Abstract
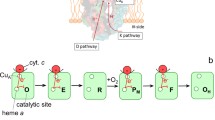
In this minireview an overview is presented of the kinetics of electron transfer within the cytochrome bc 1 complex, as well as from cytochrome bc 1 to cytochrome c. The cytochrome bc 1 complex (ubiquinone:cytochrome c oxidoreductase) is an integral membrane protein found in the mitochondrial respiratory chain as well as the electron transfer chains of many respiratory and photosynthetic bacteria. Experiments on both mitochondrial and bacterial cyatochrome bc 1 have provided detailed kinetic information supporting a Q-cycle mechanism for electron transfer within the complex. On the basis of X-ray crystallographic studies of cytochrome bc 1, it has been proposed that the Rieske iron–sulfur protein undergoes large conformational changes as it transports electrons from ubiquinol to cytochrome c 1. A new method was developed to study electron transfer within cytochrome bc 1 using a binuclear ruthenium complex to rapidly photooxidize cytochrome c 1. The rate constant for electron transfer from the iron–sulfur center to cytochrome c 1 was found to be 80,000 s−1, and is controlled by the dynamics of conformational changes in the iron–sulfur protein. Moreover, a linkage between the conformation of the ubiquinol binding site and the conformational dynamics of the iron–sulfur protein has been discovered which could play a role in the bifurcated oxidation of ubiquinol. A ruthenium photoexcitation method has also been developed to measure electron transfer from cytochrome c 1 to cytochrome c. The kinetics of electron transfer are interpreted in light of a new X-ray crystal structure for the complex between cytochrome bc 1 and cytochrome c.
Similar content being viewed by others
References
Ahmed AJ, Smith HT, Smith MB and Millett F (1978) Effect of speci c lysine modi cation on the reduction of cytochrome c by succinate–cytochrome c reductase. Biochemistry 17: 2479–2483
Axelrod HL, Abresch EC, Okamura MY, Yey AP, Rees DC and Feher G (2002) X-ray structure determination of the cytochrome c 2: reaction center electron transfer complex from Rhodobacter sphaeroides. J Mol Biol 319: 501–515
Bartoschek S, Johansson M, Geierstanger BH, Okun JG, Lancaster CR, Humpfer E, Yu L, Yu CA, Griesinger C and Brandt U (2001) Three molecules of ubiquinone bind speci cally to mitochondrial cytochrome bc 1 complex. J Biol Chem 276: 35231–35234
Beratan DN, Betts JN and Onuchic JN (1991) Protein electron transfer rates set by the bridging secondary and tertiary structure. Science 252: 1285–1288
Bowyer JR and Crofts AR (1981) Inhitibion of electron transport in R. capsulatus by a ubiquinone analogue. In: Dutton PL, Leigh J and Scarpa A (eds) Frontiers of Biological Energetics, Vol I, pp 326–333. Academic Press, New York
Bowyer JR and Trumpower BL (1981) Pathways of electron transfer in the cytochrome bc 1 complexes of mitochondria and photosynthetic bacteria. In: Skulachev BP and Hinkle PC (eds) Chemiosmotic Proton Circuits in Biologial membranes, pp 105–122. Addison-Wesley, Reading, Massachusetts
Brandt U (1996) Energy conservation by bifurcated electron-transfer in the cytochrome bc 1 complex. Biochim Biophys Acta 1275: 41–46
Brandt U (1998) The chemistry and mechanics of ubihydro-quinone oxidation at center P (Qo )of the cytochrome bc 1 complex. Biochim Biophys Acta 1365: 261–268
Broger C, Salardi S and Azzi A (1983) Interaction between isolated cytochrome c 1 and cytochrome c. Eur J Biochem 131: 349–352
Brugna M, Rodgers S, Schricker A, Montoya G, Kazmeier M, Nitschike W and Sinning I (2000) A spectroscopic method for observing the domain movement of the Rieske iron–sulfur protein. Proc Natl Acad Sci USA 97: 2069–2074
Crofts AR and Meinhardt SW (1982) A Q-cycle mechanism for the cyclic electron transfer chain of Rp. sphaeroides. Biochem Soc Trans 10: 210–203
Crofts AR and Wang Z (1989) How rapid are the internal reactions of the ubiquinol: cytochrome c 2 oxidoreductase? Photosynth Res 22: 69–87
Crofts AR, Guergova-Kuras M, Huang L, Kuras R, Zhang Z and Berry EA (1999) Mechanism of ubiquinol oxidation by the bc 1 complex: role of the iron–sulfur protein and its mobility. Biochemistry 38: 15791–15806
Crofts AR, Guergova-Kuras M, Duras R, Ugulava N, Li J and Hong S (2000) Proton-coupled electron transfer at the Qo site: what type of mechanism can account for the high activation barrier? Biochim Biophys Acta 1459: 456–466
Darrouzet E and Daldal F (2002)Movement of the iron–sulfur subunit beyond the ef loop of cytochrome b is required for multiple turnovers of the bc 1 complex but not for a single turnover Qo site catalysis. J Biol Chem 277: 3471–3476
Darrouzet E, Valkova-Valchanova M and Daldal R (2000) Probing the role of the Fe–S subunit hinge region during Qo site catalysis in Rhodobacter capsulatus bc 1 complex. Biochemistry 39: 15475–15483
Darrouzet E, Valkova-Valchanova M and Daldal F (2002) The [2Fe–2S ]cluster E m as an indicator of the iron–sulfur subunit position in the ubihydroquinone oxidation site of the cytochrome bc 1 complex. J Biol Chem 277: 3464–3470
Darrouzet E, Cooley JW and Daldal F (2004) The cytochrome bc 1 complex and its homologue the b 6 f complex: similarities and differences. Photosynth Res 79: 25–44
Davidson V (1996) Unraveling the kinetic complexity of interprotein electron transfer reactions. Biochemistry 35: 14035–14039
De Vries S, Albracht SPJ, Berden JA and Slater EC (1982) The pathway of electron through QH2 cytochrome c oxidoreductase studied by pre-steady-state kinetics. Biochim Biophys Acta 681: 41–53
Ding H, Robertson DE, Daldal F and Dutton PL (1992) Cytochrome bc 1 complex [2Fe–2S ]cluster and its interaction with ubiquinone and ubihydroquinone at the Qo site: a double-occupancy Qo site model. Biochemistry 31: 3144–3158
Ding H, Moser CC, Robertson DE, Tokito MK, Daldal F and Dutton PL (1995) Ubiquinone pair in the Qo site central to the primary energy conversion reactions of cytochrome bc 1 complex. Biochemistry 24: 15979–15996
Engstrom G, Xiao K, Yu C-A, Yu L, Durham B and Millett F (2002) Photoinduced electron transfer between the Rieske iron–sulfur protein and cytochrome c 1 in the Rhodobacter sphaeroides cytochrome bc 1 complex: effects of pH, temperature, and driving force. J Biol Chem 277: 31072–31078
Engstrom G, Rajagukguk R, Saunders AJ, Patel C, Raja-gukguk S, Merbitz-Zahradnik T, Xiao K, Pielak G, Trum-power B, Yu C-A, Gu L, Durham B and Millett F (2003) Design of a ruthenium-labeled cytochrome c derivative to study electron transfer with the cytochrome bc 1 complex. Biochemistry 42: 2816–2824
Gao X, Wen X, Yu C-A, Esser L, Tsao S, Quinn B, Zhang L, Yu L and Xia D (2002) The crystal structure of mitochon-drial cytochrome bc 1 in complex with famoxadone: the role of aromatic–aromatic interaction in inhibition. Biochemistry 41: 11692–11702
Ghosh M, Wang C, Ebert E, Vadlamuri S and Beattie DS (2001) Substituting leucine for alanine-86 in the tether region of the iron–sulfur protein of the cytochrome bc 1 complex affects the mobility of the [2Fe–2S ]Domain. Biochemistry 40: 327–335
Gutweniger HE, Grassi C and Bisson C (1983) Interaction between cytochrome c and ubiquinone–cytochrome c oxido-reductase: a study with water-soluble carbodiimides. Biochem Biophys Res Comm 116: 272–283
Hansen KC, Schultz BE, Wang F Chan SI (2000) Reaction of E. coli cytochrome bo 3 and mitochondrial cytochrome bc 1 with a photoreleasable decylubiquinol. Biochim Biophys Acta 1456: 121–137
Hong S, Ugulava N, Guergova-Kuras M and Crofts AR (1999) The energy landscape for ubihydroquinone oxidation at the Qo site of the bc 1 complex in Rhodobacter sphaeroides. J Biol Chem 274: 33931–33944
Hunte C, Koepke J, Lange C, Rossmanith T and Michel H (2000) Structure at 2. 3 Å resolution of the cytochrome bc 1 complex from the yeast Saccharomyces cerevisiae co crystallized with an antibody Fv fragment. Struct (London) Fold Des 8: 669–684
Hunte C, Solmaz S and Lange C (2002) Electron transfer between yeast cytochrome bc 1 complex and cytochrome c: a structural analysis. Biochim Biophys Acta 1555: 21–28
Iwata S, Lee JW, Okada K, Lee JK, Wata M, Rasmussen B, Link TA, Ramaswamy S and Jap BK (1998) Complete structure of the 11-subunit bovine mitochondrial cytochrome bc 1 complex. Science 281: 64–71
Jordan DB, Livingston RS, Bisaha JJ, Duncan KE, Pember SO, Picollelli MA, Schwartz RS, Sternberg JA and Tang XS (1999) Oxazolidinediones: a new chemical class of fungicides and inhibitors of mitochondrial cytochrome bc 1 function. Extended summaries: IUPAC Conference. Pestic Sci 55: 197–218
Kim H, Xia D, Yu C-A, Xia J-Z, Kachurin AM, Zhang L Yu L and Deisenhofer J (1998) Inhibitor binding changes domain mobility in the iron–sulfur protein of the mitochondrial bc 1 complex from bovine heart. Proc Natl Acad Sci USA 95: 8026–8033
King TE, Yu C-A, Yu L and Chiang Y (1975) An examination of the components, sequence, mechanism and their uncertainties involved in mitochondrial electron transport from succinate to cytochrome c. In: Quagliariello E, Papa S, Palmieri F, Slater EC and Sliprandi N (eds) Electron Transfer Chains and Oxidatitve Phosphorylation, pp 105–118. North-Holland Publishing, Amsterdam
Konig BW, Oshero. N, Wilms J, Muijsers AO, Dekker HL, and Margoliash E (1980) Mapping of the interaction domain for puri ed cytochrome c 1 on cytochrome c. FEBS Lett 111: 359–398
Kurisu G, Zhang H, Smith JL and Cramer WA (2003) Structure of the cytochrome b 6 f complex of oxygenic photosynthesis: tuning the cavity. Science 302: 1009–1014
Lange C and Hunte C (2002) Crystal structure of the yeast cytochrome bc 1 complex with its bound substrate cytochrome c. Proc Natl Acad Sci USA 99: 2800–2805
Li J, Osyczka A, Conover RC, Johnson MK, Qin H, Daldal F and Knaff DB (2003) Role of acidic and aromatic amino acids in Rb. capsulatus cytochrome c 1. A site-directed mutagenesis study. Biochemistry 42: 8818–8830
Link TA (1994) Two p K values of the oxidized Rieske [2Fe–2S ] cluster observed by CD spectroscopy. Biochim Biophys Acta 1185: 81–84
Marcus RA (1956) On the theory of oxidation–reduction reactions involving electron transfer. J Chem Phys 24: 966–978
Meinhardt SW and Crofts AR (1982a) The site and mechanism of action of myxothiazol as an inhibitor of electron transport in Rp. Sphaeroides FEBS Lett 149: 223–227
Meinhardt SW and Crofts AR (1982b) Kinetic and thermodynamic resolution of cytochrome c 1 and cytochrome c 2 from Rhodopseudomonas sphaeroides. FEBS Lett 149: 223–227
Mitchell P (1975) Proton motice redox mechanism of the cytochrome bc 1 complex in the respiratory chain: proton motive ubiquinone cycle. FEBS Lett 56: 1–6
Moore GR and Pettigrew GW (1990) Cytochromes c: Evolutionary, Structural and Physicochemical Aspects. Springer-Verlag, New York
Moser CC, Keske JM, Warncke K, Farid RS and Dutton PL (1992) Nature of biological electron transfer. Nature 355: 796–802
Nett JH, Hunte C and Trumpower BL (2000) Changes to the length of the flexible linker region of the Rieske protein impair the interaction of ubiquinol with the cytochrome bc 1 complex. Eur J Biochem 267: 5777–5782
Pelletier H and Kraut J (1992) Crystal structure of a complex between electron transfer partners, cytochrome c peroxidase and cytochrome c. Science 258: 1748–1755
Sadoski RC, Engstrom G, Tian H, Zhang L, Yu CA, Y, L, Durham B and Millett F (2000) Use of a photoactivated ruthenium dimer complex to measure electron transfer between the Rieske iron–sulfur protein and cytochrome c 1 in the cytochrome bc 1 complex. Biochemistry 39: 4231–4236
Shinkarev VP, Crofts AR and Wraight CA (2001) The electric eld generated by photosynthetic reaction center induces rapid reversed electron transfer in the bc 1 complex. Biochem-istry 40: 12584–12590
Slater EC and De Vries S (1980) Identification of the BAL-labile factor. Nature 288: 717–718
Smith HT, Ahmed A and Millett F (1981) Electrostatic interaction of cytochrome c with cytochrome c 1 and cyto-chrome oxidase J Biol Chem 256: 4984–4990
Snyder CH, Guierrez-Cirlos EB and Trumpower BL (2000) Evidence for a concerted mechanism of ubiquinol oxidation by the cytochrome bc 1 complex. J Biol Chem 275: 13555–13541
Speck SH, Ferguson-Miller S, Oshero. N and Margoliash E (1979) Definition of cytochrome c binding domains by chemical modi cation: kinetics of reaction with beef mitochondrial reductase. Proc Natl Acad Sci USA 76: 155–160
Stonehuerner J, OBrien P, Geren L, Millett F, Steidl J, Yu L and Yu C-A (1983) Identification of the binding site on cytochrome c 1 for cytochrome c. J Biol Chem 260: 5392–5398
Stroebel D, Choquet Y, Popot JL and Picot D (2003) An atypical heme in the cytochrome b 6 f complex. Nature 426: 413–418
Tian H, Yu L, Mather MW and Yu C-A (1998) Flexibility of the neck region of the Rieske iron–sulfur protein is functionally important in the cytochrome bc 1 complex. J Biol Chem 273: 27953–27959
Tian H, White S, Yu L and Yu C-A (1999) Evidence for the head domain movement of the Rieske iron–sulfur protein in electron transfer reaction of the cytochrome bc 1 complex. J Biol Chem 274: 7146–7152
Tian H, Sadoski R, Zhang L, Yu C-A, Yu L, Durham B and Millett F (2000) Definition of the interaction domain for cytochrome c on the cytochrome bc 1 complex. Steady-state and rapid kinetic analysis of electron transfer between cytochrome c and Rhodobacter sphaeroides cytochrome bc 1 surface mutants. J Biol Chem 275: 9587–9595
Trumpower BL (1981) Function of the iron–sulfur protein of the cytochrome bc 1 segment of the respiratory chain. Biochim Biophys Acta 639: 129–155
Trumpower BL (1990) The protonmotive Q cycle. Energy transduction by coupling of proton translocation to electron transfer by the cytochrome bc 1 complex. J Biol Chem 265: 11409–11412
Trumpower BL (2002) A concerted, alternating sites mechanism of ubiquinol oxidation by the dimeric cytochrome bc 1 complex. Biochim Biophys Acta 1555: 166–173
Trumpower BL and Gennis RB (1994) Energy transduction by cytochrome complexes in mitochondrial and bacterial respiration: the enzymology of coupling electron transfer reactions to transmembrane proton translocation. Annu Rev Biochem 63: 675–716
T'sai A, Olson JS and Palmer G (1983) The oxidation of yeast complex III: evidence for a very rapid electron equilibrium between cytochrome c 1 and the iron–sulfur center. J Biol Chem 258: 2122–2125
Ugulava NB and Crofts AR (1998) CD-monitored redox titration of the Rieske Fe–S protein of Rb. sphaeroides: pH dependence of the midpoint potential in isolated bc 1 complex and in membranes. FEBS Lett 440: 409–413
Valkova-Valchanova MB, Saribas AS, Gibney BR, Dutton PL and Daldal F (1998) Isolation and characterization of a two-subunit cytochrome b–c 1 subcomplex from Rhodobacter capsulatus and reconstitution of its ubihydroquinone oxidation (Qo )site with puri ed Fe–S protein subunit. Biochem-istry 37: 16242–16251
Wang K, Mei H, Geren L, Miller MA, Saunders A, Wang X, Waldner JL, Pielak GJ, Durham B and Millett F (1996) Design of a ruthenium cytochrome c derivative to measure electron transfer to the radical cation and the oxyferryl heme in cytochrome c peroxidase. Biochemistry 35: 15107–15119
Wang K, Zhen Y, Sadoski R, Grinnell S, Geren L, Ferguson-Miller S, Durham B and Millett F (1999) Definition of the interaction domain for cytochrome c on cytochrome c oxidase: II. Rapid kinetic analysis of electron transfer from cytochrome c to Rhodobacter sphaeroides cytochrome oxidase surface mutants. J Biol Chem 274: 38042–38050
Wikstrom MKF and Berden J (1972) Oxidoreduction of cytochrome b in the presence of antimycin. Biochim Biophys Acta 283: 403–420
Xia D, Yu C-A, Kim H, Xia J-Z, Kachurin AM, Zhang L Yu L and Desienhofer J (1997) Crystal structure of the cytochrome bc 1 complex from bovine heart mitochondria science 277: 60–66
Xiao K, Engstrom G, Rajagukguk S, Yu C-A, Yu L, Durham B and Millett F (2003) Effect of famoxadone on photo-induced electron transfer between the iron–sulfur center and cytochrome c 1 in the cytochrome bc 1 Complex. J Biol Chem 278: 11419–11426
Yu CA, Yu L and King TE (1973) Kinetics of electron transfer between cardiac cytochrome c 1 and c. J Biol Chem 248: 528–533
Zhang Z, Huang L, Shulmeister VM, Chi Y-I, Kim KK, Hung L-W, Crofts AR, Berry EA and Kim S-H (1998) Electron transfer by domain movement in cytochrome bc 1 Nature 392: 677–684
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Millett, F., Durham, B. Kinetics of Electron Transfer within Cytochrome bc 1 and Between Cytochrome bc 1 and Cytochrome c . Photosynthesis Research 82, 1–16 (2004). https://doi.org/10.1023/B:PRES.0000040434.77061.ca
Issue Date:
DOI: https://doi.org/10.1023/B:PRES.0000040434.77061.ca