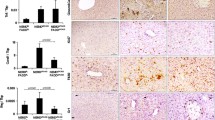
Abstract
The protein Bid is a participant in the pathway that leads to celldeath (apoptosis), mediating the release of cytochromec from mitochondria in response to signals from ‘death’ receptors known as TNFR1/Fas on the cell surface1,2,3,4,5,6,7. It is a member of the pro-apoptotic Bcl-2 family8 and is activated as a result of its cleavage by caspase 8, one of a family of proteolytic cell-death proteins. To investigate the role of Bid in vivo, we have generated mice deficient for Bid. We find that when these mice are injected with an antibody directed against Fas, they nearly all survive, whereas wild-type mice die from hepatocellular apoptosis and haemorrhagic necrosis. About half of the Bid-deficient animals had no apparent liver injury and showed no evidence of activation of the effector caspases 3 and 7, although the initiator caspase 8 had been activated. Other Bid-deficient mice survived with only moderate damage: all three caspases (8 and 37) were activated but their cell nuclei were intact and no mitochondrial cytochrome c was released. We also investigated the effects of Bid deficiency in cultured cells treated with anti-Fas antibody (hepatocytes and thymocytes) or with TNFα. (fibroblasts). In these Bid−/− cells, mitochondrial dysfunction was delayed, cytochrome c was not released, effector caspase activity was reduced and the cleavage of apoptosis substrates was altered. This loss-of-function model indicates that Bid is a critical substrate in vivo for signalling by death-receptor agonists, which mediates a mitochondrial amplification loop that is essential for the apoptosis of selected cells.





Similar content being viewed by others
References
Luo, X., Budihardjo, I., Zou, H., Slaughter, C. & Wang, X. Bid, a Bcl-2 interacting protein, mediates cytochrome c release from mitochondria in response to activation of cell surface death receptors. Cell 94, 481–490 (1998).
Li, H., Zhu, H., Xu, C. J. & Yuan, J. Cleavage of BID by caspase 8 mediates the mitochondrial damage in the Fas pathway of apoptosis. Cell 94, 491–501 (1998).
Gross, A. et al. Caspase-cleaved BID targets mitochondria and is required for cytochrome c release, while BCL-XL prevents this release but not tumor necrosis factor-R1/Fas death. J. Biol. Chem. 274, 1156–1163 (1999).
Han, Z., Bhalla, K., Pantazis, P., Hendrickson, E. A. & Wyche, J. H. Cif (cytochrome c efflux-inducing factor) activity is regulated by Bcl-2 and caspases and correlates with the activation of Bid. Mol. Cell. Biol. 19, 1381–1389 (1999).
Sun, X. M. et al. Distinct caspase cascades are initiated in receptor-mediated and chemical-induced apoptosis. J. Biol. Chem. 274, 5053–5060 (1999).
Chou, J., Li, H., Salvesen, G., Yuan, J. & Wagner, G. Solution structure of BID, an intracellular amplifier of apoptotic signaling. Cell 96, 615–624 (1999).
McDonnell, J., Fushman, D., Milliman, C., Korsmeyer, S. & Cowburn, D. Solution structure of the proapoptotic molecule BID: a structural basis for apoptotic agonist and antagonists. Cell 96, 625–634 (1999).
Wang, K., Yin, X. M., Chao, D. T., Milliman, C. L. & Korsmeyer, S. J. BID: a novel BH3 domain-only death agonist. Genes Dev. 10, 2859–2869 (1996).
Wang, K. et al. BID, a proapoptotic BCL-2 family member, is localized to mouse chromosome 6 and human chromosome 22q11. Genomics 53, 235–238 (1998).
Tartaglia, L. A., Ayres, T. M., Wong, G. H. W. & Goeddel, D. V. Anovel domain within the 55 kd TNF receptor signals cell death. Cell 74, 845–853 (1993).
Nagata, S. Apoptosis: telling cells their time is up. Curr. Biol. 6, 1241–1243 (1996).
Boldin, M. P., Goncharow, M., Goltsev, Y. V. & Wallach, D. Involvement of MACH, a novel MORT1/FADD-interacting protease, in Fas/APO-1- and TNF receptor-induced cell death. Cell 85, 803–815 (1996).
Muzio, M. et al. FLICE, a novel FADD-homologous ICE/CED-3-like protease, is recruited to the CD95 (Fas/API-1) death-inducing signaling complex. Cell 85, 817–827 (1996).
Ogasawara, J. et al. Lethal effect of the anti-Fas antibody in mice. Nature 364, 806–809 (1993).
Thornberry, N. A. et al. Acombinatorial approach defines specificities of members of the caspase family and granzyme B. Functional relationships established for key mediators of apoptosis. J. Biol. Chem. 272, 17907–17911 (1997).
Liu, X., Kim, C. N., Yang, J., Jemmerson, R. & Wang, X. Induction of apoptotic program in cell-free extracts: requirement for dATP and cytochrome c. Cell 86, 147–157 (1996).
Jemmerson, R. et al. Aconformational change in cytochrome c of apoptotic and necrotic cells is detected by monoclonal antibody binding and mimicked by association of the native antigen with synthetic phospholipid vesicles. Biochemistry 38, 3599–3609 (1999).
Scaffidi, C. et al. Two CD95 (APO-1/Fas) signaling pathways. EMBO J. 17, 1675–1687 (1998).
Xuesong, L., Zou, H., Slaughter, C. & Wang, X. DFF, a heterodimeric protein that functions downstream of caspase-3 to trigger DNA fragmentation during apoptosis. Cell 89, 175–184 (1997).
Enari, M. et al. Acaspase-activated DNase that degrades DNA during apoptosis and its inhibitor ICAD. Nature 391, 43–50 (1998).
Lacronique, V. et al. Bcl-2 protects from lethal hepatic apoptosis induced by an anti-Fas antibody in mice. Nature Med. 2, 80–86 (1996).
Rodriguez, I. et al. Abcl-2 transgene expressed in hepatocytes protects mice from fulminant liver destruction but not from rapid death induced by anti-Fas antibody injection. J. Exp. Med. 183, 1031–1036 (1996).
Li, P. et al. Cytochrome c and dATP-dependent formation of Apaf-1/caspase-9 complex initiates an apoptotic protease cascade. Cell 91, 479–489 (1997).
Varfolomeev, E. E. et al. Targeted disruption of the mouse caspase 8 gene ablates cell death induction by the TNF receptors, Fas/Apo1 and DR3 and is lethal prenatally. Immunity 9, 267–276 (1998).
Kuida, K. et al. Reduced apoptosis and cytochrome c-mediated caspase activation in mice lacking caspase 9. Cell 94, 325–337 (1998).
Hakem, R. et al. Differential requirement for caspase 9 in apoptotic pathways in vivo. Cell 94, 339–352 (1998).
Cecconi, F., Alvarezbolado, G., Meyer, B. I., Roth, K. A. & Gruss, P. Apaf1 (Ced-4 homolog) regulates programmed cell death in mammalian development. Cell 94, 727–737 (1998).
Yoshida, H. et al. Apaf1 is required for mitochondrial pathways of apoptosis and brain development. Cell 94, 739–750 (1998).
Adams, J. M. & Cory, S. The Bcl-2 family: arbiters of cell survival. Science 281, 1322–1326 (1998).
Srinivasan, A. et al. In situ immunodetection of activated caspase-3 in apoptotic neurons in the developing nervous system. Cell Death Differ. 5, 1004–1016 (1998).
Acknowledgements
We thank R. P. Sekaly for anti-casp-3 antibody, X. Wang for anti-DEF45 (ICAD) antibody, A. Srinivasan of IDUN Pharmaceuticals for anti-casp-8 and CM1 antibodies, C. Kanagawa for help with intravenous injections, and D. Maher for preparing the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yin, XM., Wang, K., Gross, A. et al. Bid-deficient mice are resistant to Fas-induced hepatocellular apoptosis. Nature 400, 886–891 (1999). https://doi.org/10.1038/23730
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/23730
- Springer Nature Limited
This article is cited by
-
mTOR inhibition suppresses Myc-driven polyposis by inducing immunogenic cell death
Oncogene (2023)
-
Some mice lacking intrinsic, as well as death receptor induced apoptosis and necroptosis, can survive to adulthood
Cell Death & Disease (2022)
-
Non-steroidal anti-inflammatory drugs induce immunogenic cell death in suppressing colorectal tumorigenesis
Oncogene (2021)
-
PUMA-mediated epithelial cell apoptosis promotes Helicobacter pylori infection-mediated gastritis
Cell Death & Disease (2020)
-
Caspase-1 initiates apoptosis in the absence of gasdermin D
Nature Communications (2019)