Abstract
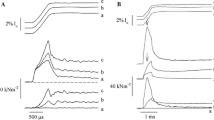
Muscle force is generated by cycling cross-bridges between actin and myosin filaments. In smooth muscle, cyclic attachment and detachment of cross-bridges is thought to be induced by a Ca2+- and calmodulin-dependent myosin light chain kinase which phosphorylates myosin. The relaxation that occurs after Ca2+ removal is usually ascribed to dephosphorylation of myosin by a phosphatase as non-phosphorylated myosin is unable to form force-generating criss-bridges1. Recently, Dillon et al.2 claimed, however, that dephosphorylation of attached cross-bridges may impede cross-bridge detachment, thus forming so-called ‘latch bridges’. Here we present evidence that after a Ca2+- and calmodulin-induced contraction of chemically skinned guinea pig Taenia coli3, the rapid removal of Ca2+ impedes the detachment of the myosin cross-bridges from the actin filament; force can then be maintained without energy consumption. The extremely slowly detaching cross-bridges which maintain the force after Ca2+ removal may indeed correspond to the ‘latch bridges’ mentioned above.
Similar content being viewed by others
References
Adelstein, R. S. & Eisenberg, E. A. Rev. Biochem. 49, 921–956 (1980).
Dillon, P. F., Aksoy, M. O., Driska, S. P. & Murphy, R. A. Science 211, 495–497 (1981).
Sparrow, M. P., Mrwa, U., Hofmann, F. & Rüegg, J. C. FEBS Lett. 125, 141–145 (1981).
Moisescu, D. G. Nature 262, 610–613 (1976).
Murphy, R. A., Driska, S. P. & Cohen, D. M. in Excitation–Contraction Coupling in Smooth Muscle (eds Casteels, R., Godfraind, T. & Rüegg, J. C.) 417–424 (Elsevier, Amsterdam, 1977).
Stein, L. A., Schwarz, R. P. Jr, Chock, P. B. & Eisenberg, E. Biochemistry 18, 3895–3909 (1979).
Schneider, M., Sparrow, M. & Rüegg, J. C. Experientia 37, 980–982 (1981).
Rüegg, J. C. Physiol. Rev. 51, 201–248 (1971).
Siegman, M. J., Butler, T. M., Mooers, S. U. & Davies, R. E. J. gen. Physiol. 76, 609–629 (1980).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Güth, K., Junge, J. Low Ca2+ impedes cross-bridge detachment in chemically skinned Taenia coli. Nature 300, 775–776 (1982). https://doi.org/10.1038/300775a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/300775a0
- Springer Nature Limited
This article is cited by
-
Comparison of the effects of 2,3-butanedione monoxime on force production, myosin light chain phosphorylation and chemical energy usage in intact and permeabilized smooth and skeletal muscles
Journal of Muscle Research and Cell Motility (1994)
-
Influence of 2,3-butanedione monoxime on heart energy metabolism
Basic Research in Cardiology (1993)
-
Flash photolysis studies of relaxation and cross-bridge detachment: higher sensitivity of tonic than phasic smooth muscle to MgADP
Journal of Muscle Research and Cell Motility (1993)
-
Complex stiffness of smooth muscle cytoplasm in the presence of Ca-activated brevin
Journal of Muscle Research and Cell Motility (1991)
-
The action of brevin, an F-actin severing protein, on the mechanical properties and ATPase activity of skinned smooth muscle
Journal of Muscle Research and Cell Motility (1990)