Abstract
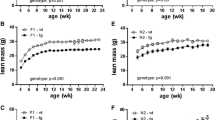
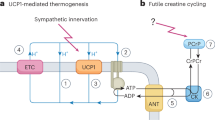
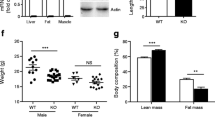
Uncoupling protein-3 (UCP-3) is a recently identified member of the mitochondrial transporter superfamily1,2 that is expressed predominantly in skeletal muscle1,2. However, its close relative UCP-1 is expressed exclusively in brown adipose tissue, a tissue whose main function is fat combustion and thermogenesis. Studies on the expression of UCP-3 in animals and humans in different physiological situations support a role for UCP-3 in energy balance and lipid metabolism3,4. However, direct evidence for these roles is lacking. Here we describe the creation of transgenic mice that overexpress human UCP-3 in skeletal muscle. These mice are hyperphagic but weigh less than their wild-type littermates. Magnetic resonance imaging shows a striking reduction in adipose tissue mass. The mice also exhibit lower fasting plasma glucose and insulin levels and an increased glucose clearance rate. This provides evidence that skeletal muscle UCP-3 has the potential to influence metabolic rate and glucose homeostasis in the whole animal.



Similar content being viewed by others
References
Boss, O. et al. Uncoupling protein-3, a new member of the mitochondrial carrier family with tissue-specific expression. FEBS Lett. 408, 39–42 (1999).
Vidal-Puig, A., Solanes, G., Grujic, D., Flier, J. S. & Lowell, B. B. UCP3—an uncoupling protein homolog expressed preferentially and abundantly in skeletal-muscle and brown adipose-tissue. Biochem. Biophys. Res. Commun. 235, 79– 82 (1997).
Samec, S., Seydoux, J., Dulloo, A. G. Role of UCP homologs in skeletal-muscles and brown adipose-tissue—mediators of thermogenesis or regulators of lipids as fuel substrate. FASEB J. 12, 715–724 ( 1998).
Samec, S., Seydoux, J., Dulloo, A. G. Post-starvation gene expression of skeletal muscle uncoupling protein 2 and uncoupling protein 3 in response to dietary fat levels and fatty acid composition—A link with insulin resistance. Diabetes 48, 436–441 ( 1999).
Brennan, K. J. & Hardeman, E. C. Quantitative analysis of the human alpha-skeletal actin gene in transgenic mice. J. Biol. Chem. 268, 719–725 ( 1993).
Grunning, P. et al. Differential patterns of transcript accumulation during human myogenesis. Mol. Cell. Biol. 7, 4100– 4114 (1987).
Widdowson, P. S., Upton, R., Buckingham, R., Arch, J. & Williams, G. Inhibition of food response to intracerebroventricular injection of leptin is attenuated in rats with diet-induced obesity. Diabetes 46, 1782– 1785 (1997).
Bellisle, F., Louis-Sylvestre, J., Demozay, F., Blazy, D. & Le Magnen, J. Cephalic phase of insulin secretion and food stimulation in humans, a new perspective. Am. J. Physiol. 249, E639–E645 ( 1985).
Leblanc, J. & Brondel, L. Role of palatability on meal-induced thermogenesis in human subjects. Am. J. Physiol. 248 , E333–E336 (1985).
Diamond, P., Brodel, L. & LeBlanc, J. Palatability and postprandial thermogenesis in dogs. Am. J. Physiol. 248, E75– E79 (1985).
Arch, J. R. et al. Treatment of obesity with thermogenic beta-adrenoceptor agonists, studies on BRL 26830A in rodents. Int. J. Obes. 8 (suppl. 1), 1–11 (1984).
Gordon, C. J. Temperature Regulation in Laboratory Rodents (Cambridge Univ. Press, 1993).
Stock, M. J. Gluttony and thermogenesis revisited. Int. J. Obes. 23, 1105–1117 (1999).
Largis, E. E., Burns, M. G., Muenkel, H. A., Dolan, J. A. & Claus, T. H. Antidiabetic and antiobesity effects of a highly selective β3-adrenoceptor agonist (CL 316,243). Drug Dev. Res. 32, 69–76 (1994).
Weyer, C., Tataranni, A., Snitker, S., Danforth, E. & Ravussin, E. Increase in insulin action and fat oxidation after treatment with CL 316,243, a highly selective beta3-adrenoceptor agonist in humans. Diabetes 47, 1555– 1561 (1998).
Liu, X., Perusse, F. & Bukoweicki, L. J. Mechanisms of the antidiabetic effects of the beta 3-adrenergic agonist CL-316243 in obese Zucker-ZDF rats. Am. J. Physiol. 274, R1212–R1219 (1998).
Cadenas, S. et al. UCP2 and UCP3 rise in starved rat skeletal muscle but mitochondrial proton conductance is unchanged. FEBS Lett. 462, 257–260 (1999).
Argyropoulos, G. et al. Effects of mutations in the human uncoupling protein 3 gene on the respiratory quotient and fat oxidation in severe obesity and type 2 diabetes. J. Clin. Invest. 102, 1345– 1351 (1998).
Otabe, S. et al. A genetic variation in the 5′ flanking region of the UCP3 gene is associated with body mass index in humans in interaction with physical activity. Diabetologia 43, 245– 249 (2000).
Chung, W. K. et al. the long isoform uncoupling protein-3 (UCP3L) in human energy homeostasis. Int. J. Obes. 23, S49– S50 (1999).
Hogan, B., Lacy, E., Beddington, R. & Constantini, F. Manipulating the Mouse Embryo 2nd edn (Cold Spring Harbor Press, 1994).
Holland, P. M., Abramson, R. D., Watson, R. & Gelfand, D. H. Detection of specific polymerase chain reaction product by utilizing the 5′-3′exonuclease activity of Thermus aquaticus DNA polymerase. Proc. Natl Acad. Sci. USA 88, 7276–7280 (1991).
Livak, K. J. Flood, S. J., Marmaro, J., Giusti, W. & Deetz, K. Oligonucleotides with fluorescent dyes at opposite ends provide a quenched probe system useful for detecting PCR product and nucleic acid hybridization. PCR Methods Appl. 4, 357–362 (1995).
Ernster, L. & Nordenbrand, K. Skeletal muscle mitochondria. Methods Enzymol. 10, 86– 94 (1967).
Young, P. W. et al. Repeat treatment of obese mice with BRL 49653, a new potent insulin sensitizer, enhances insulin action in white adipocytes. Association with increased insulin binding and cell-surface GLUT4 as measured by photoaffinity labeling. Diabetes 44, 1087– 1092 (1995).
Rogers, D. C. et al. Use of SHIRPA and discrimainant analysis to characterise marked differences in the behavioural phenotype of six inbred mouse strains. Behav. Brain. Res. 105, 207–217 (1999).
Jones, B. J. & Roberts, D. J. The quantitative measurement of motor inco-ordination in naive mice using an accelerating rotarod. J. Pharm. Pharmacol. 20, 302–304 (1968).
Acknowledgements
S.C. was supported by a Marie Curie (EC) fellowship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Clapham, J., Arch, J., Chapman, H. et al. Mice overexpressing human uncoupling protein-3 in skeletal muscle are hyperphagic and lean. Nature 406, 415–418 (2000). https://doi.org/10.1038/35019082
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/35019082
- Springer Nature Limited
This article is cited by
-
A transdermal treatment with MC903 ameliorates diet-induced obesity by reducing visceral fat and increasing myofiber thickness and energy consumption in mice
Nutrition & Metabolism (2023)
-
CD47 antisense oligonucleotide treatment attenuates obesity and its-associated metabolic dysfunction
Scientific Reports (2023)
-
Early high-fat feeding improves histone modifications of skeletal muscle at middle-age in mice
Laboratory Animal Research (2020)
-
Ccr7 null mice are protected against diet-induced obesity via Ucp1 upregulation and enhanced energy expenditure
Nutrition & Metabolism (2019)
-
Cytosolic lipid excess-induced mitochondrial dysfunction is the cause or effect of high fat diet-induced skeletal muscle insulin resistance: a molecular insight
Molecular Biology Reports (2019)