Abstract
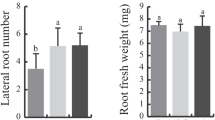
Multicellular organisms need to modulate proliferation and differentiation in response to external conditions. An important role in these processes plays the mitogen-stimulated induction of cyclin D (cycD) gene expression. D-type cyclins have been identified as the crucial intracellular sensors for cell-cycle regulation in all eukaryotes. However, cycD deletions have been found to cause specific phenotypic alterations in animals but not yet in plants. An insertional mutation of a so far uncharacterized Arabidopsis cycD gene did not alter the plant phenotype. To gain new insights into CycD function of land plants, we generated targeted cycD gene knockouts in the moss Physcomitrella patens and observed a surprisingly limited disruption phenotype. While wild-type plants reacted to exogenous glucose sources with prolonged growth of juvenile stages and retarded differentiation, cycD knockouts exhibited developmental progression independent of sugar supply. On the other hand, growth rate, cell sizes or plant size were not affected. Thus, we conclude that Physcomitrella CycD might not be essential for cell-cycle regulation but is important for coupling the developmental progression to nutrient availability.
Similar content being viewed by others
References
Bierfreund, N.M., Reski, R. and Decker, E.L. 2003. Use of an inducible reporter gene system for the analysis of auxin distribution in the moss Physcomitrella patens. Plant Cell Rep., published online 28 May 2003, http://dx.doi.org/10.1007/s00299-003-0646-1.
Burssens, S., Van Montagu, M. and Inzé, D. 1998. The cell cycle in Arabidopsis. Plant Physiol. Biochem. 36: 9–19.
Cockcroft, C.E., den Boer, B.G., Healy, J.M. and Murray, J.A.H. 2000. Cyclin D control of growth rate in plants. Nature 405: 575–579.
Cove, D.J. 1992. Regulation of development in the moss Physcomitrella patens. In: V.E.A. Russo, S. Brody, D. Cove and S. Ottolenghi, S. (Eds.) Development. The Molecular Genetic Approach, Springer, Heidelberg, pp. 179–193.
den Boer, G.W. and Murray, J.A.H. 2000. Triggering the cell cycle in plants. Trends Cell Biol. 10: 245–250.
Francis, D. 1998. Cell size and organ development in higher plants.In: D. Francis, D. Dudits and D. Inzé (Eds.) Plant Cell Division, Portland Press, London, pp. 187–206.
Gaudin, V., Lunness, P.A., Fobert, P.R., Towers, M., Riou-Khamlichi, C., Murray, J.A.H., Coen, E. and Doonan, J.H. 2000.The expression of D-cyclin genes defines distinct developmental zones in snapdragon apical meristems and is locally regulated by the cycloidea gene. Plant Physiol. 122: 1137–1148.
Geng, Y., Whoriskey, W., Park, M.Y., Bronson, R.T., Medema, R.H., Li, T., Weinberg, R.A. and Sicinski, P. 1999. Rescue of cyclin D1 deficiency by knockin cyclin E. Cell 97: 767–777.
Hohe, A., Rensing, S.A., Mildner, M., Lang, D. and Reski, R. 2002a. Day length and temperature strongly influence sexual reproduction and expression of a novel MADS-box gene in the moss Physcomitrella patens. Plant Biol. 4: 595–602.
Hohe, A., Decker, E.L., Gorr, G., Schween, G. and Reski, R. 2002b. Tight control of growth and cell differentiation in photoautotrophically growing moss Physcomitrella patens bioreactor cultures. Plant Cell Rep. 20: 1135–1140.
Holtorf, H., Guitton, M.-C. and Reski, R. 2002. Plant functional genomics. Naturwissenschaften 89: 235–249.
Hu, Y., Bao, F. and Li, J. 2000. Promotive effect of brassinosteroids on cell division involves a distinct CycD3-induction pathway in Arabidopsis. Plant J. 24: 693–701.
Imaizumi, T., Kadota, A., Hasebe, M. and Wada, M. 2002. Cryptochrome light signals control development to suppress auxin sensitivity in the moss Physcomitrella patens. Plant Cell 14: 373–388.
John, P., Mews, M. and Moore, R. 2001. Cyclin/Cdk complexes: their involvement in cell cycle progression and mitotic division. Protoplasma 216: 119–142.
Matsushime, H., Roussel, M.F., Ashmun, R.A. and Sherr, C.J. 1991. Colony-stimulating factor 1 regulates novel cyclins during the G1 phase of the cell cycle. Cell 65: 701–713.
Meijer, M. and Murray, J.A.H. 2000. The role and regulation of Dtype cyclins in the plant cell cycle. Plant Mol. Biol. 43: 621–633.
Oakenfull, E.A., Riou-Khamlichi, C. and Murray, J.A.H. 2002. Plant D-type cyclins and the control of G1 progression. Phil. Trans. R. Soc. Lond. B Biol. Sci. 357: 749–760.
Rensing, S.A., Rombauts, S., van de Peer, Y. and Reski, R. 2002. Moss transcriptome and beyond. Trends Plant Sci. 7: 535–538.
Reski, R. 1998a. Physcomitrella and Arabidopsis: the David and Goliath of reverse genetics. Trends Plant Sci. 6: 209–210.
Reski, R. 1998b. Development, genetics and molecular biology of mosses. Bot. Acta 111: 1–15.
Reski, R., Reynolds, S., Wehe, M., Kleber-Janke, T. and Kruse, S. 1998. Moss (Physcomitrella patens) expressed sequence tags include several sequences which are novel for plants. Bot. Acta 111: 143–149.
Riou-Khamlichi, C., Huntley, R., Jacqmard, A. and Murray, J.A.H. 1999. Cytokinin activation of Arabidopsis cell division through a D-type cyclin. Science 283: 1541–1544.
Riou-Khamlichi, C., Menges, M., Healy, J.M. and Murray, J.A.H. 2000. Sugar control of the plant cell cycle: differential regulation of Arabidopsis D-type cyclin gene expression. Mol. Cell Biol. 20: 4513–4521.
Rossi, V. and Varotto, S. 2002. Insights into the G1/S transition in plants. Planta 215: 345–356.
Roudier, F., Fedorova, E., Gyorgyey, J., Feher, A., Brown S., Kondorosi, A. and Kondorosi, E. 2000. Cell cycle function of a Medicago sativa A2-type cyclin interacting with a PSTAIREtype cyclin-dependent kinase and a retinoblastoma protein. Plant J. 23: 73–83.
Schaefer, D.G. 2001. Gene targeting in Physcomitrella patens. Curr. Opin. Plant Biol. 4: 143–150.
Schween, G., Fleig, S. and Reski, R. 2002. High-throughput-PCR screen of 15,000 transgenic Physcomitrella plants. Plant Mol. Biol. Rep. 20: 43–47.
Schween G., Gorr G., Hohe A., and Reski R. 2003. Unique tissuespecific cell cycle in Physcomitrella. Plant Biol. 5: 50–58.
Sherr, C.J. 1993. Mammalian G1 cyclins. Cell 73: 1059–1065.
Sherr, C.J. 1994. G1 phase progression: cycling on cue. Cell 79: 551–555.
Sherr, C.J. 1996. Cancer cell cycles. Science 274: 1672–1677.
Sicinski, P., Donaher, J.L., Parker, S.B., Li, T., Fazeli, A., Gardner, H., Haslam, S.Z., Bronson, R.T., Elledge. S.J. and Weinberg, R.A. 1995. Cyclin D1 provides a link between development and oncogenesis in the retina and breast. Cell 82: 621–630.
Soni, R., Carmichael, J.P. and Murray, J.A.H. 1993. Parameters affecting lithium acetate-mediated transformation of Saccharomyces cerevisiae and development of a rapid and simplified procedure. Curr. Genet. 24: 455–459.
Soni, R., Carmichael, J.P., Shah, Z.H. and Murray, J.A.H. 1995. A family of cyclin D homologs from plants differentially controlled by growth regulators and containing the conserved retinoblastoma protein interaction motif. Plant Cell 7: 85–103.
Strepp, R., Scholz, S., Kruse, S., Speth, V. and Reski, R. 1998. Plant nuclear gene knockout reveals a role in plastid division for the homolog of the bacterial cell division protein FtsZ, an ancestral tubulin. Proc. Natl. Acad. Sci USA 95: 4368–4373.
Swaminathan, K., Yang, Y., Grotz, N., Campisi, L. and Jack, T. 2000. Cyclin D1 provides a link between development and oncogenesis in the retina and breast. Plant Physiol. 124: 1658–1667.
Vandepoele, K., Raes, J., De Veylder, L., Rouzé, P., Rombauts, S. and Inzé, D. 2002. Genome-wide analysis of core cell cycle genes in Arabidopsis. Plant Cell 14: 903–916.
Vicente, O., Moreno, R.M.B. and Heberle-Bors, E. 1991. Pollen cultures as a tool to study plant development. Cell Biol. Rev. 25: 295–305.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lorenz, S., Tintelnot, S., Reski, R. et al. Cyclin D-knockout uncouples developmental progression from sugar availability. Plant Mol Biol 53, 227–236 (2003). https://doi.org/10.1023/B:PLAN.0000009295.07016.87
Issue Date:
DOI: https://doi.org/10.1023/B:PLAN.0000009295.07016.87