Abstract
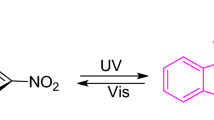
The crystal structure of the photochromism compound 3-{3,3-dimethyl-6'-nitrospiro[2'H-chromene-2,2'-(2,3-dihydro-1H-indole)]-1-yl}-propanoic acid, SP1, has been characterized by X-ray crystallography. We compared it with the crystal structure of 3-{2-[2-(3,5-dinitro-2-oxidophenyl)ethyl]-3,3-dimethyl-3H-indol-1-ium-1-yl}-propanoic acid. SP2 solvated with two DMF molecules that was obtained earlier. We found that great differences existed between them. Such differences can also be easily found by their [1H]NMR and IR spectra. Their photochromic behaviors in solutions were investigated by UV-vis absorption with the aid of ultraviolet irradiation, leading to the conclusion that SP1 tended to the closed form while SP2 tended to be the open form both in solution and in the solid state.
Similar content being viewed by others
References
Fischer, E.; Hirschberg, Y. J. Chem. Soc. 1952, p. 4522.
Hirschberg, Y. J. Amer. Chem. Soc. 1956, 78, 2304.
Bertelson, R. C. Photochromism; Brown, G. H. Ed.; Wiley (Interscience): New York, 1971.
Guglielmetti, R. Photochromism: Molecules and Systems; Dürr, H., Bouas-Laurent, H. Eds., Elsevier: Amsterdam, 1990.
Rim, R. H.; Hirschberg, Y; Fischer, E. J. Phys. Chem. 1962, 66, 2470.
Heiligman, R.; Hirschberg, Y.; Fischer, E. J. Chem. Soc. 1961, p. 156; Heiligman, R.; Hirschberg, Y.; Fischer, E. J. Phys. Chem. 1962, 66, 2465, and references therein.
Becker, R. S.; Michl, J. J. Phys. Chem. 1966, 88, 5931.
Becker, R. S.; Kolc, J. J. Phys. Chem. 1967, 71, 4045.
Li, X. L.; Wang, Y. M.; Matsuura, T.; Meng, J. B. Heterocycles 1999, 51, 2639.
Li, X. L.; Li, J. L.; Wang, Y. M.; Matsuura, T.; Meng, J. B. Mol. Cryst. Liq. Cryst. 2000, 344, 295.
Li, X. L.; Wang, Y. M.; Matsuura, T.; Meng, J. B. Mol. Cryst. Liq. Cryst. 2000, 344, 301.
Zhang, P.; Meng, J. B.; Li, X. L.; Matsuura, T.; Wang, Y. M. Heterocyclic Chem. 2002, 39, 1.
Zou, W. X.; Chen, P. L.; Gao, Y.; Meng, J. B. Acta Crystallogr. E 2003, 59, o337.
Sheldrick, G. M. SHELXTL. Version 5.10; Bruker AXS Inc.: Madison, Wisconsin, 1998.
Aldoshin, S. M.; Atovmyan, O. L.; Ponomarev, V. I. Khim. Fiz. 1984, 3, 787.
Brown, G.; Marsh, R. E. Acta Crystallogr. 1963, p. 191.
Basak, K.; Mazumdar, S. K. Cryst. Struct. Commun. 1982, p. 609.
Fukuyo, H.; Hirotzu, K.; Higuchi, K. Acta Crystallogr. B 1982, 28, 640.
Gridunova, G. V.; Shklover, V. E.; Yu, Struchkov, T.; Chayanov, B. L., Kristallografiya 1983, 28, 286.
Sazhnikov, V. A.; Alsoshin, S. M.; Efimov, S. P.; Budyka, M. Vses. Soveshchanie Fotokhim. Tezisy Dokl. Suzdal' Ivanovo, 1985, p. 135.
Shustov, G. N.; Zolotoy, A. B.; Kostyanovskii, R. G. Tetrahedron 1982, 38, 2319.
Kennard, O. Molecular Structures and Dimensions; Dosthoek's Unitgevers mij Utrecht: The Netherlands, 1972.
Biryukov, P.; Unkovskii, B. V. In Kristallokhimiya [Crystal Chemistry]; Izdatelstro VINITI: Moscow, 1974; Vol. 9.
Biryukov, P.; Unkovskii, B. V. In Kristallokhimiya [Crystal Chemistry]; Izdatelstro VINITI: Moscow, 1976; Vol. 11.
Coppens, P.; Schmidt, G. M. J. Acta Crystallogr. A 1965, 18, 62.
Coppens, P.; Schmidt, G. M. J. Acta Crystallogr. A 1965, 18, 654.
Desiraju, G. R.; Steiner, T., Eds. The Weak Hydrogen Bond in Structural Chemistry and Biology; Oxford University Press: New York, 1999.
Flannery, J. B. J. Amer. Chem. Soc. 1968, 90, 5660.
Norris, W.; Tyer, Jr.; Becker, R. S. J. Amer. Chem. Soc. 1970, 92, 1289.
Kholmanskii, S.; Dyumaev, K. M. Dokl. Akad. Nauk SSSR 261, p. 704.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zou, W.X., Huang, H.M., Gao, Y. et al. Structures of Two Spiropyrans in the Open and Closed Form. Structural Chemistry 15, 317–321 (2004). https://doi.org/10.1023/B:STUC.0000026746.98051.d6
Issue Date:
DOI: https://doi.org/10.1023/B:STUC.0000026746.98051.d6