Abstract
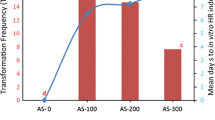
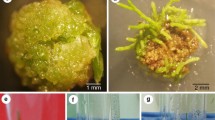
Transformed roots of Lupinus mutabilis cv. Potosi induced by Agrobacterium rhizogenes strain R1601 were cultured on Murashige and Skoog-based medium lacking kanamycin sulphate, or with this antibiotic at 40 mg l−1. The neomycin phosphotransferase gene in the genome of transformed roots was confirmed by non-radioactive Southern hybridisation. Neomycin phosphotransferase protein was detected by ELISA. Transformed roots synthesised isoflavones, but not quinolizidine alkaloids; the latter are typical secondary metabolites of lupin normally produced in aerial parts of the plant. Genistein and 2′-hydroxygenistein, were the main secondary metabolites in cultured, transformed roots, whereas the glycoside genistin was more abundant in roots of non-transformed plants. Wighteone concentrations in transgenic roots were higher than those of non-transformed roots. Transformed roots produced twice the concentration of isoflavones compared with roots from non-transformed plants, indicating that Ri plasmid T-DNA genes modified isoflavone concentration and pattern of biosynthesis.
Similar content being viewed by others
References
Alhasan SA, Aranha O & Sarkar FH (2001) Genistein elicits pleiotropic molecular effects on head and neck cancer cells. Clin. Cancer Res. 7: 4174-4181
Babaoglu M, Power JB & Davey MR (2000a) Genetic engineering of grain legumes: key transformation events. AgbiotechNet 2 June, ABN 050: 1-8
Babaoglu M, McCabe M, Power JB & Davey MR (2000b) Agrobacterium-mediated transformation of Lupinus mutabilis using shoot apical explants. Acta Physiol. Plant. 22: 111-119
Baiza AM, Quiroz-Moreno A, Ruiz JA & Loyola-Vargas VM (1999) Genetic stability of hairy root cultures of Datura stramonium. Plant Cell Tiss. Org. Cult. 59: 9-17
Baumel PJ, Jeschke WD, Rath N, Czygan F-C & Proksch P (1995) Modelling of quinolizidine alkaloid net flows in Lupinus albus and between L. albus and the parasite Cuscuta reflexa: new insights into the site of quinolizidine alkaloid synthesis. J. Exp. Bot. 46: 1721-1730
Berlin J, Fecker L, Rugenhagen C, Sator C, Strack D, Witte L & Wray V (1991a) Isoflavone glycoside formation in transformed and non-transformed suspension and hairy cultures of L. polyphyllus and L. hartwegii. Z. Naturforsch. 46c: 725-734
Berlin J, Rugenhagen C, Rippert M & Erdogan S (1991b) Effects of culture conditions on isoflavonoid levels of transformed and nontransformed cultures of Lupinus- A comparison of suspension and hairy root cultures. Z. Naturforsch. 46c: 735-742
Boehm R, Sommer S, Li S-M & Heide L (2000) Genetic engineering on shikonin biosynthesis: expression of the bacterial ubiA gene in Lithospermum erythrorhizon. Plant Cell Physiol. 41: 911-919
Cho H-J, Farrand SK, Noel GR & Widholm JM (2000) Highefficiency induction of soybean hairy roots and propagation of the soybean cyst nematode. Planta 210: 195-204
Damiano C & Monticelli S (1998) In vitro fruit trees rooting by Agrobacteirum rhizogenes wild type of infection. Electron. J. Biotechnol. 1: 1-8 (http:www.ejb.org/content/vol11/ issue3/full/4/index.html)
Dellaporta SL, Wood J & Hicks JB (1983) A plant DNA minipreparation: version II. Plant Mol. Biol. Rep. 4: 9-21
Fukutake M, Takahashi M, Ishida K, Kawamura H, Sugimura T & Wakabayashi K (1996) Quantification of genistein and genistin in soybeans and soybean products. Food Chem. Toxicol. 34: 457-461
Hoeck JA, Fehr WR, Murphy PA & Welke GA (2000) Influence of genotype and environment on isoflavone contents of soybean. Crop Sci. 40: 48-51
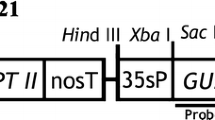
Jefferson RA, Kavanagh TA & Bevan MW (1987) GUS fusions: ß-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. EMBO J. 6: 3901-3907
Joao KHL & Brown TA (1994) Long-term stability of root cultures of tomato transformed with Agrobacterium rhizogenes R1601. J. Exp. Bot. 45: 641-647
Jung W, Yu O, Lau SMC, O'Keefe DP, Odell J, Fader G & McGonigle B (2000) Identification and expression of isoflavone synthase, the key enzyme for biosynthesis of isoflavones in legumes. Nature Biotechnol. 18: 208-212
Kneer R, Poulev AA, Olesinski A & Raskin I (1999) Characterization of the elicitor-induced biosynthesis and secretion of genistein from roots of Lupinus luteus L. J. Exp. Bot. 50: 1553-1559
Kovats E (1958) Gaschromatographische charakterisierung organischer verbindungen. 1. Retentionsindices aliphatischer halogenide, alkohole, aldehyde und ketone. Helv. Chim. Acta 41: 1915-1932
Krishnamurthy KV, Suhasini K, Sagare AP, Meixner M, de Kathen A, Pickardt T & Schieder O (2000) Agrobacterium-mediated transformation of chickpea (Cicer arietinum L.) embryo axes. Plant Cell Rep. 19: 235-240
Kumar V, Jones B & Davey MR (1991) Transformation by Agrobacterium rhizogenes and regeneration of transgenic shoots of the wild soybean Glycine argyrea. Plant Cell Rep. 10: 135-138
Li H, Wylie SJ & Jones MGK (2000) Transgenic yellow lupin (Lupinus luteus). Plant Cell Rep. 19: 634-637
McCabe MM, Power JB, de Laat AMM & Davey MR (1996) Detection of single copy genes in DNA from transgenic plants by non-radioactive Southern blot analysis. Mol. Biotechnol. 7: 1-6
Merkli A, Christen P & Kapetanidis I (1997) Production of diosgenin by hairy root cultures of Trigonella foenum-graecum L. Plant Cell Rep. 16: 632-636
Molvig L, Tabe LM, Eggum BO, Moore AE, Craig S, Spencer D & Higgins TJV (1997) Enhanced methionine levels and increased nutritive value of seeds of transgenic lupins (Lupinus angustifolius L.) expressing a sunflower seed albumin gene. Proc. Natl. Acad. Sci. USA 94: 8393-8398
Moore L, Warren G & Strobel GA (1979) Involvement of a plasmid in the hairy root disease of plants caused by Agrobacterium tumefaciens. Plasmid 2: 617-626
Mugnier J (1988) Establishment of new axenic hairy root lines by inoculation with Agrobacterium rhizogenes. Plant Cell Rep. 7: 9-12
Murashige T & Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant. 15: 473-497
Pigeaire A, Abernethy D, Smith PM, Simpson K, Fletcher N, Lu C-Y, Atkins CA & Cornish E (1997) Transformation of a grain legume (Lupinus angustifolius L.) via Agrobacterium tumefaciens-mediated gene transfer to shoot apices. Mol. Breed. 3: 341-349
Porter JR (1991) Host range and implications of plant infection by Agrobacterium rhizogenes. Crit. Rev. Plant Sci. 10: 387-421
Pythoud F, Sinkar VP, Nester EW & Gordon MP (1987) Increased virulence of Agrobacterium rhizogenes conferred by the vir region pTiBo542: application to genetic engineering of poplar. Bio/Technol. 5: 1323-1327
Rech EL, Golds TJ, Husnain T, Vainstein MH, Jones B, Hammatt N, Mulligan BJ & Davey MR (1989) Expression of a chimaeric resistance gene introduced into the wild soybean Glycine canescens using a cointegrate Ri plasmid vector. Plant Cell Rep. 8: 3-36
Saalbach I, Pickardt T, Machemehl F, Saalbach G, Schieder O & Muntz K (1994) A chimeric gene encoding the methionine-rich 2S albumin of the Brazil (Bertolletia excelsa H.B.K) is stably expressed and inherited in transgenic grain legumes. Mol. Gen. Genet. 242: 226-236
Sambrook J, Fritsch EF & Maniatis T (1989) Molecular cloning. A Laboratory Manual Vol. 3, 2nd Edition, Cold Spring Harbor Laboratory Press, Cold Spring Harbor, USA
Shanks JV & Morgan J (1999) Plant’ hairy root’ culture. Curr. Opin. Biotechnol. 10: 151-155
Siefkes-Boer HJ, Noonan MJ, Bullock DW & Conner AJ (1995) Hairy root transformation system in large seeded grain legumes. Israel J. Plant Sci. 43: 1-5
Taylor BH, Amasino RM, White FF, Nester EW & Gordon MP (1985) T-DNA analysis of plants regenerated from hairy root tumours. Mol. Gen. Genet. 201: 554-557
Tepfer D (1990) Genetic transformation using Agrobacterium rhizogenes. Physiol. Plant. 79: 140-146
Win W, Cao ZX, Peng XX, Trush MA & Li YB (2002) Different effects of genistein and resveratrol on oxidative DNA damage in vitro. Mutation Res.-Genet. Toxicol. Environ. Mut. 513: 113-120
Wink M (1987) Site of lupanine and sparteine biosynthesis in intact plants and in vitro organ cultures. Z. Naturforsch. 42c: 868-872
Wink M (1993) Quinolizidine alkaloids. In:Waterman P (ed) Methods in Plant Biochemistry Vol. 8 (pp. 197-239). Academic Press, London
Yu O, Junk W, Shi J, Croes RA, Fader GM, McGonigle B & Odell JT (2000) Production of the isoflavones genistein and daidzein in non-legume dicot and monocot tissues. Plant Physiol. 124: 781-794
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Babaoglu, M., Davey, M.R., Power, J.B. et al. Transformed Roots of Lupinus mutabilis: Induction, Culture and Isoflavone Biosynthesis. Plant Cell, Tissue and Organ Culture 78, 29–36 (2004). https://doi.org/10.1023/B:TICU.0000020386.03780.ea
Issue Date:
DOI: https://doi.org/10.1023/B:TICU.0000020386.03780.ea