Abstract
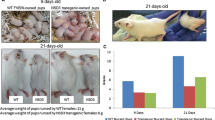
Female transgenic mice, in which a murine c-myc gene has been placed under the transcriptional control of the mouse mammary tumor virus long terminal repeat, are prone to developing mammary adenocarcinomas. Owing to the manner in which these mammary tumors develop, it is clear that exogenous expression of the c-myc transgene is necessary to but insufficient for murine mammary tumorigenesis. The genetic background of study mice has been shown to influence the phenotype induced by different transgenes; furthermore, mammary tumor initiation and progression induced by different transgenes has been shown to be susceptible to significant modification with alterations in and mixing of the genetic background of the study mice. We bred MMTV-c-myc transgenic mice onto a mixed genetic background that resulted in a very significant suppression of mammary tumor incidence for parous mice, bred continuously starting at 10 weeks of age. In this paper, we show that mammary tumor incidence is significantly elevated in these mixed background MMTV-c-myc transgenic mice when they are bred continuously, starting at 7 weeks of age. Early breeding of these mice did not influence mammary tumor multiplicity, latency, histopathology, or number of pregnancies at time of tumor development. These results are the first to demonstrate that breeding age influences mammary tumor incidence in MMTV-c-myc transgenic mice. They suggest that mammary gland susceptibility to tumorigenesis, resulting from the expression of c-myc, may vary with glandular development as is seen for chemical carcinogens.
Similar content being viewed by others
References
Amundadottir LT, Johnson MD, Merlino G, Smith GH and Dickson RB (1995) Synergistic interaction of transforming growth factor α and c-myc in mouse mammary and salivary gland tumorigenesis. Cell Growth Diff 6: 737–748.
Cardiff RD (1996) The biology of mammary transgenes: five rules. J Mammary Gland Biol Neoplasia 1: 61–73.
Cardiff RD, Anver MR, Gusterson BA, Hennighausen L, Jensen RA, Merino MJ et al. (2000) The mammary pathology of genetically engineered mice: the Consensus report and recommendations from the Annapolis meeting. Oncogene 19: 968–988.
Desai KV, Xiao N, Wang W, Gangi L, Greene J, Powell JI et al. (2002) Initiating oncogenic event determines gene-expression patterns of human breast cancer models. Proc Natl Acad Sci USA 99: 6967–6972.
Doerfler W, Schubbert R, Heller H, Kämmer C, Hilger-Eversheim K, Knoblauch M et al. (1997) Integration of foreign DNA and its consequences in mammalian systems. Trends Biotechnol 15: 297–301.
Doetschmann T (1999) Interpretation of phenotype in genetically engineered mice. Lab Anim Sci 49: 137–143.
Donehower LA, Andre J, Berard DS, Wolford RG and Hager GL (1980) Construction and characterization of molecular clones containing integrated mouse mammary tumor virus sequences. Cold Spring Harbor Symp Quant Biol 44: 1153–1159.
Donehower LA, Huang AL and Hager GL (1981) Regulatory and coding potential of the mouse mammary tumor virus long terminal redundancy. J Virol 37: 226–238.
Hennighausen L and Robinson GW (1998) Think globally, act locally: the making of a mouse mammary gland. Genes Dev 12: 449–455.
Le Voyer T, Lu Z, Babb J, Lifsted T, Williams M and Hunter KW (2000) An epistatic interaction controls the latency of a transgene-induced mammary tumor. Mamm Genome 11: 883–889.
Leder A, Pattengale PK, Kuo A, Stewart TA and Leder P (1986) Consequences of widespread deregulation of the c-myc gene in transgenic mice: multiple neoplasms and normal development. Cell 45: 485–495.
Lifsted T, Le Voyer T, Williams M, Muller W, Klein-Szantos A, Buetow KH et al. (1998) Identification of inbred mouse strains harboring genetic modifiers of mammary tumor age of onset and metastatic progression. Int J Cancer 77: 640–644.
MacLeod KF and Jacks T (1999) Insights into cancer from transgenic mouse models. J Path 187: 43–60.
Mangues R, Schwartz S, Seidman I and Pellicer A (1995) Promoter demethylation isMMTV/N-rasN transgenic mice required for transgene expression and tumorigenesis. Mol Carcinogen 14: 94–102.
Robertson KD (2002) DNA methylation and chromatin – unraveling the tangled web. Oncogene 21: 5361–5379.
Rowse GJ, Ritland SR and Gendler SJ (1998) Genetic modulation of neu proto-oncogene-induced mammary tumorigenesis. Cancer Res 58: 2675–2679.
Russo J and Russo IH (1996) Experimentally induced mammary tumors in rats. Breast Cancer Res Treat 39: 7–20.
Stewart TA, Pattengale PK and Leder P (1984) Spontaneous mammary adenocarcinomas in transgenic mice that carry and express MTV/myc fusion genes. Cell 38: 627–637.
Zhou H, Chen W, Qin X, Lee K, Liu L, Markowitz SD et al. (2001) MMTV promoter hypermethylation is linked to spontaneous and MNU associated c-neu expression and mammary carcinogenesis in MMTV-c-neu transgenic mice. Oncogene 20: 6009–6017.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jamerson, M.H., Johnson, M.D., Furth, P.A. et al. Early Parity Significantly Elevates Mammary Tumor Incidence in MMTV-c-myc Transgenic Mice. Transgenic Res 12, 747–750 (2003). https://doi.org/10.1023/B:TRAG.0000005247.69329.ca
Issue Date:
DOI: https://doi.org/10.1023/B:TRAG.0000005247.69329.ca