Abstract
Background: Although microemulsion-based nanoparticles (MEs) may be useful for drug delivery or scavenging, these benefits must be balanced against potential nanotoxicological effects in biological tissue (bio-nano interface). We investigated the actions of assembled MEs and their individual components at the bio-nano interface of thrombosis and hemolysis in human blood.
Methods: Oil-in-water MEs were synthesized using ethylbutyrate, sodium caprylate, and pluronic F-68 (ME4) or F-127 (ME6) in 0.9% NaClw/v. The effects of MEs or components on thrombosis were determined using thrombo-elastography, platelet contractile force, clot elastic modulus, and platelet counting. For hemolysis, ME or components were incubated with erythrocytes, centrifuged, and washed for measurement of free hemoglobin by spectroscopy.
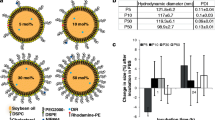
Results and conclusions: The mean particle diameters (polydispersity index) for ME6 and ME4 were 23.6 ± 2.5 nm (0.362) and 14.0 ± 1.0 nm (0.008), respectively. MEs (0, 0.03, 0.3, 3 mM) markedly reduced the thromboelastograph maximal amplitude in a concentration-dependent manner (49.0 ± 4.2, 39.0 ± 5.6, 15.0 ± 8.7, 3.8 ± 1.3 mm, respectively), an effect highly correlated (r2 = 0.94) with similar changes caused by pluronic surfactants (48.7 ± 10.9, 30.7 ± 15.8, 20.0 ± 11.3, 2.0 ± 0.5) alone. Neither oil nor sodium caprylate alone affected the thromboelastograph. The clot contractile force was reduced by ME (27.3 ± 11.1–6.7 ± 3.4 kdynes/cm2, P = 0.02, n = 5) whereas the platelet population not affected (175 ± 28–182 ± 23 106/ml, P = 0.12, n = 6). This data suggests that MEs reduced platelet activity due to associated pluronic surfactants, but caused minimal changes in protein function necessary for coagulation. Although pharmacological concentrations of sodium caprylate caused hemolysis (EC50 = 213 mM), MEs and pluronic surfactants did not disrupt erythrocytes. Knowledge of nanoparticle activity and potential associated nanotoxicity at this bio-nano interface enables rational ME design for in vivo applications.
Similar content being viewed by others
References
Amiji M. & K. Park, 1992. Prevention of protein adsorption and platelet adhesion on surfaces by PEO/PPO/PEO triblock copolymers. Biomaterials 13, 682–692.
Amiji M. & K. Park, 1993. Surface modification of polymeric biomaterials with poly(ethylene oxide), albumin, and heparin for reduced thrombogenicity. J. Biomater. Sci. Polym. Ed. 4, 217–234.
Bjoraker D.G., 1991. The thromboelastograph D coagulation analyzer. Anesthesiol. Rev. 18,34–40.
Carr M.E., 1995. Measurement of platelet force: The hemo dyne hemostasis analyzer. Clin. Lab. Manage. Rev. 9,312–318, 320.
Csordas A. & M. Rybczynska, 1988. Critical temperatures for the interaction of free fatty acids with the erythrocyte membrane. Biochim. Biophys. Acta. 944,155–163.
Danielson G.K., L.D. Dubilier & L.R. Bryant, 1970. Use of Pluronic F-68 to diminish fat emboli and hemolysis during cardiopulmonary bypass. A controlled clinical study. J. Thorac. Cardiovasc. Surg. 59,178–184.
Dietl K.H., 2002. Oral dosing of tacrolimus and cyclosporine microemulsion – results from a large multicenter study in renal transplantation. Transplant. Proc. 34,1659–1660.
Gore S.D., L.J. Weng W.D. Figg S. Zhai R.C. Donehower G. Dover M.R. Grever C. Griffin L.B. Grochow A. Hawkins, K. Burks Y. Zabelena & C.B. Miller, 2002. Impact of prolonged infusions of the putative differentiating agent sodium phenyl-butyrate on myelodysplastic syndromes and acute myeloid leukemia. Clin. Cancer Res. 8,963–970.
Jumaa M. & B.W. Muller, 2000. Lipid emulsions as a novel system to reduce the hemolytic activity of lytic agents: Mechanism of the protective effect. Eur. J. Pharm. Sci. 9,285–290.
Lapr’e J.A. D.S. Termont A.K. Groen & M.R. Van der, 1992. Lytic effects of mixed micelles of fatty acids and bile acids. Am. J. Physiol. 263,G333–G337.
Lowe K.C., B.A. Furmidge & S. Thomas, 1995. Haemolytic properties of pluronic surfactants and effects of purification. Artif. Cells Blood Substit. Immobil. Biotechnol. 23,135–139.
Morey T.E., S. Rajasekaran A.E. Martynyuk C.N. Seubert & D.M. Dennis, 2001. Intralipid attenuates bupivacaine-induced QRS prolongation and sodium channel block in guinea pig isolated heart and ventricular myocytes. Anesthesiology 95, A981.
Orringer E.P., J.F. Casella K.I. Ataga M. Koshy P. Adams-Graves L. Luchtman-Jones T. Wun M. Watanabe, F. Shafer A. Kutlar M. Abboud M. Steinberg B. Adler P. Swerdlow C. Terregino S. Saccente B. Files S. Ballas, R. Brown S. Wojtowicz-Praga & J.M. Grindel, 2001. Purified poloxamer 188 for treatment of acute vaso-occlusive crisis of sickle cell disease: A randomized controlled trial. JAMA 286, 2099–2106.
Padilla F., J.O. Wear & W.H. Van Wagner, 1975. Effect of fluorocarbon emulsions on the mechanical fragility of normal and sickle cells: In vitro studies. Fed. Proc. 34,1510–1512.
Park K., H.S. Shim M.K. Dewanjee & N.L. Eigler, 2000. In vitro and in vivo studies of PEO-grafted blood-contacting cardiovascular prostheses. J. Biomater. Sci. Polym. Ed. 11, 1121–1134.
Sakaeda T. & K. Hirano, 1998. Effect of composition on biological fate of oil particles after intravenous injection of O/W lipid emulsions. J. Drug Target 6, 273–284.
von Corswant C., P. Thoren & S. Engstrom, 1998. Triglyceride-based microemulsion for intravenous administration of sparingly soluble substances. J. Pharm. Sci. 87,200–208.
Zabka M. & M. Benkova, 1995. Microemulsions containing local anaesthetics. Part 6: Influence of microemulsion vehicle on in vivo effect of pentacaine. Pharmazie 50, 703–704.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Morey, T.E., Varshney, M., Flint, J.A. et al. Activity of microemulsion-based nanoparticles at the human bio-nano interface: concentration-dependent effects on thrombosis and hemolysis in whole blood. J Nanopart Res 6, 159–170 (2004). https://doi.org/10.1023/B:NANO.0000034625.30336.c1
Issue Date:
DOI: https://doi.org/10.1023/B:NANO.0000034625.30336.c1