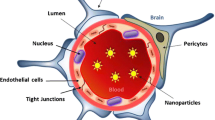
Abstract
Purpose. Two novel types of nanoparticles were evaluated as potential carriers for drugs across the blood–brain barrier (BBB).
Methods. Nanoparticles were composed of biocompatible materials including emulsifying wax (E. Wax) or Brij 72. Brij 78 and Tween 80 were used as surfactants for E.Wax nanoparticles (E78 NPs) and Brij 72 nanoparticles (E72 NPs), respectively. Both nanoparticle formulations were prepared from warm microemulsion precursors using melted E. Wax or Brij 72 as the oil phase. Nanoparticles were radiolabeled by entrapment of [3H]cetyl alcohol, and entrapment efficiency and release of radiolabel were evaluated. The transport of E78 and E72 NPs across the BBB was measured by an in situ rat brain perfusion method.
Results. Both formulations were successfully radiolabeled by entrapment of [3H]cetyl alcohol; ∼98% of radiolabel remained associated with nanoparticles at experimental conditions. The transfer rate (Kin) of E78 NPs from perfusion fluid into the brain was 4.1 ± 0.5 × 10-3 ml/s/g, and the permeability-surface area product (PA) was 4.3 ± 0.7 × 10-3 ml/s/g. The values for Kin and PA for E72 NPs were 5.7 ± 1.1 × 10-3 ml/s/g and 6.1 ± 1.4 × 10-3 ml/s/g, respectively.
Conclusions. For both nanoparticle types, statistically significant uptake was observed compared to [14C]sucrose, suggesting central nervous system uptake of nanoparticles. The mechanism underlying the nanoparticle brain uptake has yet to be fully understood.
Similar content being viewed by others
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Koziara, J.M., Lockman, P.R., Allen, D.D. et al. In Situ Blood–Brain Barrier Transport of Nanoparticles. Pharm Res 20, 1772–1778 (2003). https://doi.org/10.1023/B:PHAM.0000003374.58641.62
Issue Date:
DOI: https://doi.org/10.1023/B:PHAM.0000003374.58641.62