Abstract
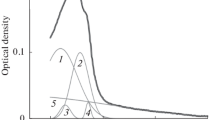
RECENT work by Weber1–3 has shown that the ultraviolet fluorescent spectra of globular proteins differ from those predicted for the summation of the independent contributions of their aromatic amino-acid constituents. Those examples investigated which contained tyrosine, but no tryptophan, showed a tyrosine emission spectrum, but with a reduced quantum yield. Those containing both tyrosine and tryptophan showed an emission spectrum predominantly resembling that of tryptophan alone, with an emission maximum in the range 320–350 mµ. The present communication will be concerned solely with proteins of the second category.
Similar content being viewed by others
References
Weber, G., Biochem. J., 75, 335 (1960).
Teale, F., and Weber, G., Biochem. J., 65, 476 (1957).
Weber, G., Biochem. J., 75, 345 (1960).
Weber, G., and Teale, F., Disc. Farad. Soc., 27, 134 (1959).
Forster, T., Fluoreszenz Organischen Verbindugen (Vanderhoeck and Ruprecht, Gottingen, 1951).
Yang, J., and Foster, J., J. Amer. Chem. Soc., 76, 1588 (1954).
Williams, E., and Foster, J., J. Amer. Chem. Soc., 82, 242 (1960).
Katz, S., and Klotz, I., Arch. Biochem. Biophys., 44, 351 (1953).
Edelhoch, H., J. Amer. Chem. Soc., 79, 6100 (1957).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
STEINER, R., EDELHOCH, H. Influence of pH and Urea on the Ultra-violet Fluorescence of Several Globular Proteins. Nature 192, 873–874 (1961). https://doi.org/10.1038/192873a0
Issue Date:
DOI: https://doi.org/10.1038/192873a0
- Springer Nature Limited