Abstract
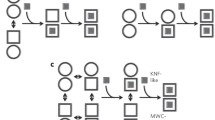
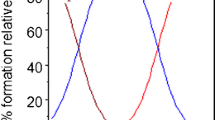
A RECENT theoretical investigation of the binding of a ligand to a polymerizing protein system1 extends the model of allosteric action based on isomerization2 to include reversible polymerization as a basis of these effects. We discuss here the possibility of defining uniquely an allosteric system from binding data in conjunction with other experimental evidence, and we consider results on the binding of succinate to aspartate transcarbamylase3 (EC 2.1.3.2) and of guanosinetriphosphate (GTP) to bovine liver glutamate dehydrogenase4 (EC 1.4.1.3).
Similar content being viewed by others
References
Nichol, L. W., Jackson, W. J. H., and Winzor, D. J., Biochemistry, 6, 2449 (1967).
Monod, J., Wyman, J., and Changeux, J-P., J. Mol. Biol., 12, 88 (1965).
Changeux, J-P., Gerhart, J. C., and Schachman, H. K., Biochemistry, 7, 531 (1968).
Frieden, C., and Colman, R. F., J. Biol. Chem., 242, 1705 (1967).
Klotz, I. M., Arch. Biochem., 9, 109 (1946).
Frieden, C., J. Biol. Chem., 242, 4045 (1967).
Changeux, J-P., and Rubin, M. M., Biochemistry, 7, 538 (1968).
Scatchard, G., Ann. NY Acad. Sci., 51, 660 (1949).
Weber, K., Nature, 218, 1116 (1968).
Wiley, D. C., and Lipscomb, W. N., Nature, 218, 1119 (1968).
Eisenberg, H., and Tomkins, G. M., J. Mol. Biol., 31, 37 (1968).
Frieden, C., J. Biol. Chem., 234, 809 (1959).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
NICHOL, L., SMITH, G. & WINZOR, D. Curve-fitting of Allosteric Binding Data. Nature 222, 174–176 (1969). https://doi.org/10.1038/222174a0
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1038/222174a0
- Springer Nature Limited
This article is cited by
-
Cooperativity of ligand binding as a function of monomer-dimer equilibrium parameters and acceptor concentration
Journal of Protein Chemistry (1987)