank S. Artavanis-Tsakonas and M. Rand for human Notch1-ICD construct; C. Kintner for Xenopus notch-ICD and Su(H)-Dbm constructs; and J. Chen for zebrafish ephrin-B2 and EphB4 probes. J.P.L. was supported by the Sarnoff Foundation. This work is supported by grants from the National Institutes of Health and a sponsored research agreement with Genentech to M.C.F.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhong, T., Childs, S., Leu, J. et al. Gridlock signalling pathway fashions the first embryonic artery. Nature 414, 216–220 (2001). https://doi.org/10.1038/35102599
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/35102599
- Springer Nature Limited
This article is cited by
-
Single-cell transcriptomic analysis of vascular endothelial cells in zebrafish embryos
Scientific Reports (2022)
-
Molecular and genetic mechanisms in brain arteriovenous malformations: new insights and future perspectives
Neurosurgical Review (2022)
-
The roles and activation of endocardial Notch signaling in heart regeneration
Cell Regeneration (2021)
-
Atypical chemokine receptor ACKR3/CXCR7 controls postnatal vasculogenesis and arterial specification by mesenchymal stem cells via Notch signaling
Cell Death & Disease (2020)
-
Single-cell transcriptomic analysis identifies the conversion of zebrafish Etv2-deficient vascular progenitors into skeletal muscle
Nature Communications (2020)