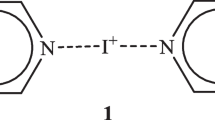Abstract
RECENTLY, several authors have postulated the existence of positive monovalent bromine ions on purely theoretical grounds1,2, or in order to explain the fact that hypobromous acid (HOBr) acts as a powerful brominating agent in strongly acid solutions3. This latter mechanism of substitution is very similar to that of nitration in strong sulphuric acid solution as suggested by several investigators4–6. Pyridine complexes of positive iodine and bromine ions have been prepared by the reaction of the halogen with silver nitrate in organic solvents7,8.
Similar content being viewed by others
References
Hinshelwood, C. N., J. Chem. Soc., 696 (1947).
Weiss, J., Ann. Rep. Chem. Soc., 44, 79 (1947).
Derbyshire, D. H., and Waters, W. A., Nature, 164, 446 (1949).
Melander, L., Nature, 163, 599 (1949).
Gillespie, R. J., Hughes, E. D., Ingold, C. K., Millen, D. J., and Reed, R. I., Nature, 163, 599 (1949).
Bonner, T. G., James, M. E., Lowen, A. M., and Williams, G., Nature, 163, 955 (1949); see also their bibliography.
Uschakow, M. I., and Tchistow, W. O., J. allgem. Chemie (Russian), 1 (63), 1258 (1931); 4, 194 (1934); Ber. deut. Chem. Ges., 68, 824 (1935).
Carlsohn, H., Ber. deut. Chem. Ges., 68, 2209 (1935); Z. angew. Chem., 46, 747 (1933).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
GONDA-HUNWALD, K., GRÁF, G. & KÖRÖSY, F. Positive Bromine Ions. Nature 166, 68–69 (1950). https://doi.org/10.1038/166068a0
Issue Date:
DOI: https://doi.org/10.1038/166068a0
- Springer Nature Limited
This article is cited by
-
Positive Bromine Ions?
Nature (1951)