Abstract
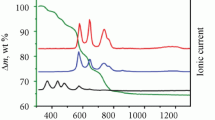
IN connexion with earlier investigations on uranium oxides in this Institute, it was observed that uranium dioxide (UO2) can react with oxygen at so low a temperature as 120° C. To obtain more exact information about this low-temperature oxidation of UO2, the oxide was heated in purified oxygen at different temperatures from 100° to 265° C. and the reaction products examined by the X-ray powder method1.
Similar content being viewed by others
References
Cf. Haraldsen, H., Tidsskr. Kjemi, Bergv. and Metallurgi, 7, 98 (1947). Grønvold, F., lecture held at the 6. Nordiske Kjemikermøte in Lund (1947).
Lydén, R., Finska Kemistsamf. Medd., 48, 124 (1939).
Biltz, W., and Müller, H., Z. anorg. allg. Chem., 163, 257 (1927).
Jolibois, P., C.R. Acad. Sci., Paris, 224, 1395 (1947).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
GEØNVOLD, F., HARALDSEN, H. Oxidation of Uranium Dioxide (UO2). Nature 162, 69–70 (1948). https://doi.org/10.1038/162069a0
Issue Date:
DOI: https://doi.org/10.1038/162069a0
- Springer Nature Limited
This article is cited by
-
Microstructural analysis of pitchblende in the Miaoershan area using TEM lattice imaging
Mineralogy and Petrology (2019)
-
Investigation of reaction conditions on synthesis of UO2.34 and UO2 via hydrothermal route
Journal of Radioanalytical and Nuclear Chemistry (2017)
-
Determination of specific surface area of uranium oxide powders using differential thermal analysis technique
Journal of Thermal Analysis (1995)