Abstract
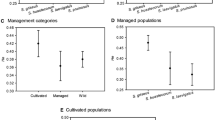
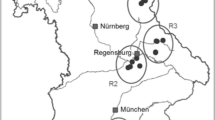
Bouteloua gracilis (blue grama grass) native populations have been shown to be highly variable, however the genetic basis of this variability has not been well established. Determining the extent of genetic variability within and among plant populations have important repercussions for the management and conservation of species, and in particular for those subjected to intensive use such as forage plants. Using RAPD, this study was undertaken to investigate the genetic variability of four B. gracilis native populations developed in three grasslands and one shrubland at the southernmost part of the North American Graminetum in México. Significant differences in grass aboveground production were found among the study sites, while considerable genetic variation within each of the four blue grama populations evaluated was detected. The molecular analysis, based on 55 individuals, revealed a total of 108 scorable repeatable bands, with 99 of them being polymorphic (overall polymorphism= 91.7%). Within every population each individual was genetically distinct and no population-specific bands (fixed marker differences) were identified. Pair-wise ΦST comparisons indicated that the four blue grama populations examined were significantly different in their genetic constitution (P<0.001). AMOVA revealed that most of the genetic variation detected in Bouteloua gracilis was explained by intra- (88.53%), rather than by inter-population (11.47%) differences. UPGMA based on the ΦST values indicated that the blue grama population collected from the shrubland displayed the RAPD profiles that most differed among the study sites. Possible causes of these results could reside on intensive grazing reducing, and proper management conserving, the forage production and genetic diversity of blue grama native populations. Our results are consistent with previous studies analyzing population genetic variation in outcrossing grasses and, in particular, with ecological and cytological evidence for a high genetic variability in native populations of B. gracilis. The implications of our findings and prospective studies to be undertaken using molecular tools in the study of blue grama biology and ecology are discussed.
Similar content being viewed by others
References
Aguado-Santacruz G.A. 1987. Cambios en la vegetación de áreas excluidas del pastoreo en los Llanos de Ojuelos. B.Sc. thesis, Departamento de Biología. Universidad Autónoma de Aguascalientes. Aguascalientes, México.
Aguado-Santacruz G.A. 1994. Análisis de algunos efectos del pastoreo en agostaderos del Altiplano Central. Campo Experimental Vaquerías. CIPAC/Instituto Nacional de Investigaciones Forestales y Agropecuarias. Ojuelos, Jalisco, México. Folleto Técnico No. 4.
Aguado-Santacruz G.A. and Fierro L.C. 1988. Respuesta de la vegetación a la exclusión del ganado en tres pastizales de los Llanos de Ojuelos. Manejo de Pastizales-SOMMAP 1: 11–;15.
Aguado-Santacruz G.A. and García-Moya E. 1998. Environmental factors and community dynamics at the southernmost part of the North American Graminetum. I. On the contribution of climatic factors to temporal variation in species composition. Plant Ecology 135: 13–29.
Aguado-Santacruz G.A., García-Moya E., Creque J.A., Meyer S. and Flores-Flores J.L. 2002. Environmental factors and community dynamics at the southernmost part of the North American Graminetum. II. Temporal plant assemblages determined by rainfall patterns. Plant Ecology 158: 49–63.
Aguado-Santacruz G.A., Luna M. and Giner R.A. 1989. Respuesta de la vegetación y el suelo de un pastizal de los Llanos de Ojuelos al pastoreo inmoderado. Manejo de Pastizales-SOMMAP 1: 1–3.
Aguilera M.O. and Lauenroth W.K. 1993. Seedling establishment in adult neighborhoods-intraspecific constraints in the regeneration of the bunchgrass Bouteloua gracilis. Journal of Ecology 81: 253–261.
Allred B.W. 1950. Practical grassland management. Sheep and Goat Raiser Magazine. San Angelo, Texas, USA.
Canfield R.H. 1941. Application of the line interception method in sampling range vegetation. Journal of Forestry 34: 388–394.
Carney H.J. 1989. On competition and the integration of population, community and ecosystem studies. Functional Ecology 3: 637–641.
Coffin D.P. and Lauenroth W.K. 1989. Spatial and temporal variation in the seed bank of a semiarid grassland. American Journal of Botany 76: 53–58.
Coffin D.P. and Lauenroth W.K. 1990. A gap dynamics simulation model of succession in a semiarid grassland. Ecological modeling 49: 229–266.
Coffin D.P. and Lauenroth W.K. 1992. Spatial variability in seed production of the perennial bunchgrass Bouteloua gracilis (Gramineae). American Journal of Botany 79: 347–353.
Crnokrak P. and Merilä J. 2002. Genetic population divergence: markers and traits. Trends in Ecology and Evolution 17: 501.
Dawson I.K., Chalmers K.J., Waugh R. and Powell W. 1993. Detection and analysis of genetic variation in Hordeum spontaneum populations from Israel using RAPD markers. Molecular Ecology 2: 151–159.
Detling J.K. 1979. Processes controlling blue grama production on the shortgrass prairie. pp. 25–;42. In: French N.R. (ed.), Perspectives in Grassland Ecology. Springer-Verlag, New York, New York, USA.
Excoffier L., Smouse P.E. and Quattro J.M. 1992. Analysis of molecular variance inferred from metric distances among DNA haplotypes: application to human mitochondrial DNA restriction data. Genetics 131: 479–491.
Fair J., Lauenroth W.K. and Coffin D.P. 1999. Demography of Bouteloua gracilis in a mixed prairie: analysis of genets and individuals. Journal of Ecology 87: 233–243.
Ferdinandez Y.S.N., Somers D.J. and Coulman B.E. 2001. Estimating the genetic relationship of hybrid bromegrass to smooth bromegrass and meadow bromegrass using RAPD markers. Plant Breeding 120: 149–153.
Green C.E. and Goetz H. 1973. Morphological variation in three ecotypes of Agropyron smithii Rydb. and Bouteloua gracilis (H.B.K.) Lag. ex Steud. North Dakota State Agricultural Experimental Station Bulletin 491.
Gustafsson A. 1946. Apomixis in higher plants. Otto Harrassowitz, Leipzig, Germany.
Hamrick J.L. 1990. Isozymes and the analysis of genetic structure in plant populations. pp. 87–105. In: Soltis E.D. and Soltis P.S. (eds), Isozymes in Plant Biology. Chapman and Hall, London, UK.
Hamrick J.L. and Godt M.J.W. 1989. Allozyme diversity in plant species. pp. 43–;63. In: Bown A.D.H, Clegg M.T., Kahler A.L. and Weir B.S. (eds), Plant Population Genetics, Breeding and Genetic Resources. Sinauer Associates, Sunderland, Massachusetts, USA.
Harper J.L. 1977. Population Biology of Plants. Academic Press, London, UK.
Hendry A.P. 2002. Qst > = ≠ < Fst ?. Trends in Ecology and Evolution 17: 502.
Ho C.Y., McMaugh S.J., Wilton A.N., McFarlane I.J. and Mackinlay A.G. 1997. DNA amplification variation within cultivars of turf-type Couch grasses (Cynodon spp). Plant Cell Reports 16: 797–801.
Hogbin P.M. and Peakall R. 1999. Evaluation of the contribution of genetic research to the management of the endangered plant Zieria prostrata. Conservation Biology 13: 514–522.
Huff D.R. 1997. RAPD characterization of heterogeneous perennial ryegrass cultivars. Crop Science 37: 557–564.
Huff D.R., Peakall R. and Smouse P.E. 1993. RAPD variation within and among natural populations of outcrossing buffalograss [Buchloë dactyloides (Nutt.) Engelm.]. Theoretical and Applied Genetics 86: 927–934.
Huff D.R., Quinn J.A., Higgins B. and Palazzo A.J. 1998. Random amplified polymorphic DNA (RAPD) variation among native little bluestem [Schizachyrium scoparium (Michx.) Nash] populations from sites of high and low fertility in forest and grassland biomes. Molecular Ecology 7: 1591–1597.
Hyder D.N., Everson A.C. and Bement R.E. 1971. Seedling morphology and seeding failures with blue grama. Journal of Range Management 24: 287–292.
John T.V., St. and Coleman D.C. 1983. The role of mycorrhizae in plant ecology. Canadian Journal of Botany 61: 1005–1014.
Johnson N.C., Zak D.R., Tilman D. and Pfleger F.L. 1991. Dynamics of vesicular-arbuscular mycorrhizae during old-field succession. Oecologia 86: 349–358.
Kölliker R., Stadelmann F.J., Reidy B. and Nösberger J. 1998. Fertilization and defoliation frequency affect genetic diversity of Festuca pratensis Huds. in permanent grasslands. Molecular Ecology 7: 1557–1567.
Kresovich S., Lamboy W.F., Li R., Ren J., Szewc-McFadden A.K. and Bliek S.M. 1994. Application of molecular methods and statistical analyses for discrimination of accessions and clones of Vetiver grass. Crop Science 34: 805–809.
Lee S.B. and Taylor J.W. 1990. Isolation of DNA from fungal mycelia and single spores. Pp. 282–;287. In: Innis M.A., Gelfand D.H., Sninsky J.J. and White T.J. (eds), PCR Protocols. A Guide to Methods and Applications. Academic Press, San Diego, California, USA.
Loveless M.D. and Hamrick J.L. 1984. Ecological determinants of genetic structure in plant populations. Annual Review of Ecology and Systematics 15: 65–95.
McGinnies W.J., Laycock W.A., Tsuchiya T., Yonker C.M. and Edmunds D.A. 1988. Variability within a native stand of blue grama. Journal of Range Management 41: 391–;395.
McKay J.K. and Latta R.G. 2002. Adaptive population divergence: markers, QTL and traits. Trends in Ecology and Evolution 17: 285–291.
McMillian C. 1956. Nature of the plant community. I. Uniform garden and light period studies of five grass taxa in Nebraska. Ecology 37: 330–340.
Miller M.P. 2000. AMOVA-PREP. Department of Biological Sciences. Northern Arizona University. Flagstaff, Arizona, USA.
Molina C. 1990. Caracterización del banco de semillas de un pastizal con diferente grado de deterioro. M.Sc. thesis, Centro de Botánica. Colegio de Postgraduados. Chapingo, México.
Moran G.F. 1992. Patterns of genetic diversity in Australian tree species. New Forests 6: 49–66.
Mosher R.A. and Smith E. 1997. Quantifying apomixis in two populations of sideoats grama. In: Ninth Annual Undergraduate Biology Research Conference. The University of Arizona (http://www.blc.arizona.edu/ubrp/conference/abstracts).
Mueller I.M. 1941. An experimental study of the rhizomes of certain prairie plants. Ecological Monographs 11: 165–188.
Nei M. 1972. Genetic distance between populations. American Naturalist 106: 283–292.
Nei M. 1973. Analysis of gene diversity in subdivided populations. Proceedings of the National Academy of Sciences USA 70: 3321–3323.
Nei M and Li W-H. 1979. Mathematical model for studying genetic variation in terms of restriction endonucleasas. Proceedings of the National Academy of Sciences USA 76: 5269–5273.
Newsham K.K. and Watkinson A.R. 1998. Arbuscular mycorrhizas and the population biology of grasses. pp. 286–;310. In: Cheplick G.P. (ed.), Population Biology of Grasses. Cambridge University Press, Cambridge, UK.
Newton A.C., Allnutt T., Gillies A.C.M., Lowe A. and Ennos R.A. 1999. Molecular phylogeography, intraspecific variation and the conservation of tree species. Trends in Ecology and Evolution 14: 140–145.
Nybom H. and Bartish I.V. 2000. Effects of life history traits and sampling strategies on genetic diversity estimates obtained with RAPD markers in plants. Perspectives in Plant Ecology, Evolution and Systematics 3: 93–114.
Ouborg N.J., Piquot Y. and Van Groenendael J.M. 1999. Population genetics, molecular markers and the study of dispersal in plants. Journal of Ecology 87: 551–568.
Pérez H., Bravo S., Ongaro V., Castagnaro A., García-Seffino L. and Taleisnik E. 1999. Chloris gayana cultivars: RAPD polymorphism and field performance under salinity. Grass and Forage Science 54: 289–296.
Pool R.J. 1948. Marching with the grasses. University of Nebraska Press, Nebraska, USA.
Riegel A. 1940. A study of the variations in the growth of blue grama grass from seed produced in various sections of the Great Plains region. Transactions of the Kansas Academy of Science 43: 155–171.
Ritland K. 2000. Marker-inferred relatedness as a tool for detecting heritability in nature. Molecular Ecology 9: 1195–1204.
Samuel M.J. 1985. Growth parameter differences between populations of blue grama. Journal of Range Management 38: 339–342.
Samuel M.J. and Hart R.H. 1995. Observations on spread and fragmentation of blue grama clones in disturbed rangeland. Journal of Range Management 48: 508–510.
Sangwan N.S., Yadav U. and Sangwan R.S. 2001. Molecular analysis of genetic diversity in elite Indian cultivars of essential oil trade types of aromatic grasses (Cymbopogon species). Plant Cell Reports 20: 437–444.
Shannon C.E. and Weaver W. 1949. The Mathematical Theory of Communication. University of Illinois Press, Urbana, Illinois, USA.
Snaydon R.W. and Davies T.M. 1982. Rapid divergence of populations in response to recent changes in soil conditions. Evolution 30: 289–297.
Snyder L.A. and Harlan J.R. 1953. A cytological study of Bouteloua gracilis from Western Texas and Eastern New Mexico. American Journal of Botany 40: 702–707.
Steel R.G.D and Torrie J.H. 1960. Principles and Procedures of Statistics. McGraw-Hill. New York, USA.
Stubbendieck J., Launchbaugh J.L., Burzlaff D.F. and McCully W.G. 1973. Stoloniferous blue grama. Journal of Range Management 26: 230–231.
Watkinson A.R. and Powell J.C. 1993. Seedling recruitment and the maintenance of clonal diversity in plant populations-a computer simulation of Ranunculus repens. Journal of Ecology 81: 707–717.
Wilson A.M. and Briske D.D. 1979. Seminal and adventitious root growth of blue grama seedlings on the Central Plains. Journal of Range Management 32: 209–213.
Yeh F.C., Yang R. and Boyle T. 1999. POPGENE version 1.31. Microsoft Window-based Freeware for population genetic analysis. University of Alberta/Centre for International Forestry Research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Aguado-Santacruz, G.A., Leyva-López, N.E., Pérez-Márquez, K.I. et al. Genetic variability of Bouteloua gracilis populations differing in forage production at the southernmost part of the North American Graminetum. Plant Ecology 170, 287–299 (2004). https://doi.org/10.1023/B:VEGE.0000021706.12328.61
Issue Date:
DOI: https://doi.org/10.1023/B:VEGE.0000021706.12328.61