Abstract
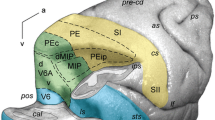
Experiments on three groups of rhesus macaques (intact and with bilateral removal of field 7 and the sulcus principalis) were performed to study the functional characteristics of the associative areas of the cortex while the monkeys learned visual discrimination. Significant differences in learning processes associated with removal of structures and the properties of the stimuli were seen in all animals, in the form of different types of learning curves. As compared with intact monkeys, removal of field 7 had no effect on learning processes for images with properties such as spatial frequency, color, and animal images, though there was a significant worsening in the characteristics of learning during visual discrimination of spatial relationships between objects. Learning processes became unstable, the number of peaks and troughs on learning curves increased, and as a result the training periods were significantly lengthened and 85% of the animals were unable to achieve the learning criterion. Removal of the sulcus principalis significantly worsened the characteristics of discrimination of the sizes of geometric objects, the spatial relationships between them, and stimuli of different colors. The stable reaction time and the probability of refusal in most cases also increased for monkeys of both these groups. Cluster analysis based on the quantitative characteristics of learning processes, despite individual differences between the monkeys, demonstrated a tendency for stimuli to be separated into classes corresponding to different types of information. These data show that the result of sensory processing is that several (at least three) functional visual information flows are generated and that different areas of the cortex deal with these different flows.
Similar content being viewed by others
REFERENCES
S. A. Aivazyan, V. M. Bukhshtaber, N. S. Enyukov, and L. D. Meshaklin, Applied Statistics [in Russian], Finansy i Statistika, Moscow (1989).
A. S. Batuev, G. P. Dem'yanenko, A. A. Orlov, and V. I. Shefer, Neuronal Mechanisms of the Conscious Monkey Brain [in Russian], Nauka, Leningrad (1986).
K. N. Dudkin, Visual Perception and Memory [in Russian], Nauka, Leningrad (1985).
K. N. Dudkin, V. K. Kruchinin, Yu. V. Skryminskii, and I. V. Chueva, Automated Methods for Studies of the Mechanisms of Behavior [in Russian], Nauka, Leningrad (1989).
K. N. Dudkin and I. V. Chueva, “Relationship between the characteristics of behavior in rhesus macaques and the properties of visual objects,” Ros. Fiziol. Zh. im. I. M. Sechenova, 81,No. 9, 25–34 (1995).
K. N. Dudkin, I. V. Chueva, F. N. Makarov, and I. V. Orlov, “Relationship between the characteristics of learning and the properties of visual objects in rhesus macaques with bilateral removal of field 7 of the parietal cortex,” Ros. Fiziol. Zh. im. I. M. Sechenova, 84,No. 3, 145–156 (1998).
K. N. Dudkin, I. V. Chueva, and F. N. Makarov, “The role of the prefrontal parietal areas of the cortex in learning and memory processes in monkeys,” Ros. Fiziol. Zh. im. I. M. Sechenova, 86,No. 11, 1458–1470 (2000).
I. P. Pavlov, Complete Collected Works [in Russian], Moscow, Leningrad (1951), Vol. 3, Books 1 and 2.
P. Bailey and C. von Bonin, The Neocortex of Macaca Mulatta, Urbana (1947).
S. M. Courtney, L. Petit, J. M. Maisog, L. G. Ungerleider, and J. V. Haxby, “An area specialized for spatial working memory in human frontal cortex,” Science, 279, 1347–1351 (1998).
J. Hyvarinen, The Parietal Cortex of Monkey and Man, Springer, Berlin (1982).
R. T. Knight, W. R. Staines, D. Swick, and L. L. Chao, “Prefrontal cortex regulates inhibition and excitation in distributed neural networks,” Acta Psychol. (Amst.), 101,No. 2–3, 159–178 (1999).
V. C. Matthew and P. S. Goldman-Rakic, “Matching patterns of activity in primate prefrontal area 8a and parietal area 7ip neurons during a spatial working memory task,” J. Neurophysiol., 79, 2919–2940 (1998).
Y. Miyashita and T. Hayashi, “Neural representation of visual objects: encoding and top-down activation,” Curr. Opin. Neurobiol., No. 10, 187–194 (2000).
V. B. Mountcastle, J. C. Lynch, and A. Georgopoulos, “Posterior parietal association cortex of the monkey: command functions of operation within extrapersonal space,” J. Neurophysiol., 38,No. 4, 871–908 (1975).
S. P. O'Scalaidhe, F. A. W. Wilson, and P. S. Goldman-Rakic, “Face-selective neurons during passive viewing and working memory performance of rhesus monkeys: evidence for intrinsic specialization of neuronal coding,” Cereb. Cortex, 9,No. 5, 459–475 (1999).
M. Petrides and D. N. Pandya, “Projections to the frontal cortex from the posterior parietal region in the rhesus monkey,” J. Comp. Neurol., 228, 105–116 (1984).
J. Quintana and J. Fuster, “Spatial and temporal factors in the role of prefrontal and parietal cortex in visuomotor integration,” Cereb. Cortex, 3, 122–132 (1993).
E. T. Rolls, “Brain mechanisms for invariant visual recognition and learning,” Behav. Processes, 33, 113–138 (1994).
M. Sakagami and K. Tsutsui, “The hierarchical organization of decision making in the primate prefrontal cortex,” Neurosci. Res., 34,No. 2, 79–89 (1999).
M. Sums, J. K. Hietanen, R. Hari, R. J. Ilmoniemi, and O. V. Lounasmaa, “Face-specific responses from the human inferior occipito-temporal cortex,” Neurosci, 77, 49–55 (1997).
K. Tanaka, “Mechanisms of visual object recognition: monkey and object human studies,” Curr. Opin. Neurobiol., No. 7, 523–529 (1997).
M. J. Webster, J. Bachevalier, and L. G. Ungerleider, “Connections of inferior temporal areas TEO and TE with parietal and frontal cortex in macaque monkeys,” Cereb. Cortex, 4,No. 5, 470–483 (1994).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Dudkin, K.N., Chueva, I.V. & Makarov, F.N. Functional Characteristics of the Associative Areas of the Cortex Involved in Visual Information Discrimination Learning Processes in Monkeys. Neurosci Behav Physiol 34, 83–96 (2004). https://doi.org/10.1023/B:NEAB.0000003251.76447.e5
Issue Date:
DOI: https://doi.org/10.1023/B:NEAB.0000003251.76447.e5